CCP Score Emerges as a Prognostic Marker in Resected Stage I Lung Cancer
Resection of stage I lung adenocarcinoma can lead to 5-year survival rates of up to 87%, but whether adjuvant therapy can improve overall survival (OS) in this subset of patients is not yet clear.
Corey J. Langer, MD
Resection of stage I lung adenocarcinoma can lead to 5-year survival rates of up to 87%, but whether adjuvant therapy can improve overall survival (OS) in this subset of patients is not yet clear.1,2
The results of the CALGB 9633 study have suggested that patients with stage Ib lung adenocarcinoma whose tumors are >4 cm may benefit from adjuvant chemotherapy. Recommendations regarding the use of adjuvant therapy in this stage I subset, such as those of the National Comprehensive Cancer Network (NCCN), are therefore based on tumor size.3
Nonetheless, there is evidence that factors other than size alone, such as cribiform pattern in acinar predominant tumors, solid predominant histologic subtype, lymphatic vessel invasion, and tumor microenvironment may also be important determinants of prognosis in stage I tumors.4-6A recent report published inOncotargethas sought to identify a molecular expression signature that can be used to further stratify patients with stage Ia or Ib lung adenocarcinoma into high- or low-risk strata.1
KEY FINDINGS
The authors evaluated a cohort of patients with stage I lung adenocarcinoma who had undergone resection, using a 46-gene panel to assess cell cycle progression (CCP) genes, and a molecular prognostic score (mPS), which included the CCP score as well as stage. The evaluable population included 1103 patients, with a majority being women (61%), with a history of smoking (68.1%), and stage Ia disease (72.4%).
Results for lung cancerspecific mortality (LCSM), on multi-variate analysis, showed that CCP score was an independent and significant prognostic marker (HR, 1.6; P = .006), conferring a 1.6-fold increased risk per interquartile range for this endpoint; age at diagnosis, surgical procedure, tumor size, lymphatic in- vasion, and morphologic grade were also significant predictors of LCSM in the analysis.
mPS was also a significant predictor of 5-year LCSM, conferring an approximately 1.8-fold increased risk per interquartile range (HR, 1.77; P = .006); as with CCP, age at diagnosis, surgical procedure, tumor size, lymphatic invasion, and histological grade were also significant predictors of 5-year LCSM in the mPS analysis. A significant difference was observed for the endpoint of 5-year LCSM when a previously defined threshold of 27 was used to stratify the mPS into low mPS (<27; n = 614) and high mPS (>27; n = 489) cohorts (96% vs. 81%; P <.001), respectively.
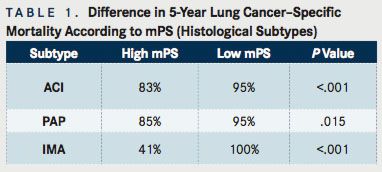
(TABLE 1)
Further analyses examined the endpoint of mortality according to histological subtype. The results showed worse 5-year LCSM rates for all histological subtypes, with high mPS compared to those with low mPS. For the subtypes of intermediate-grade acinar predominant (ACI), papillary predominant (PAP), and invasive mucinous adenocarcinoma (IMA), the difference between groups were all statistically significant
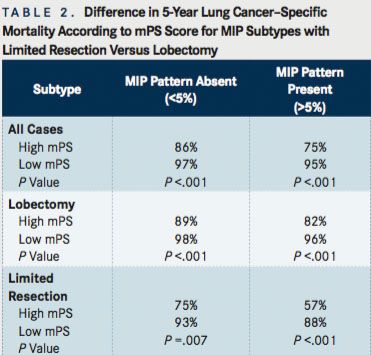
(TABLE 2)
Additional analyses were conducted for patients with micropapillary (MIP) subtype tumors, which are usually associated with a worse prognosis,7 who had undergone limited resection versus those undergoing lobectomy. The results again showed that patients with high mPS had signi cantly lower survival compared to those with low mPS (P <.001), inclusive of those with the presence (>5%) or absence (<5%) of the MIP pattern. The greatest difference was observed among patients with the MIP subtype who had undergone a limited resection, as compared to those undergoing lobectomy.
Exploratory analyses also investigated the impact of CCP and mPS on OS in this dataset. In multivariate analysis, it was found that CCP score (HR, 1.33; P = .014) was a significant prognostic marker for 5-year OS. Similarly, in multivariate analysis, mPS was also shown to be a significant prognostic marker for 5-year OS (HR, 1.41; P = .016). An association between mPS and the percentage of solid pattern in their tumors was also observed in the study; as the percentage of solid pattern increased, so too did the proportion of patients with high mPS score (P <.001).
In addition, whereas high mPS score was less commonly observed in tumors with epidermal growth factor receptor (EGFR) or KRAS mutations (35% and 30%, respectively) roughly equal proportions of wild-type mutational status patients had high and low mPS (48% and 52%, respectively).
CONCLUSIONS AND IMPLICATIONS
Data from this analysis, conducted in a large cohort of patients with resected stage I adenocarcinoma, have validated this prognostic signature, and show an association between both the CCP and mPS with 5-year LCSM. The authors concluded that use of this Clinical Laboratory Improvement Amendmentcertified assay may provide a more reproducible and quantitative means to assess clinical aggressiveness of stage I adenocarcinoma and determine the appropriateness of adjuvant chemotherapy in this population.
In a recent interview withTargeted Therapies in Oncology, Corey J. Langer, MD, professor of Medicine at the Perelman School of Medicine, and director of Thoracic Oncology at the Abramson Cancer Center at the University of Pennsylvania, cited the lingering uncertainties regarding the optimal treatment for stage I, node negative lung adenocarcinoma.
While he recognized the utility of gene signature assays such as the one described in this study, in that “they may be potentially prognostic, [and] may separate out those who are going to do really well, from those who are at much higher risk for relapse in early stage,” Langer also noted the need for ad- ditional studies.
“The big question is what do we do with these data,” he said. “One could envision, perhaps, studies in those with poor prognostic signatures, (comparing the standard of observation) to a platinum- based regimen. Then, perhaps, in those with good prognostic signatures, looking at lesser resections. Maybe we can get by with less than a lobectomy and do a sublobar resection or segmentectomy in that group. That would be a study I’d love to see, considering we’re talking about relatively small tumors that seem to have potentially more favorable genetic signatures.”
References:
- Eguchi T, Kadota K, Chaft J, et al. Cell cycle progression score is a marker for ve-year lung cancer-speci c mortality risk in patients with resected stage I lung adenocarcinoma. Oncotarget. 2016; 7(23): 35241-35256.
- Rami-Porta R, Bolejack V, Crowley J, et al. The IASLC lung cancer staging project: Proposals for the revisions of the T descriptors in the forthcoming eighth edition of the TNM classi cation for lung cancer. J Thorac Oncol. 2015; 10:990-1003.
- Strauss GM, Herndon JE 2nd, Maddaus MA, et al. Adjuvant paclitaxel plus carboplatin compared with observation in stage IB non-small-cell lung cancer: CALGB 9633 with the Cancer and Leukemia Group B, Radiation Therapy Oncol- ogy Group, and North Central Cancer Treatment Group Study Groups. J Clin Oncol. 2008; 26(31):5043-5051.
- Kadota K, Yeh YC, Sima CS, et al. The cribriform pattern identi es a subset of acinar predominant tumors with poor prognosis in patients with stage I lung adenocarcinoma: a conceptual proposal to classify cribriform predominant tumors as a distinct histologic subtype. Mod Pathol. 2014; 27(5):690-700.
- Ujiie H, Kadota K, Chaft JE, et al. Solid Predominant Histologic Subtype in Resected Stage I Lung Adenocarcinoma Is an Independent Predictor of Early, Extrathoracic, Multisite Recurrence and of Poor Postrecurrence Survival. J Clin Oncol. 2015; 33(26):2877-2884.
- Nitadori J, Bograd AJ, Kadota K, Sima CS, Rizk NP, Morales EA, Rusch VW, Travis WD and Adusumilli PS. Impact of micropapillary histologic subtype in selecting limited resection vs lobectomy for lung adenocarcinoma of 2cm or smaller. J Natl Cancer Inst. 2013; 105:1212-1220.

Rugo Surveys Peers on Treatment After Metastatic Relapse of HR+, HER2– Breast Cancer
April 20th 2024During a Case-Based Roundtable® event, Hope S. Rugo, MD, FASCO, moderated a discussion on treatment options for a patient whose breast cancer recurred several years after adjuvant therapy.
Read More