MRD Emerging as a Biomarker in Acute Leukemia
In results published in April in the New England Journal of Medicine, corresponding author Peter J. M. Valk, PhD, of the Department of Hematology, Erasmus University Medical Center, and colleagues found that MRD positivity was associated with a higher rate of relapse and a lower rate of relapse-free survival and overall survival for patients with newly diagnosed AML.
Propelled by strong results in clinical trials, minimal residual disease (MRD) has emerged as a biomarker for directing treatment and as a predictor for outcomes in patients with acute lymphoblastic leukemia (ALL) and acute myeloid leukemia (AML).
In results published in April in theNew England Journal of Medicine, corresponding author Peter J. M. Valk, PhD, of the Department of Hematology, Erasmus University Medical Center, and colleagues found that MRD positivity was associated with a higher rate of relapse and a lower rate of relapse-free survival (RFS) and overall survival (OS) for patients with newly diagnosed AML.1
Researchers obtained samples of bone marrow or peripheral blood from 482 patients with previously untreated AML (n = 428) or refractory anemia with excess of blasts collected from 2001 to 2013. Patients had to be in either complete remission (CR) or CR with incomplete hematologic recovery (CRi), with less than 5% blast cells in the bone marrow, after receiving 2 cycles of induction chemotherapy.
The investigators used multiparameter flow cytometry (MFC) and targeted next-generation sequencing (NGS) at diagnosis to detect mutations that could serve as a marker of residual disease. They detected mutations in 54 genes commonly found in patients with hematologic cancers. Overall, 430 patients (89.2%) were positive for at least 1 such mutation.
Compared with no detection, the detection of molecular MRD positivty was associated with a significantly higher relapse rate (55.4% vs 31.9%; HR, 2.14; P <.001) and lower rates of RFS (36.6% vs 58.1%; HR for relapse or death, 1.92; P <.001) and OS (41.9% vs 66.1%; HR, 2.06; P <.001).
The 430 patients who were positive for at least 1 mutation were randomly assigned to either a training cohort (n = 283) or a validation cohort (n = 147). In the training cohort, the detection of any persistent mutation during CR was associated with an increased risk for relapse. At 4 years, the relapse rate was 48.2% with detection compared with 32.4% with no detection (P = .03). Investigators found that the correlation of persistent mutations with an increased relapse risk appeared to be independent of allele frequency.
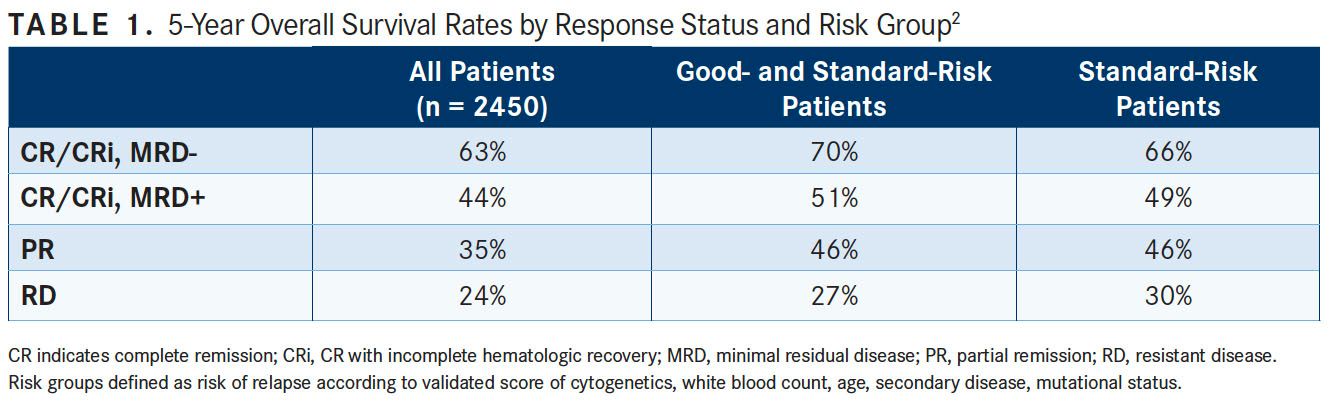
In late March, the Journal of Clinical Oncology published results from the NCRI AML17 trial of 2450 adults older than 60 years with AML or high-risk myelodysplastic syndrome who underwent prospective MFC-MRD assessment. Investigators hoped to determine the extent of the predictive value of MRD status following each course of induction chemotherapy.2
Investigators categorized responses after course 1 (C1) as resistant disease (RD), partial remission (PR), and CR or CRidefined as absolute neutrophil count <1000/μL or thrombocytopenia <100,000/ μL). CR/CRi was further subdivided by MFC-MRD assay into MRD-positive or MRD-negative status.
Patients with favorable or standard risk, including NPM1 wild-type patients, received a second induction of daunorubicin/cytosine arabinoside. Treatment in course 2 (C2) was intensified for patients with high-risk factors.
Among patients had achieved a CR post C1, the presence of MRD data was associated with secondary AML, and the absence of an NPM1 mutation. Five-year OS survival was 52% in patients with MRD data versus 50% in those without. In adjusted analyses, the presence of MRD data was not associated with survival (HR, 0.99; 95% CI, 0.84-1.16; P = .9).
Five-year OS from PR and MRD-positive responses after C1 were similar, particularly for good- to standard-risk subgroups (RD, 27%; PR, 46%; CR/CRi and MRD-positive, 51%; CR/CRi and MRD-negative, 70%; P <.001). Adjusted analyses confirmed significant OS differences in the good- to standard-risk subgroups between RD and PR/MRD-positive but not between PR and MRD positive (TABLE 1).
Compared with those who had CR, MRD-positive patients who had CRi after C1 had poorer OS rates (19% vs 45%; P = .001), with a smaller effect after C2. Investigators found that the prognostic effect of C2 MFC-MRD status remained significant when adjusting for C1 response, for both relapse (HR, 1.88; 95% CI, 1.50-2.36; P <.001) and OS (HR, 1.77; 95% CI, 1.41-2.22; P <.001).
MRD positivity appeared to be less discriminatory in poor-risk patients by stratified analyses. For the NPM1 wild-type standard-risk subgroup, MRD-positive status following C2 was significantly associated with poorer outcomes compared with MRD-negative status for OS (33% vs 63%; P = .003).
MRD-positive patients also derived greater benefit from stem cell transplantation compared with those who were MRD-negative (HR, 1.68; 95% CI, 0.75-3.85; P = .16 for interaction).
“MFC-MRD can improve outcome stratification by extending the definition of partial response after first induction and may help predict NPM1 wild-type standard-risk patients with poor outcome who benefit from transplant in the first CR,” wrote Sylvie D. Freeman, MD, PhD, of Birmingham Medical School, and colleagues.
Freeman et al went on to write that, to date, there has been no accepted approach to distinguish those patients likely to be cured with chemotherapy alone from those whose response is likely to be successful after salvage therapy following relapse, if they do relapse, or from patients those who would benefit from transplantation in first remission.
“Our results suggest that allogeneic transplant in first remission could be directed to those who are MRD-positive rather than MRD-negative,” they wrote. “This is the first indication that MRD status might have utility in directing therapy for NPM1 wild-type patients at standard risk despite their molecular heterogeneity. Large patient datasets likely requiring collaborative efforts will determine whether integrating MFC-MRD status with genomic profiles further informs outcome prediction.”
THE PROGNOSTIC POWER OF MRD DRIVES DRUG DEVELOPMENT
In March, the FDA granted an accelerated approval to blinatumomab (Blincyto) for the treatment of adult and pediatric patients with B-cell precursor (BCP) ALL who are in remission but still have MRD. The decision made blinatumomab the first FDA-approved treatment for patients specifically with MRD-positive ALL, and the first and only approved therapy for MRD. The FDA’s Oncologic Drugs Advisory Committee had previously recommended the approval based on results from the phase II BLAST study. In BLAST, blinatumomab induced a nearly 80% complete MRD response rate in patients with MRD-positive ALL who were in hematologic CR.3
“Because patients who have MRD are more likely to relapse, having a treatment option that eliminates even very low amounts of residual leukemia cells may help keep the cancer in remission longer,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in a statement upon announcement of the approval.
BLAST was a single-arm trial of up to 4 cycles of blinatumomab for treatment of patients with B-cell precursor ALL in CR or CR with partial platelet recovery and MRD >0.1%. The primary efficacy endpoint of BLAST was complete MRD response, defined as absence of detectable MRD using an assay with a sensitivity <0.01% after 1 cycle of blinatumomab.
Of 116 patients who received at least 1 dose of blinatumomab, the FDA identified 87 in CR with hematologic recovery and baseline MRD >0.1%, including 61 in first CR, 25 in second CR, and 1 in third CR. Sixty-nine patients (79.3%; 95% CI, 70.4- 87.6) achieved a complete MRD response within the first cycle.
The full analysis set from BLAST included 113 patients who were determined to be MRD-positive, based on a measurement of ≥0.1% from an assay with a minimum sensitivity of 0.01% after ≥2 weeks from their last chemotherapy treatment. Among the evaluable patients in this group, 77.9% (95% CI, 69.1%-85.1%) achieved MRD CR within the first cycle. Two additional patients achieved MRD CR after the second cycle.
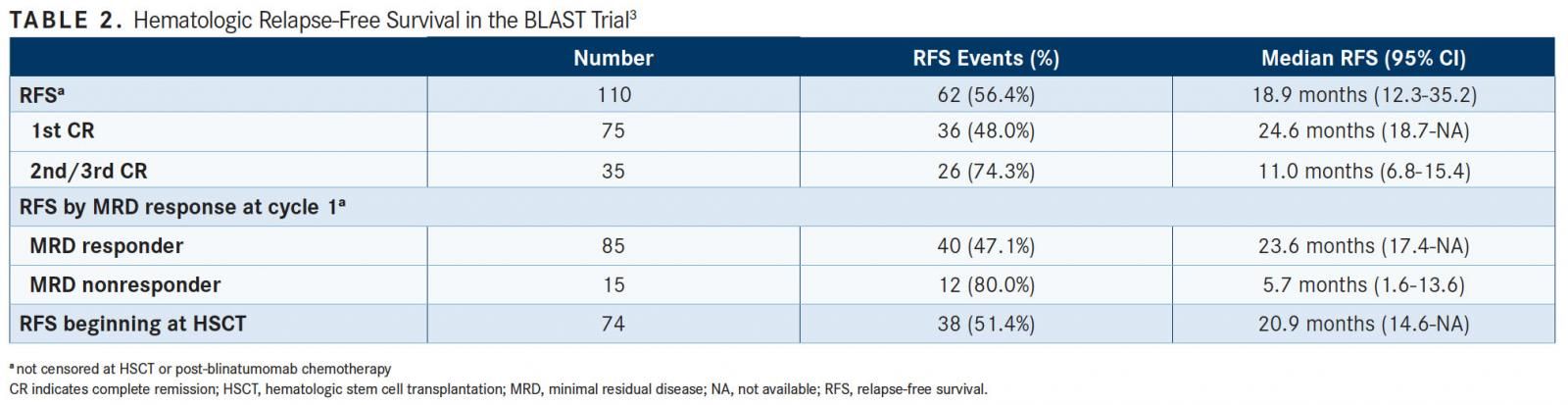
Using the 87-patient FDA efficacy analysis set, the 18-month RFS rate was 56% and the estimated median RFS was 22.3 months. The estimated median RFS time in first CR at the time of treatment with blinatumomab was 24.6 months (95% CI, 18.7-not applicable), and the median RFS time in the second or third CR was 11.0 months (95% CI, 6.8-15.4) (TABLE 2). RFS was numerically longer for patients in first CR than for those in the second or third CR.
The estimated median RFS time was 23.6 months (95% CI, 17.4-not applicable) for patients with a complete MRD response and 5.7 months (95% CI, 1.6-13.6) for MRD nonresponders.
A propensity score analysis for the patients in first remission, with or without hematopoietic recovery, in BLAST and in Study 20120148 demonstrated that the RFS for patients treated with blinatumomab was significantly greater than in historical controls. Study 20120148 was a retrospective cohort study investigating the hematological RFS and OS in adult patients with Philadelphia chromosomenegative BCP ALL in hematological CR with MRD.
The estimated median RFS time with propensity score weighted analyses was 35.2 months (95% CI, 24.2-not estimable) for the blinatumomab group and 8.3 months (95% CI, 6.23-11.9) for the control group (log-rank P <.0001).
MRD ONLY AS GOOD AS ITS ASSAY
As noted by Roland B. Walter, MD, PhD, MS, an associate member in the Clinical Research Division, Fred Hutchinson Cancer Research Center, in an editorial accompanying the findings from Freeman et al, results from MRD tests are not perfectly predictive. Even without subsequent therapy, not every patient who is MRD-positive will relapse and not every patient who is MRD-negative will be cured, even with standard therapy.4
“The prognostic significance of any MRD test result critically depends on the assay characteristics,” he wrote. “Current assays are neither standardized nor harmonized, which is perceived as [an] important hurdle for the embracement of MRD-based response definitions. Even though work toward correcting this deficiency is ongoing, methodologies continue to evolve rapidly, and it will take time before mature assays become available with reproducible operating characteristics when implemented across laboratories or institutions.”
It’s possible that using both NGS and MFC to determine MRD status will deliver the best outcomes.
Although Freeman et al observed distinct prognostic groups for 5-year OS after incorporating MFC-MRD with established response criteria of PR and RD, Valk et al concluded that the detection of residual leukemia using both methods was associated with an excessively high probability (approximately 75%) of relapse. Furthermore, the absence of detection of residual disease with both methods correlated with a relatively low probability of relapse (approximately 25%).
“In this study, gene sequencing and multiparameter flow cytometry each had independent and additive prognostic value with respect to rates of relapse and survival in patients with AML,” Valk and colleagues wrote. “Thus, the combined use of sequencing and flow cytometry during complete remission warrants further development and evaluation in clinical practice.”
More research will help to further clarify the role of MRD status in AML and ALL and the best method for testing MRD that can be used to guide treatment.
References:
- Jongen-Lavrencic M, Grob T, Hanekamp K, et al. Molecular minimal residual disease in acute myeloid leukemia. N Engl J Med. 2018;378(13):1189-1199. doi: 10.1056/NEJMoa1716863.
- Freeman SD, Hills RK, Virgo P, et al. Measurable residual disease at induction redefines partial response in acute myeloid leukemia and stratifies outcomes in patients at standard risk without NPM1 mutations [published online March 30, 2018]. J Clin Oncol. doi: 10.1200/JCO.2017.76.3425.
- FDA briefing document: Oncologic Drugs Advisory Committee Meeting: BLA 125557 S-013 Blincyto (blinatumomab) Applicant: Amgen, Inc. FDA website. www. fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/ OncologicDrugsAdvisoryCommittee/UCM599298.pdf. Accessed March 29, 2018.
- Walter RB. Minimal residual disease testing after induction chemotherapy for acute myeloid leukemia: moving beyond prognostication? [published online March 30, 2018]. J Clin Oncol. doi: 10.1200/JCO.2018.78.3266.

Biomarker Testing Paves the Way for Better Targeted Therapies in NSCLC
April 16th 2024At a live virtual event, Edward S. Kim, MD, MBA, discussed the evolving landscape of biomarker testing before making treatment decisions for patients with early-stage non–small cell lung cancer (NSCLC).
Read More
Creating Solutions for a 'Continual State of Transition' in Cancer Care
April 15th 2024In a Peers & Perspectives in Oncology feature article, we focus on the importance of the transition-of-care process for patients with cancer as they move from the inpatient to outpatient setting, as well as between lines of therapy with comments from Marc J. Braunstein, MD, PhD, and Michael Shusterman, MD.
Read More