Current Approaches to Diagnosis and Risk Stratification in Chronic Lymphocytic Leukemia
<p>CLL is a malignancy characterized by the clonal proliferation and accumulation of small, mature-appearing CD5-positive B lymphocytes in the blood, bone marrow, and secondary lymphoid tissues.</p>
Chronic lymphocytic leukemia (CLL) is a malignancy characterized by the clonal proliferation and accumulation of small, mature-appearing CD5-positive B lymphocytes in the blood, bone marrow, and secondary lymphoid tissues.1A CLL diagnosis is established by the presence of more than 5x109/L peripheral lymphocytes co-expressing CD5, CD19, and CD23, and weakly expressing CD20, CD79b, and surface immunoglobulin.2Small lymphocytic lymphoma (SLL), in which the same leukemic cell population is mostly restricted to the bone marrow and lymphoid tissues, represents a clinical variant of CLL and is similarly managed.3
CLL is the most common adult leukemia in Western countries. Its incidence increases with age, and with aging populations, its prevalence and mortality in Western countries will continue to rise. Improved diagnostic methods and more frequent blood testing have also led to increasing identification of early-stage CLL among younger patients.2,4Approximately 10% to 15% of patients with CLL are younger than 55 years.2,4
Recent years have seen major advances in diagnostic approaches in CLL, and disease assessment may now include analysis of multiple genetic mutations in addition to recurrent cytogenetic changes.1Furthermore, with the development of novel treatment approaches, the significance of prognostic markers is shifting. Awareness of current approaches to CLL diagnosis and assessment of disease criteria relevant to current risk stratification and treatment selection strategies is therefore prerequisite to tailor treatment for each patient.
EPIDEMIOLOGY AND RISK FACTORS
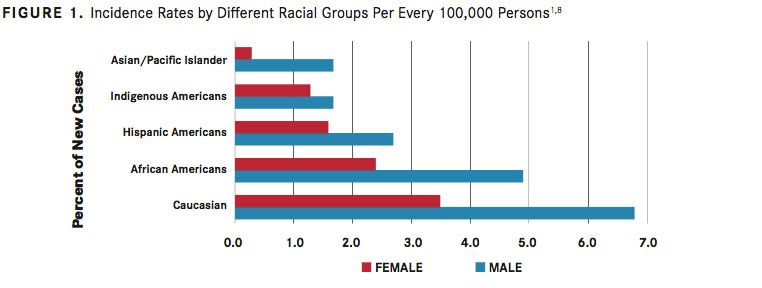
The incidence of CLL varies between geographic locations and is lower in Eastern Asian populations, such as China, Korea, and Japan, than in Western countries.1Lower incidence of CLL is maintained in Asian individuals emigrating to Western countries and their progeny.5With an age-adjusted incidence of approximately 4 per 100,000 inhabitants in Europe and the United States, CLL is the most common adult leukemia in adults in Western countries.2In the United States, CLL accounts for approximately one-third of all annual new leukemia diagnoses (20,110 of 62,130 in 2017).6,7According to estimates based on the US National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database, CLL is most frequent in white populations in the United States (6.8 per 100,000 men and 3.5 per 100,000 women), followed by African Americans (4.9 per 100,000 men and 2.4 per 100,000 women).1,8Rates are lower among Hispanic Americans (2.7 per 100,000 men and 1.6 per 100,000 women), Indigenous Americans (1.7 per 100,000 men and 1.3 per 100,000 women), and individuals of Asian or Pacific Island descent (1.7 per 100,000 men and 0.3 per 100,000 women) (FIGURE 11,8).
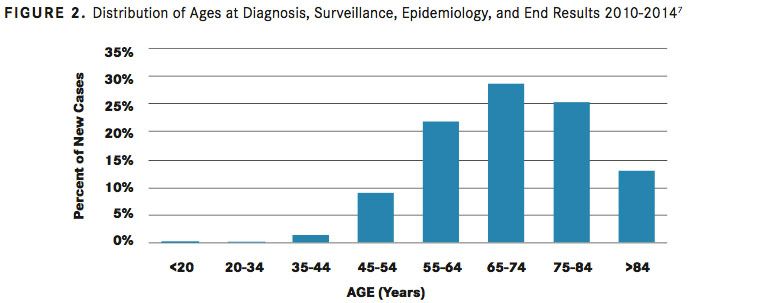
The risk for CLL increases with age (FIGURE 27). More than 70% of patients are older than 65 years at diagnosis, the median age at diagnosis is 72 years, and the incidence rate increases to >30:100,000/year at an age of more than 80 years.2Men are at an approximately 2-fold higher risk for developing CLL than women (male:female ratio of 1.9).6
A genetic contribution to CLL susceptibility has been clearly established. Family members of patients with CLL have an 8.5-fold increased risk for developing the disease compared with the general population.9,10 Genome-wide association studies have identified polymorphisms in more than 25 loci associated with familial CLL, including candidate genes involved in B-cell biology, apoptotic pathways, and regulatory microRNAs that may contribute to disease development.1114
Studies in veterans have established exposure to Agent Orange as an environmental risk factor for CLL.15Insecticide exposure and farming history have also been associated with a higher risk for developing CLL,16whereas blood transfusions and ionizing radiation have not.17,18
The precursor state to CLL, characterized as monoclonal B-cell lymphocytosis (MBL), is defined by the presence of less than 5000 monoclonal B cells, in the absence of lymphadenopathy, organomegaly, cytopenia, and clinical symptoms.2,19
Risk factors for MBL include increasing age and genetic disposition, including overlapping polymorphisms with those identified in CLL.19Population studies have shown that MBL is characterized by a bimodal distribution of clonal B cell counts that can be cat- egorized as low-count and high-count MBL (less than or equal to or greater than 0.5x109/L clonal B cells, respectively). Low-count MBL rarely progresses to CLL, whereas the annual rate of progression from high-count MBL to CLL requiring therapy is 1% to 2%.20
EVOLVING METHODS OF CLL DIAGNOSIS
Many patients with CLL are asymptomatic and diagnosis follows the detection of lymphocytosis in a routine complete blood count (CBC), ie, above the normal adult upper limit of approximately 3500 cells per μL, typically ≥10,000 cells per μL.1If disease symptoms are present, they may include fatigue, involuntary weight loss, excessive night sweats, abdominal fullness with early satiety, increased frequency of infections, and symptoms of an autoimmune cytopenia, as well as enlarged lymph nodes, hepatomegaly, and splenomegaly, which can be detected by palpation.1
Essential components of the CLL diagnosis include blood immunophenotyping by low cytometry and uorescence in situ hybridization (FISH), lymph node biopsy if a peripheral analysis is not sufficient, and absolute B monoclonal B lymphocyte counts.3The initial workup should include a physical exam with palpation of node-bearing areas, determination of liver and spleen size, performance status, complete blood count, differential blood counts, platelets, and a metabolic panel.3
Immunophenotyping by low cytometry is required to establish CLL diagnosis based on cell identity, clonality, and quantity (<5x109/L peripheral CLL cells).2,3 CLL cells co-express CD5, CD19, and CD23 antigens, weakly express CD20, CD79b, and surface immunoglobulin, with each clone restricted to expression of either kappa or lambda immunoglobulin light chains, and are negative for cyclin D1. Flow cytometric analysis for these markers is therefore used to establish a differential diagnosis from other B-cell lymphoproliferative disorders such as marginal zone lymphoma, lymphoplasmacytic lymphoma, and mantle cell lymphoma (MCL), all of which express B-cell surface antigens, but usually do not express CD23 and have negative or low CD43 expression.2,3MCL cells express CD5 but also exhibit enhanced expression of the gene encoding cyclin D1 (CCND1) due to the (11;14) translocation (t[11;14]), which is absent from CLL cells.1As such, FISH analysis for t(11;14) can help to distinguish MCL from CLL.
CLL cells in blood smears are small, mature lymphocytes with a narrow rim of cytoplasm and a dense nucleus without detectable nucleoli and with partially aggregated chromatin. Smudged cells, also known as Gumprecht nuclear shadows, are also mor- phologically characteristic for CLL.4Larger, atypical lymphocytes, cleaved cells, or prolymphocytes may be seen, but if the latter exceed 55%, prolymphocytic leukemia needs to be considered.1,21
Bone marrow biopsy is not considered essential for the diagnostic workup in CLL but may be needed if immunophenotyping results are unclear. According to the International Workshop of Chronic Lymphocytic Leukemia (iwCLL) 2008 guidelines for the diagnosis and treatment of patients with CLL, a bone marrow asipirate and biopsy may be desirable in both practice and prior to enrollment in a clinical trial (TABLE 1).21If a biopsy is performed, 4 different patterns of infiltration may be observed, which are nodular, interstitial, mixed nodular/interstitial, or diffuse (advanced disease).1Imaging using computed tomography (CT) scans can be beneficial for assessing the tumor load, monitoring disease progression, and determining the effects of investigational treatments in clinical trials, but it’s not recommended in asymptomatic patients or for staging.2,3
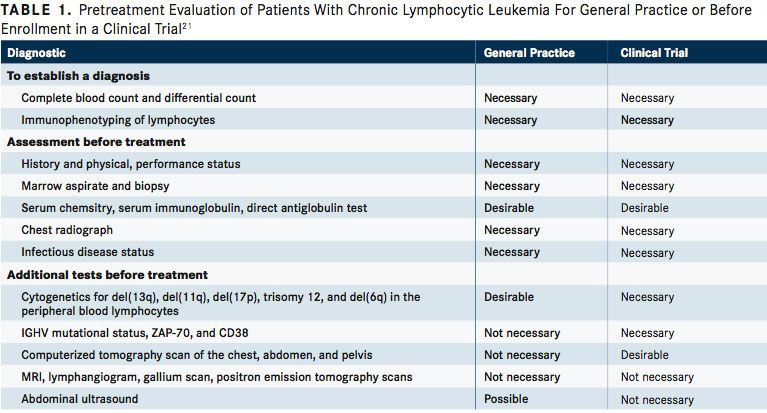
More than 80% of patients with previously untreated CLL have cytogenetic abnormalities, most commonly a deletion in chromosome 13q14.3 (del[13q]; 55%), followed by del(11q) (18%), trisomy 12 (16%), and (del[17p]) (7%), and del(6q) (6%).22Recommended analyses include interphase cytogenetic analysis with FISH for the detection of the del(17p), which affects p53 expression, and if negative, molecular genetics is recommended for the detection of a TP53 mutation.2,3 While FISH testing for cytogenetic abnormalities is suggested for pretreatment evaluation by the iwCLL 2008 guidelines, the guidelines especially recommend these tests in patients being enrolled in clinical trials.21
Molecular genetic analysis should also include mutational status of the immunoglobulin heavy chain variable region gene (IGHV). Additional analyses considered informative for prognosis and treatment selection are stimulated metaphase analyses to identify complex karyotypes, extended FISH for additional cytogenetic abnormalities, and polymerase chain reaction or sequencing for mutations in NOTCH1, SF3B1, TP53, or MYD88.2,3
CLL STAGING
Two clinical staging systems, the Rai and Binet systems, are used predominantly in the United States and Europe, respectively, to group patients with CLL into broad prognostic groups.23,24Both systems combine the presence of specific physical parameters, such as lymph node involvement, enlarged spleen and/or liver and blood parameters, including anemia or thrombocytopenia, to determine tumor burden.
The Rai system, originally including 5 groups, has been modified to define low-risk disease (former stage 0) as lymphocytosis with leukemia cells in the blood and/or marrow. Intermediate risk disease (stage I/II) is defined as enlarged nodes in any site, and splenomegaly and/or hepatomegaly (lymph nodes being palpable or not), and high-risk disease (stage II/IV) as lymphocytosis and cytopenia (hemoglobin [Hb] level less than 11 g/dL and/ or platelet count of less than 100,000/μL) (TABLE 23,23).
The Binet staging system relies on determining the number of involved areas, ie, enlarged lymph nodes of greater than 1 cm in diameter or organomegaly, and the presence anemia or thrombocytopenia. Binet stages are low risk (stage A), with less than 3 palpable enlarged sites without cytopenia; intermediate risk (stage B), with 3 or more palpable enlarged sites without cytopenia, and high risk (stage C) in all patients who have Hb of less than 10 g/dL and/or a platelet count of less than 100,000/μL, irrespective of organomegaly (TABLE 3 24).
Available survival estimates linked with staging suggest similar survival to age-matched controls for patients with low-risk disease by Rai stage (median, 150 months), shorter survival for patients with intermediate-risk disease (median, 71-101 months), and poor survival for high-risk features (median, 19 months).1,3However, these estimates reflect treatment with chemotherapy or chemoimmunotherapy, and life expectancies are increasing with newer small molecule inhibitor-based therapy.1,3
PROGNOSTIC MARKERS IN CLL
Newly diagnosed CLL is characterized by a highly variable clinical course, ranging from absence of symptoms for decades to the rapid development of symptoms or features of high-risk disease. Prognostic factors used for patient strati cation include patient factors and clinical features of the disease, and genetic, molecular, and biochemical characteristics of the CLL clone.3
In addition to staging and lymphocyte doubling time, traditional prognostic factors or clinical features associated with poorer outcome are male sex, ≥65 years, poor performance status due to medical comorbidities, high serum levels of beta-2 microglobulin (>3.5 mg/L), high absolute lymphocyte count (>50,000 cells/μL), and/or late-stage disease at diagnosis.2527Elevated serum β2 microglobulin is an independent prognostic indicator for treatment-free interval, response to treatment, and overall survival (OS) in response to first-line chemoimmunotherapy regimens,3,26andif remaining after 6 months of treatment—for inferior progres- sion-free survival (PFS) with ibrutinib (Imbruvica)-based therapies.28


The use of these diagnostic tests is encouraged by current treatment guidelines and managed care companies. John L. Fox, MD, MHA, the vice president of medical affairs of Priority Health, explained in an interview with the American Journal of Managed Care®, “The cost of the diagnostics themselves are really not an issue. Those are the upfront costs in trying to establish what the most appropriate therapy is for that particular patient.”29
Biological prognostic markers that reflect CLL cell characteristics and are used for risk stratification include cytogenetic abnormalities; IGHV mutational status; TP53 mutation; expression of ZAP-70, CD49d (also known as integrin alpha-4), or CD38; and mutations in NOTCH1, SF3B1, BIRC3, and MYD88.3Among these, del(17p) and TP53 mutations are considered pertinent for treatment selection, and multiple other markers are considered of predictive value.
Del(17p), which causes the loss of 1 TP53 allele and is associated with inactivating mutations in the other allele in 80% of patients with CLL, is considered the most important prognostic marker in CLL, with predictive value on treatment. The presence of del(17p) and/or mutations in TP53 defines a group of patients with CLL not eligible for chemoimmunotherapy regimens due to poor outcomes.22,30Targeted inhibitors of the B-cell receptor pathway and apoptosis inhibitors can produce high response rates in patients with del(17p) and are the preferred regimens in the first and subsequent lines of therapy.3The deletion is relatively rare at diagnosis (approximately 7%)22but frequent in relapsed/refractory disease, indicating acquisition or expansion of del(17q)-harboring CLL clones during treatment.31
Del(11q) has been linked to extensive lymphadenopathy, aggressive clinical course, and shorter median survival with traditional reg- imens (79 months).22However, the presence of del(11q) is not an adverse prognostic marker for response for treatment with ibrutinib.32
Among cytogenetic abnormalities, del(13q) is the only one that confers a favorable prognosis and long median survival (133 months).22Trisomy 12 has been linked to shorter survival.22
Unmutated IGHV (de ned as ≥98% homology with the germline gene sequence) re ects CLL originating from B cells that have not undergone a somatic mutation. The presence of unmutated IGHV predicts a more aggressive disease type and has traditionally been associated with significantly decreased survival compared with mutated IGHV, irrespective of disease stage.33,34Recent analyses have con rmed unmutated IGHV as a predictor of shorter survival with chemoimmunotherapy regimens including udarabine and rituximab (Rituxan; FR), and udarabine, chlorambucil, and rituximab (FCR).35,36In patients with mutated IGHV, FCR was associated with an improved survival in all cytogenetic groups except del(17p).36Unmutated IGHV does not predict adverse outcomes with ibrutinib-based regimens.37
Among prognostic surface markers detected by flow cytometry or immunohistochemistryincluding CD38, CD49d, and ZAP-70—CD49d is independent of FISH and IGHV.38Multiple studies have associated CD38 and/or ZAP-70 expression with shorter PFS and OS, but despite positive correlations with the presence of unmutated IGHV, these markers are not considered appropriate surrogate markersthe latter due to discordance and variable expression during the course of disease.3Expression of ZAP-70 may be a stronger predictor of time until treatment is needed than IGHV mutational status or CD38 levels39,40; however, standardized detec- tion of this nuclear marker is hampered by technical issues and it’s use is not yet recommended outside clinical trials.3
NOTCH1, SF3B1, and BIRC3 mutations are observed in approximately 4% to 15% of patients with newly diagnosed CLL, and 15% to 25% of patients with CLL refractory to udarabine.3These mutations have been associated with varying prognostic significance across studies, such that their impact on treatment selection remains to be established, particularly for targeted agents.
Del(17p) and TP53 mutations are currently the only disease-based predictive markers that affect treatment selection in CLL. With changing treatment options, particularly the inclusion of novel effective agents that prolong survival and have activity among patients considered high-risk in the era of chemotherapy-based regimens, the value of other prognostic markers continues to evolve.
As Steven E. Coutre, MD, professor of medicine (hematology) at the Stanford University Medical Center, summarized in an interview: “A FISH test from the peripheral blood identifies patients who are at higher risk of progression and, in some cases like with del(17p), identifies patients who respond differently to different therapies. And so, those are the kinds of insights that we’re looking for to help us better choose treatments for our patients. And I think that’s where the eld is going. Hopefully, we’ll be able to better tailor the choices we have toward specific patient groups."
References:
- Kipps TJ, Stevenson FK, Wu CJ, et al. Chronic lymphocytic leukaemia [published correction appears in Nat Rev Dis Primers. 2017;3:17008. doi:10.1038/nrdp.2017.8]. Nat Rev Dis Primers. 2017;3:16096. doi: 10.1038/nrdp.2016.96.
- Eichhorst B, Robak T, Montserrat E et al. Chronic lymphocytic leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26(suppl 5):v78-v84. doi: 10.1093/annonc/mdv303.
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma. Version 2.2017. NCCN website. www.nccn.org/professionals/physician_gls/ pdf/cll.pdf. Published February 21, 2017. Accessed May 15, 2017.
- Scarfò L, Ferreri AJ, Ghia P. Chronic lymphocytic leukaemia. Crit Rev Oncol Hematol. 2016;104:169-182. doi: 10.1016/j.critrevonc.2016.06.003.
- Pan JW, Cook LS, Schwartz SM, Weis NS. Incidence of leukemia in Asian migrants to the United States and their descendants. Cancer Causes Control. 2002;13(9):791-795.
- Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin 2017;67(1):7-30. doi: 10.3322/caac.21387
- National Cancer Institute. Cancer stat facts: Chronic lymphocytic leukemia (CLL). NCI website. https://seer.cancer.gov/statfacts/html/clyl.html. Accessed May 15, 2017.
- Li Y, Wang Y, Wang Z, Yi D, Ma S. Racial differences in three major NHL subtypes: descrip- tive epidemiology. Cancer Epidemiol. 2015;39(1):8-13. doi: 10.1016/j.canep.2014.12.001.
- Goldin LR, Björkholm M, Kristinsson SY, Turesson I, Landgren O. Elevated risk of chronic lymphocytic leukemia and other indolent non-Hodgkin’s lymphomas among relatives of patients with chronic lymphocytic leukemia. Haematologica. 2009;94(5):647-653. doi: 10.3324/haematol.2008.003632.
- Cerhan JR, Slager SL. Familial predisposition and genetic risk factors for lymphoma. Blood. 2015;126(20):2265-2273. doi: 10.1182/blood-2015-04-537498.
- Di Bernardo MC, Crowther-Swanepoel D, Broderick P, et al. A genome-wide association study identi es six susceptibility loci for chronic lymphocytic leukemia. Nat Genet. 2008;40(10):1204-1210. doi: 10.1038/ng.219.
- Crowther-Swanepoel D, Broderick P, Di Bernardo MC, et al. Common variants at 2q37.3, 8q24.21, 15q21.3 and 16q24.1 in uence chronic lymphocytic leukemia risk. Nat Genet. 2010;42(2):132-136. doi: 10.1038/ng.510.
- Speedy HE, Di Bernardo MC, Sava GP, et al. A genome-wide association study identi es multiple susceptibility loci for chronic lymphocytic leukemia. Nat Genet. 2014;46(1):56- 60. doi: 10.1038/ng.2843.
- Baumann Kreuziger LM, Tarchand G, Morrison VA. The impact of Agent Orange exposure on presentation and prognosis of patients with chronic lymphocytic leukemia. Leuk Lymphoma. 2014;55(1):63-66. doi: 10.3109/10428194.2013.794267.
- Schinasi LH, De Roos AJ, Ray RM, et al. Insecticide exposure and farm history in relation to risk of lymphomas and leukemias in the Women’s Health Initiative obser- vational study cohort. Ann Epidemiol. 2015;25(11):803-810. doi: 10.1016/j.annepi- dem.2015.08.002.
- Hjalgrim H, Rostgaard K, Vasan SK, et al. No evidence of transmission of chronic lymphocytic leukemia through blood transfusion. Blood. 2015;126(17):2059-2061. doi: 10.1182/blood-2015-03-632844.
- Radivoyevitch T, Sachs RK, Gale RP, Smith MR, Hill BT. Ionizing radiation exposures in treatments of solid neoplasms are not associated with subsequent increased risks of chronic lymphocytic leukemia. Leuk Res. 2016;43:9-12. doi: 10.1016/j.leu- kres.2016.02.008.
- Strati P, Shanafelt TD. Monoclonal B-cell lymphocytosis and early-stage chronic lymphocytic leukemia: diagnosis, natural history, and risk strati cation. Blood. 2015;126(4):454-462. doi: 10.1182/blood-2015-02-585059.
- Rawstron AC, Bennett FL, O’Connor SJ, et al. Monoclonal B-cell lymphocytosis and chronic lymphocytic leukemia. N Engl J Med. 2008;359(6):575-583. doi: 10.1056/ NEJMoa075290.
- Hallek M, Cheson BD, Catovsky D, et al. Guidelines for the diagnosis and treat- ment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer InstituteWork- ing Group 1996 guidelines [erratum published in Blood. 2018;112(13):5259. doi: 10.1182/blood-2008-10-186254.]. Blood. 2008;111(12):5446-5456. doi: 10.1182/ blood-2007-06-093906.
- Döhner H, Stilgenbauer S, Benner A et al. Genomic aberrations and survival in chronic lymphocytic leukemia. N Engl J Med. 2000;343(26):1910-1916. doi: 10.1056/ NEJM200012283432602.
- Rai KR, Sawitsky A, Cronkite EP, Chanana AD, Levy RN, Pasternack BS. Clinical staging of chronic lymphocytic leukemia. Blood. 1975;46(2):219-234. doi: 10.1182/ blood-2016-08-737650.
- Rai KR, Sawitsky A, Cronkite EP, Chanana AD, Levy RN, Pasternack BS. Clinical staging of chronic lymphocytic leukemia. Blood. 1975;46(2):219-234. doi: 10.1182/ blood-2016-08-737650.
- Binet JL, Auquier A, Dighiero G, et al. A new prognostic classi cation of chronic lympho- cytic leukemia derived from a multivariate survival analysis. Cancer. 1981;48(1):198-206.
- Hallek M, Wanders L, Ostwald M, et al. Serum beta(2)-microglobulin and serum thymidine kinase are independent predictors of progression-free survival in chronic lymphocytic leukemia and immunocytoma. Leuk Lymphoma. 1996;22(5-6):439-447.
- Wierda WG, O’Brien S, Wang X, et al. Characteristics associated with important clinical end points in patients with chronic lymphocytic leukemia at initial treat- ment. J Clin Oncol. 2009;27(10):1637-1643. doi: 10.1200/JCO.2008.18.1701.
- Oscier DG, Gardiner AC, Mould SJ, et al. Multivariate analysis of prognostic factors in CLL: clinical stage, IGVH gene mutational status, and loss or mutation of the p53 gene are independent prognostic factors. Blood. 2002;100(4):1177-1184.
- Thompson PA, O’Brien SM, Xiao L, et al. β2-microglobulin normalization within 6 months of ibrutinib-based treatment is associated with superior progression-free survival in patients with chronic lymphocytic leukemia. Cancer. 2016;122(4):565- 573. doi: 10.1002/cncr.29794.
- American Journal of Managed Care® (AJMC®). Health plans encourage use of diag- nostic tests in CLL. www.ajmc.com/insights/updates-in-the-treatment-of-chronic- lymphocytic-leukemia/health-plans-encourage-use-of-diagnostic-tests-in-cll/text- version. Published July 18, 2016. Accessed May 15, 2017.
- Zenz T, Eichhorst B, Busch R, et al. TP53 mutation and survival in chronic lympho- cytic leukemia. J Clin Oncol. 2010;28(29):4473-4479. doi: 10.1200/JCO.2009.27.8762.
- Stilgenbauer S, Sander S, Bullinger L, et al. Clonal evolution in chronic lymphocytic leukemia: acquisition of high-risk genomic aberrations associated with unmutated VH, resistance to therapy, and short survival. Haematologica. 2007;92(9):1242-1245.
- Kipps TJ, Hillmen P, Demirkan F, et al. 11q Deletion (del11q) is not a prognostic factor for adverse outcomes for patients with chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) treated with ibrutinib: pooled data from 3 ran- domized phase 3 studies. Blood. 2016;128(22): 2042.
- Damle RN, Wasil T, Fais F, et al. Ig V gene mutation status and CD38 expres- sion as novel prognostic indicators in chronic lymphocytic leukemia. Blood. 1999;94(6):1840-1847.
- Hamblin TJ, Davis Z, Gardiner A, Oscier DG, Stevenson FK. Unmutated Ig V(H) genes are associated with a more aggressive form of chronic lymphocytic leukemia. Blood. 1999;94(6):1848-1854.
- Woyach JA, Ruppert AS, Heerema NA, et al. Chemoimmunotherapy with udara- bine and rituximab produces extended overall survival and progression-free sur- vival in chronic lymphocytic leukemia: long-term follow-up of CALGB study 9712. J Clin Oncol. 2011;29(10):1349-1355. doi: 10.1200/JCO.2010.31.1811.
- Fischer K, Bahlo J, Fink AM, et al. Long-term remissions after FCR chemoimmu- notherapy in previously untreated patients with CLL: updated results of the CLL8 trial. Blood. 2016; 127(2):208-215. doi: 10.1182/blood-2015-06-651125.
- Kipps TJ, Fraser G, Coutre S, et al. Unmutated IGHV is not an adverse predictor of outcome to therapy with ibrutinib in patients with chronic lymphocytic leuke- mia/small lymphocytic lymphoma (CLL/SLL). Presented at: 2017 AACR Annual Meeting; April 1-5, 2017; Washington, DC. Abstract CT158/24.
- Shanafelt TD, Geyer SM, Bone ND, et al. CD49d expression is an independent predictor of overall survival in patients with chronic lymphocytic leukaemia: a prognostic parameter with therapeutic potential. Br J Haematol. 2008;140(5):537- 546. doi: 10.1111/j.1365-2141.2007.06965.x.
- Rassenti LZ, Huynh L, Toy TL, et al. ZAP-70 compared with immunoglobulin heavy-chain gene mutation status as a predictor of disease progression in chronic lymphocytic leukemia. N Engl J Med. 2004;351(9):893-901.
- Rassenti LZ, Jain S, Keating MJ, et al. Relative value of ZAP-70, CD38, and immunoglobulin mutation status in predicting aggressive disease in chronic lymphocytic leukemia. Blood. 2008;112(5):1923-1930. doi: 10.1182/ blood-2007-05-092882.
- American Journal of Managed Care® (AJMC®). Pathophysiology, prognostic factors, and potential risks that in uence treatment in CLL. www.ajmc.com/insights/ updates-in-the-treatment-of-chronic-lymphocytic-leukemia/pathophysiology-prog- nostic-factors-and-potential-risks-that-in uence-treatment-in-cll/text-version. Published August 4, 2016. Accessed May 17, 2017.
Novel Approaches Focus on Limiting Toxicity in Older Patients With ALL
April 22nd 2024The major challenges for clinicians treating older patients with acute lymphoblastic leukemia surround the emergence of resistance to existing therapies and the toxicities associated with current chemotherapies.
Read More
Ornstein Advises on Starting Dose and Management of Lenvatinib in RCC
April 21st 2024During a Case-Based Roundtable® event, Moshe Ornstein, MD, MA, provided guidance on dosing and toxicity concerns in a patient treated with lenvatinib plus pembrolizumab for advanced renal cell carcinoma.
Read More