Current Treatment Strategies in Nonsquamous Non-Small Cell Lung Cancer
Nonsquamous non‒small cell lung cancer (NSCLC) is a heterogeneous disease with multiple treatment options dependent upon staging, presence of metastasis, and patient factors—including presence of comorbidities—among other considerations. As such, current treatment options include surgical resection, chemotherapy, radiation, immunotherapy, and targeted therapy.
1While conventional treatment for patients without driver mutations is platinum-based chemotherapy, much of the field has shifted to first-line use of agents such as bevacizumab (Avastin) or pembrolizumab (Keytruda) plus pemetrexed (Alimta) and carboplatin.
As explained by Thu Oanh Dang, PharmD, BCOP, clinical oncology pharmacist at Memorial Sloan Kettering Cancer Center, New York, “for about 2 and a half decades after cisplatin became widely used in the late 1970s, the therapeutic armamentarium of the medical oncologist for the treatment of NSCLC was limited largely to cytotoxic chemotherapy.”2Despite advances in chemotherapy, however, "the development of therapeutic resistance to both cytotoxic chemotherapy and targeted therapy is universal" for patients with advanced or metastatic disease. Fortunately, the development and approval of checkpoint inhibitors and antiVEGF therapies over the past decade provide attractive options for delaying recurrence and improving overall survival (OS).
Accelerated Approval for Pembrolizumab Combined With Pemetrexed/Carboplatin
In May 2017, pembrolizumab, an immune checkpoint inhibitor, was granted accelerated approval by the FDA as a part of first-line combination therapy with pemetrexed and carboplatin for untreated, metastatic, nonsquamous NSCLC, regardless of programmed death ligand-1 (PD-L1) expression levels.3The accelerated approval expanded on previous indications for pembrolizumab, which was already approved as a first-line monotherapy for patients with NSCLC with high PD-L1 expression or second-line therapy for NSCLC with any PD-L1 expression.1
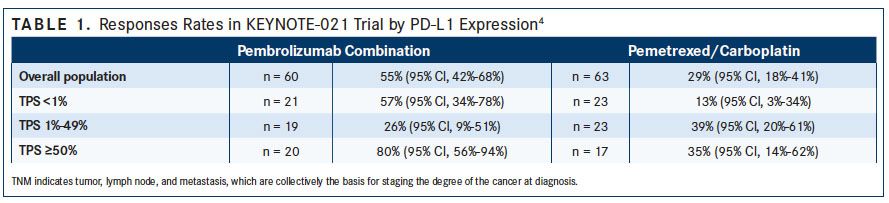
Approval for pembrolizumab as first-line combination therapy was based on cohort G of the phase II KEYNOTE-021 study that evaluated carboplatin and pemetrexed with or without pembrolizumab. In this cohort, 123 patients with chemotherapy-naïve, stage IIIb/IV, nonsquamous NSCLC were stratified by PD-L1 expression, and were randomly assigned to receive 4 cycles of carboplatin and pemetrexed with or without 24 months of pembrolizumab. Pemetrexed maintenance therapy was allowed in both groups.4
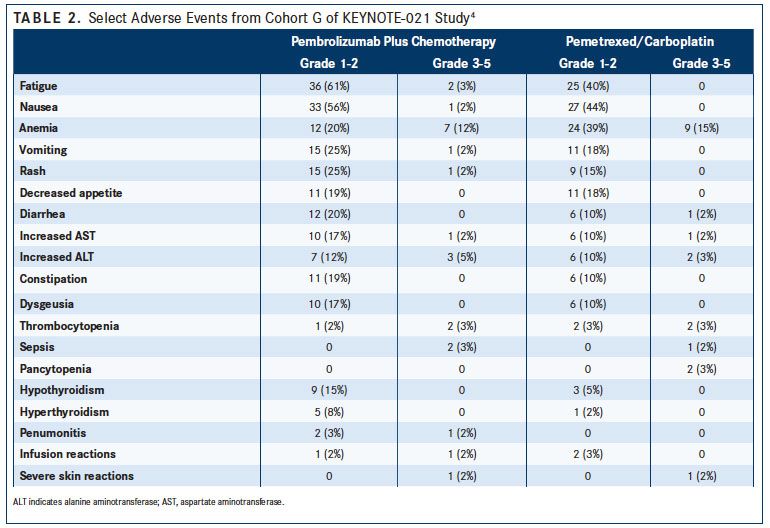
After a median follow-up of 10.6 months, more patients in the pembrolizumab group compared with the chemotherapy group achieved an objective response (55% vs 29%, respectively; P = .0016).4For patients in the pembrolizumab group, no difference in objective response rate (ORR) was noted for patients with a PD-L1 tumor proportion score (TPS) of less than 1% (57%) or patients with a PD-L1 TPS of at least 1% (54%). The researchers, however, noted an increased ORR among patients with PD-L1 expression of 50% or more compared with those with a score of 1% to 49% (80% vs 26%) (TABLE 1). The incidence of severe treatment-related adverse events (AEs) was similar between the groups (39% vs 26%), with anemia (12%) and decreased neutrophil count (5%) occurring most commonly in the pembrolizumab group (TABLE 2).4
In a news release from Merck, the developer of pembrolizumab, announcing the accelerated FDA approval for pembrolizumab as a first-line combination therapy agent, Corey Langer, MD, lead investigator of the KEYNOTE-021 study, noted, “This approval marks an important milestone in the treatment of lung cancer. Now, pembrolizumab in combination with pemetrexed and carboplatin can be prescribed in the first-line setting for patients with metastatic nonsquamous nonsmall cell lung cancer, irrespective of PD-L1 expression.” Importantly, emphasized, Langer, director of the Thoracic Oncology Program at Penn Medicine in Philadelphia, Pennsylvania, "Physicians should continue to use each patient’s individual characteristicsincluding biomarker status, histology, and other clinical factors—to determine the best treatment plan for each person."5Phase III trials confirming these findings are ongoing.6
PD-L1 Expression and First-Line Pembrolizumab
Before pembrolizumab was approved for use as first-line combination therapy, it was approved in October 2016 as monotherapy for metastatic, nonsquamous NSCLC with high PD-L1 expression.6According to Lee Pai-Scherf, MD, of the Center for Drug Evaluation and Research at the FDA, and colleagues, this indication could impact “about a quarter of patients with [metastatic NSCLC].”6This approval was based on the benefit demonstrated in the KEYNOTE-010 and KEYNOTE-024 clinical trials.7,8
In the randomized, open-label phase III KEYNOTE-024 study, pembrolizumab monotherapy and a platinum-based chemotherapy regimen were compared in patients with PD-L1 expression of at least 50% and no other driver mutations. The primary endpoint of median progression-free survival (PFS) was significantly longer in the pembrolizumab group (10.3 months) compared with standard-of-care chemotherapy (6.0 months; HR, 0.50; P <.001). Moreover, OS at 6 months (80.2% vs 72.4%) and ORRs (44.8% vs 27.8%) were higher in the pembrolizumab group compared with the chemotherapy group, respectively.8
Treatment-related AEs were less common in the pembrolizumab groups. Of particular interest, pneumonitis and severe skin reactions occurred in fewer than 4% of patients in either trial.7,8Dang called the results “promising,” and indicated that reported cases of durable disease control suggest that “pembrolizumab may ‘lift the tail of the curve,' which is a phenomenon similarly observed with advanced, cutaneous melanoma in the era of immunotherapy. Whereby immune checkpoint inhibitor therapy can result in a plateau at the end of a survival curve, non-immunomodulatory strategies usually result in the curve’s tail-end returning to zero.”2
Pemetrexed Disodium Injection: Beneficial for Nonsquamous NSCLC
Pemetrexed in combination with cisplatin has been shown to improve survival of patients with nonsquamous NSCLC. Compared with gemcitabine plus cisplatin, pemetrexed plus cisplatin had noninferior median OS (10.3 vs 10.3 months, respectively; HR, 0.94), median PFS (5.1 vs 4.8 months, respectively; HR, 1.04), and ORRs (24.7% vs 27.1%, respectively). Importantly, the pemetrexed group had fewer grade 3/4 AEs, including neutropenia, anemia, and thrombocytopenia (P <.001 each), febrile neutropenia (P = .002), and alopecia (P <.001). Grade 3/4 nausea, however, was more common in the pemetrexed group (P = .004).9Pemetrexed has also been evaluated in combination with carboplatin. In a clinical trial comparing pemetrexed plus cisplatin versus pemetrexed plus carboplatin, both carboplatin and cisplatin groups met their primary endpoints, but cisplatin may have been slightly more efficacious. Median PFS (6.0 vs 4.7 months), 6-month PFS rate (50.5% vs 34.9%), median OS (11.7 vs 8.9 months), and 1-year OS rate (47.5% vs 39.2%) were all modestly better in the cisplatin group compared with the carboplatin group, respectively. AEs were slightly more common for cisplatin compared with carboplatin, and included neutropenia (16.9% vs 26.2%, respectively), anemia (7.7% vs 10.8%), thrombocytopenia (3.1% vs 16.9%), and nausea (4.6% vs 7.7%).10In an expert review of pemetrexed in NSCLC, Pascale Tomasini, MD, of the Multidisciplinary Oncology and Therapeutic Innovations Department at Aix-Marseille University, Marseille, France, commented, "Pemetrexed is one of the most frequently used drugs in the management of stage IV [nonsquamous] NSCLC patients. The good efficacy and the favorable safety profile make this drug one of the most commonly used in the first-line setting."
Addition of Bevacizumab to Platinum Doublet
Bevacizumab, an anti-VEGF monoclonal antibody, was approved for the treatment of NSCLC in combination with carboplatin and paclitaxel in 2006.11 In a randomized, open-label trial, chemotherapy-naïve patients with stage IIIb/IV nonsquamous NSCLC were randomly assigned to receive carboplatin plus paclitaxel with or without bevacizumab. The bevacizumab group had significantly longer median OS (12.3 vs 10.3 months; HR, 0.79; P = .003) and longer median PFS (6.2 vs 4.5 months; HR, 0.66; P <.001). Severe AEs that were more common in patients receiving bevacizumab included neutropenia (26% vs 17% without bevacizumab), fatigue (16% vs 13%), hypertension (7% vs 0.7%), infection without neutropenia (7% vs 3%), and thrombosis/embolism (5% vs 3%), among others.12
According to the National Comprehensive Cancer Network (NCCN) guidelines,1 bevacizumab plus carboplatin and paclitaxel has a category 1 recommendation, but bevacizumab combined with other chemotherapy regimens is also an option. Compared with paclitaxel combination therapy, pemetrexed with carboplatin and bevacizumab was shown to have a similar efficacy profile in terms of OS (HR, 1.00; P = .949), with a modest improvement in PFS (HR, 0.83; P = .012). While both regimens were tolerable, the toxicity profiles were shown to be distinct between the 2 regimens. Grade 3/4 anemia, thrombocytopenia, and fatigue were more common with the pemetrexed regimen; grade 3/4 neutropenia, febrile neutropenia, sensory neuropathy, and alopecia were more common with the paclitaxel regimen.13
Treatment with bevacizumab has been associated with bleeding-related AEs; however, more recent studies with careful patient selection revealed that bevacizumab is “a safe drug with an acceptable safety profile,” according to Salvatore Lauro, MD, in the Department of Medical Oncology, Sant'Andrea Hospital, Rome, Italy, and colleagues in a review of bevacizumab treatment.14“The results of pivotal studies…argue in favor of the use of this drug,”they concluded, and went on to describe the possible benefits of bevacizumab: improved quality of life, delayed initiation of second-line treatment, and saving other effective treatments for successive lines of therapy.14
Considerations When Selecting Between First-Line Therapies
For advanced or metastatic nonsquamous NSCLC, the NCCN recommends that the “drug regimen with the highest likelihood of benefit with toxicity deemed acceptable to both the physician and the patient should be given as initial therapy.”1Determining the therapy with the highest likelihood of benefit, however, can depend upon a variety of disease factors (stage, histology, PD-L1 expression) and patient factors (ECOG performance status [PS], comorbidities, organ function).1
For patients with early-stage NSCLC, surgery with or without adjuvant chemotherapy or radiation therapy is usually the preferred treatment option.1,15For patients with locally advanced or metastatic NSCLC, clinicians now have more options: platinum doublet chemotherapy with or without bevacizumab, platinum doublet therapy with or without pembrolizumab, or pembrolizumab monotherapy.
With the approval and success of the immune checkpoint inhibitor pembrolizumab, the first tool to stratify patients after tumor genotyping will likely be PD-L1 expression level, quantified via immunohistochemistry. For patients with 50% or more PD-L1 expression, pembrolizumab monotherapy may be preferred due to the superior efficacy and tolerability compared with chemotherapy in this patient population.1,7,8Jordi Remon, MD, medical oncologist in the Cancer Medicine Department at Consorci Sanitari del Maresme, Mataró, Spain, suggested that pembrolizumab “should be considered a standard first-line treatment in NSCLC patients with a good performance status whose tumors have PD-L1 expression ≥50%.”16For patients with PD-L1 expression levels less than 50%, pembrolizumab combination therapy with pemetrexed and carboplatin is an option, although the treatment benefits are not as substantial as for patients with high PD-L1 expression.4According to Jong-Mu Sun, MD, of the Division of Hematology-Oncology at Sungkyunkwan University School of Medicine, in Seoul, South Korea, patients with weak or no PD-L1 signals may be considered for combination therapy based on the KEYNOTE-021 trial, “although data from large randomized trials are needed to confirm this. In addition, when considering the combination therapy, the physicians should keep in mind additional toxicities and should be more alert for these events. For those with low PD-L1 expression, PD-1/PD-L1 inhibitors can be reserved for second (or beyond)-line therapy following platinum-based chemotherapy.”17
Performance status and fitness are also important considerations when selecting treatments for patients with nonsquamous NSCLC. For patients with a PS of 3 or 4, the NCCN guidelines recommend best supportive care.1For patients with PS 0 to 2, systemic therapy is recommended as first-line treatment, but multiple options for treatment exist.1For better PS of 0 or 1, category 1 recommendations include various doublet chemotherapy regimens and bevacizumab with carboplatin and pemetrexed. Pembrolizumab with carboplatin and pemetrexed is included as an option but is not considered category 1, while bevacizumab with pemetrexed and platinum-based chemotherapy is.1
As explained by Tracey Evans, MD, associate professor of clinical medicine, Department of Medical Oncology at Penn Medicine, Philadelphia, Pennsylvania, in a review, “Due to the generally poor outcomes of patients with poor [performance status] treated with chemotherapy, the tendency has been to either not give chemotherapy at all or to give less toxic single-agent chemotherapy. It nevertheless stands to reason that if the patient’s poor [performance status] is caused by the cancer, treating the cancer more aggressively may lead to improved outcomes.”18Regardless of treatment selection, in most clinical trials, patients with PS 2 have worse clinical outcomes than patients with PS 0 or 1.19Nonetheless, treatment does improve outcomes compared with no treatment.
For patients with PS 2, carboplatin is preferred over cisplatin, likely due to the distinct toxicity profile, and it is recommended as a part of doublet chemotherapy.1,20Additionally, single-agent chemotherapy is an option recommended in the NCCN guidelines for this patient population. In a study of patients with advanced NSCLC and a PS of 2, combination carboplatin with pemetrexed significantly improved survival compared with pemetrexed alone (9.3 vs 5.3 months; HR, 0.62; P = .001).21Evans suggested that treatment with carboplatin-containing chemotherapy would be the optimal choice in this patient population, “for patients with PS 2 who are interested in maximizing OS and who are willing to undergo cytotoxic chemotherapy, carboplatin-based doublet therapy is indicated and is superior to single-agent therapy.”18Gemcitabine alone or in combination with docetaxel or vinorelbine is another option for patients with a PS of 2, as is single- agent paclitaxel.
Like PS, older age may be considered a negative predictor of treatment response for some regimens, although older age alone does not preclude patients from receiving treatment.22A review of cisplatin- and carboplatin-based chemotherapies in elderly patients (generally age >70 years) by Remei Blanco, MD,PhD, of Consorci Sanitari de Terrassa in Barcelona, Spain, and colleagues illuminated “The feasibility of these schedules in the elderly, as no study showed a significant difference in survival according to age, except for a combined analysis of results from 2 SWOG trials.” The study authors also explained that "a nonsignificant trend towards an [OS] benefit with platinum-based doublet therapy over gemcitabine monotherapy was reported, albeit with an increased risk of toxicity."22Pemetrexed has also been shown to be equally efficacious among older and younger patients in terms of OS (HR comparing patients aged <65 years with those aged >65 years, 0.92).23
Older age alone is not a contraindication for bevacizumab, since no increase in bleeding-related AEs has been observed in elderly patients.24However, a recent secondary analysis of the PointBreak and ECOG 4599 trials of paclitaxel plus carboplatin with or without bevacizumab evaluated the efficacy and safety of bevacizumab combination chemotherapy specifically in patients grouped by age.25In this report, OS was significantly improved by the addition of bevacizumab to carboplatin and paclitaxel for all age groups younger than 75 years (age <65 years: HR, 0.75; age 65-74 years: HR, 0.80), but no improvement was noted for those patients who were 75 years or older (HR, 1.05). AEs were significantly increased in the bevacizumab group compared with the chemotherapy-alone group among patients age 75 years or older (81% vs 56%; P <.05).25Similar results were reported in another trial that focused on patients aged ≥65 years.26Based on these data, Evans concluded that it is unclear “whether bevacizumab offers benefit, but it is reasonable to offer it to elderly patients who do not have contraindications to taking bevacizumab.”18Comorbidities and other patient factors, such as organ function, are important considerations when selecting treatment for patients with nonsquamous NSCLC. For bevacizumab, bleeding risks are often of concern, so exclusion of patients with the presence of hemoptysis is an important consideration.14For cisplatin, poor kidney function is an important contraindication due to the potential for nephrotoxicity, so carboplatin in combination with paclitaxel should be considered as an alternative.1,27
Second-Line Therapies for NSCLC
Selection of second-line therapies for nonsquamous NSCLC is dependent upon the choice of first-line therapies. According to Antonio Rossi, MD, of the Division of Medical Oncology at S.G. Moscati Hospital, in Avellino, Italy, “If the adequate choice of first-line treatment for NSCLC management is very important, the choice of second-line treatment is of vital importance, too.”28
On progression of NSCLC, preferred second-line therapy is usually an immune checkpoint inhibitor if one has not been previously given.1In CheckMate-057, nivolumab (Opdivo) was compared with docetaxel in patients with progressive NSCLC following first-line platinum-based chemotherapy.29The nivolumab group had significantly longer median OS compared with the docetaxel group (12.2 vs 9.4 months; P = .002). Additionally, the 1-year ORR was 51% for the nivolumab group compared with 39% in the docetaxel group. The ORR was more durable for patients receiving nivolumab, with a median duration of 17.2 months compared with 5.6 months in the docetaxel group. Moreover, AEs were substantially higher in the docetaxel group, with 54% of patients experiencing grade 3/4 AEs compared with 10% in the nivolumab group. The researchers noted that nivolumab was associated with similarly improved outcomes at all levels of PD-L1 expression over 1%, but improvements were lower for patients with PD-L1negative NSCLC.29
Pembrolizumab is another immune checkpoint inhibitor that has been evaluated as second-line therapy. In KEYNOTE-010, pembrolizumab was compared with docetaxel among patients with at least 1% PD-L1 expression.7Median OS was higher in the pembrolizumab groups (10.4 months for 2 mg/kg and 12.7 months for 10 mg/kg) compared with docetaxel (8.5 months), resulting in HRs of 0.71 and 0.61, respectively. Among patients with at least 50% PD-L1 expression, OS was significantly longer with pembrolizumab 2 mg/kg (14.9 months) and 10 mg/kg (17.3 months) compared with docetaxel (8.2 months). As with nivolumab, toxicity with pembrolizumab was lower compared with docetaxel, with 13%, 16%, and 35% of patients in the 2 mg/kg, 10 mg/kg, and docetaxel groups, respectively, experiencing grade 3/4 AEs.7Rossi explained, “Considering that the outcomes of the 2 doses of pembrolizumab reported in the KEYNOTE-010 trial were very similar, pembrolizumab was registered, at the lower dose of 2 mg/ kg, for the second-line treatment of patients with NSCLC of any histology with PD-L1 expression ≥1% with a companion diagnostic.”28
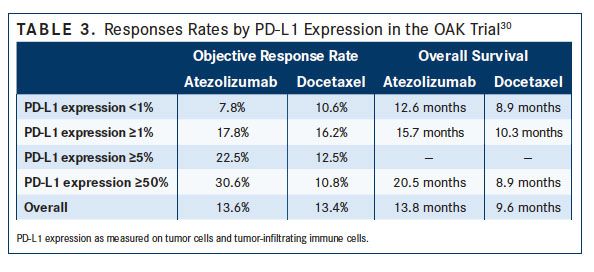
Atezolizumab (Tecentriq) is another checkpoint inhibitor that is available as second-line therapy for patients with progressive, previously treated NSCLC. In the phase III OAK trial,30patients with previously treated NSCLC were randomly assigned to receive atezolizumab or docetaxel. OS was significantly improved in the atezolizumab group (13.8 vs 9.6 months; HR, 0.73; P = .0003). In subgroup analyses, OS was improved in both PD-L1positive patients (15.7 vs 10.3 months; HR, 0.74; P = .0102) and PD-L1–negative patients (12.6 vs 8.9 months; HR, 0.75; P = .02). For patients with high PD-L1 expression, OS was 20.5 months compared with 8.9 months in the docetaxel group (HR, 0.41; P <.0001) (TABLE 3). Grade 3/4 AEs were less common with atezolizumab (15%) compared with docetaxel (43%).30In an expert review of second-line therapies for NSCLC, Fernando Santini, MD, of the Thoracic Oncology Service, Memorial Sloan Kettering Cancer Center, New York, New York, and Division of Medical Oncology, in Sao Paolo, Brazil, and of the Division of Medical Oncology, in Sao Paolo, Brazil, concluded, “the landscape of second-line treatment of patients with NSCLC has 3 checkpoint inhibitor drugs approved, with similar activities and safety profiles.” Of the 3 second-line checkpoint inhibitors, atezolizumab and nivolumab are the only 2 with approval regardless of PD-L1 status.31
Although systemic immune checkpoint inhibitors are preferred agents for second-line treatment, they are unlikely to be effective in patients who have already used these regimens. Other systemic therapy options include docetaxel, pemetrexed, gemcitabine, or ramucirumab (Cyramza) plus docetaxel. All of these agents have category 2b recommendations in the NCCN guidelines, indicating an NCCN consensus of appropriate therapy based on lower-level evidence.1
Docetaxel has been extensively studied as second-line therapy for NSCLC. In the TAX320 trial, patients were randomized to receive various doses of docetaxel compared with either vinorelbine or ifosfamide.32Although median OS was not significantly different between low-dose docetaxel, high-dose docetaxel, or the control treatment (5.7 vs 5.5 vs 5.6 months, respectively), the rate of patients achieving 1-year survival was significantly higher with docetaxel 75 mg/m2 compared with the control groups (32% vs 19%; P = .025).32"Overall, docetaxel, registered at the 3-weekly dose of 75 mg/m2, has become the standard second-line treatment against which any new drug has to be compared for registrative purposes," noted Rossi.28Docetaxel can also be combined with ramucirumab (Cyramza), an anti-VEGF agent, in the second-line setting. In the REVEL trial,33combination ramucirumab with docetaxel was compared with docetaxel alone in patients with NSCLC who had progressed after platinum-based chemotherapy. Median OS was longer in the combination group compared with docetaxel alone (10.5 vs 9.1 months; HR, 0.86; P = .023) as was PFS (4.5 vs 3.0 months; HR, 0.76; P <.0001). However, grade 3/4 AEs were more common in the ramucirumab group, and included neutropenia (49% vs 40%), febrile neutropenia (16% vs 10%), fatigue (14% vs 10%), leucopenia (14% vs 12%), and hypertension (6% vs 2%).33In an expert commentary on ramucirumab in NSCLC, Oscar Arrieta, MD, of the Instituto Nacional de Cancerología, in Mexico City, Mexico, and colleagues acknowledged that immune checkpoint therapies are preferred for patients with high PD-L1, but noted,“Patients with negative PD-L1 expression might respond better to treatment with ramucirumab.”34
For patients who were not originally treated with pemetrexed, pemetrexed provides a good second-line option, as well. Compared with docetaxel, pemetrexed was noninferior in previously treated patients with NSCLC (median survival time, 7.9 vs 8.3 months; HR, 0.99; P = .226). Docetaxel, however, was associated with a significantly higher rate of grade 3/4 AEs compared with pemetrexed, including neutropenia (40.2% vs 5.3%; P <.001), febrile neutropenia (12.7% vs 1.9%; P <.001), neutropenia with infections (3.3% vs 0.0%; P <.004), and hospitalizations for neutropenic fever (13.4% vs 1.5%; P <.001).35
Maintenance Therapy: Improving Outcomes Following First-Line Treatment
Maintenance therapy is a relatively new phenomenon of utilizing a single agent after successful treatment with first-line therapy “in an effort to improve outcomes and maximize therapy with a potentially less toxic and tolerable [treatment],” according to Stephanie Brulé, MD, of the Department of Medical Oncology at the Ottawa Hospital Cancer Centre, Ottawa, Ontario, Canada.36Two forms of maintenance therapy are used in nonsquamous NSCLC: continuation maintenance, usually with bevacizumab or pemetrexed; or switch maintenance, usually to pemetrexed.1,36Explaining the differences between the theories behind the 2 approaches, Brulé commented, “switch maintenance therapy leads to increased exposure of tumour cells to non-cross-resistant therapies. Therefore, this may lead to a decreased incidence of chemotherapy resistance and to increased antitumor effects. Further, switch maintenance therapy can act as an early form of second-line therapy,” whereas continuation maintenance capitalizes on the response that patients have already exhibited to first-line treatment.36
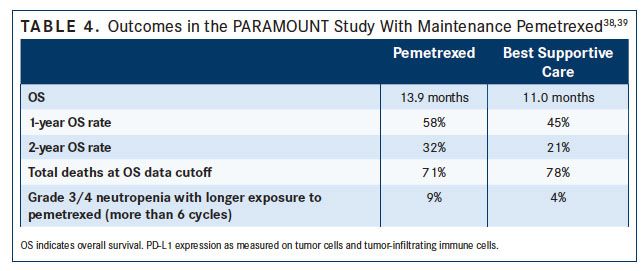
The only category 1 recommendations from NCCN for maintenance therapy are continuation maintenance with bevacizumab or pemetrexed.1 In the PARAMOUNT study,37maintenance therapy with pemetrexed was compared with best supportive care (BSC) following first-line treatment with pemetrexed plus cisplatin. Compared with BSC, maintenance therapy with pemetrexed resulted in a significant reduction in disease progression (HR, 0.62; P <.0001), improvement in median PFS (4.1 vs 2.8 months; P <.0001), and increased median OS (13.9 vs 11.0 months; P = .0195) (TABLE 4).37,38 Grade 3/4 AEs were more common in the pemetrexed group (anemia, neutropenia, and fatigue).37Nonetheless, pemetrexed maintenance therapy is considered to be well tolerated, since quality of life was shown to be similar between placebo and pemetrexed, with pemetrexed delaying worsening of pain and hemoptysis.39
Bevacizumab maintenance treatment was evaluated in a retrospective analysis of the ECOG 4599 trial,40which compared patients treated with bevacizumab plus paclitaxel and carboplatin with patients treated with carboplatin and paclitaxel alone. Bevacizumab maintenance was allowed for nonprogressors. In this study, bevacizumab induction and maintenance therapy was associated with improved OS (HR, 0.75; P <.030) and reduced PFS (HR, 0.64; P = .001), which was "suggestive of possible benefit because of bevacizumab maintenance," according to the study authors.40In the AVAPERL study, bevacizumab maintenance therapy with or without pemetrexed was evaluated.41,42Although the addition of pemetrexed to bevacizumab maintenance improved median PFS (3.7 vs 7.4 months for bevacizumab vs chemotherapy only; HR, 0.48; P <.001), no significant improvement in OS was reported (13.2 for bevacizumab vs 17.1 months for bevacizumab/pemetrexed; HR, 0.87; P = .29). Furthermore, few AEs were reported for bevacizumab alone in the ECOG 4599 and AVAPERL trials, but the addition of pemetrexed increased the number of grade 3/4 AEs.40,41Gemcitabine can also be continued for maintenance therapy. Continuation with gemcitabine significantly improved PFS (3.8 vs 1.9 months; P <.001), but did not significantly improve OS (12.1 vs 10.8 months; HR, 0.89; P = .3867).43A subgroup analysis, however, revealed that maintenance gemcitabine may improve OS among patients who had a response during induction therapy (15.2 vs 10.8 months; HR, 0.72). Switch maintenance therapy is currently recommended only with pemetrexed, based on the results of the JMEN trial.44 Pemetrexed-naïve patients who had previously received platinum-based chemotherapy were randomly assigned to receive pemetrexed or BSC following nonprogression after first-line chemotherapy. The pemetrexed group had significantly improved PFS (4.3 vs 2.6 months; HR, 0.50; P <.0001) and OS (13.4 vs 10.6 months; HR, 0.79; P = .012) compared with BSC. Although treatment discontinuations due to AEs were more common in the pemetrexed group, grade 3/4 AEs were relatively low.44
In a review of the recent advances in maintenance therapy, Ryan Gentzler, MD, at the time a fellow in the Division of Hematology/Oncology at Northwestern University Feinberg School of Medicine, in Chicago, Illinois, concluded that "it is clear that maintenance therapy will not benefit all patients," such as those who are PS 2.45“Ultimately, the decision to use maintenance chemotherapy requires a nuanced discussion between the patient and physician that adequately assesses benefits of prolonged therapy and impact in terms of toxicity, [quality of life], and financial cost.”45
Localized Treatments: Radiation and Surgery
The preferred treatment for stage I or II NSCLC is surgical resection in patients who are fit enough to tolerate it. Surgical resection can be of varied invasiveness, with the removal of an entire lung (pneumonectomy) or a part of a lung lobe (wedge resection).46Nearby lymph nodes may also be removed if deemed necessary. Paul Van Schil, MD, PhD, of the Department of Thoracic and Vascular Surgery at Antwerp University Hospital, Antwerp, Belgium, explained, “the final aim of surgical treatment is complete resection,” so clinicians will typically balance the patient’s lung health and the extent of the operation to ensure that the resection is complete as possible while maintaining good lung function.46 In a retrospective study of patients with poor lung function undergoing surgery, most patients had good outcomes following lobectomy. The study authors suggested that this indicates that “lobectomy can be safely performed in select patients considered to be high risk for resection by pulmonary function tests.”47Video-assisted thoracic surgery can be used to treat early-stage cancer located on the outside of the lung with good success. Outcomes of video-assisted surgery and open surgery are similar, with fewer complications occurring with video-assisted resection.48
Radiation therapy can be used in all stages of NSCLC, and is preferred for patients who are unable to tolerate surgery or for locally advanced NSCLC. For some patients, radiation is also used before or after surgery to reduce the risk of recurrence and metastasis; stereotactic body radiotherapy (SBRT) is typically preferred. In a prospective trial of patients receiving SBRT, 5-year OS was 52.2%, and the local control rate was 86% for tumors 3 cm or smaller.49 When video-assisted lobectomy and SBRT were compared for patients with stage I/II NSCLC, SBRT was modestly superior for locoregional control at 1 year (96.8% vs 93.3%; P = .04), but no differences in OS or distant recurrences were noted.49
Treatment Sequencing
In a proposed new treatment algorithm published in Clinics in Oncology, Rossi suggested that patients with nonsquamous NSCLC with no driver mutations and low PD-L1 expression should be initiated on either platinum-based chemotherapy with pemetrexed or bevacizumab with carboplatin and paclitaxel.50This suggestion is based upon the “right drug to right patient” approach, according to Rossi, and allows clinicians to “save” effective drugs such as checkpoint inhibitors (for both regimens) or pemetrexed (for the bevacizumab-based regimen) for later therapy sequences.51For patients with high PD-L1 expression, pembrolizumab is the recommended first-line therapy due to the increased efficacy of the agent in this patient population.1,51Second-line therapy should include agents that were not previously used in the first-line setting, usually a checkpoint inhibitor (nivolumab or atezolizumab for low or negative PD-L1 expressors) or other systemic therapy, such as pemetrexed or docetaxel.1Ramucirumab may be added to docetaxel for some patients, as well.1When selecting first-line therapy in patients who are not candidates for surgery, clinicians should always consider what options will remain available in the second-line setting. For this reason, Sun recommends that PD-L1 inhibitors should be “reserved for second (or beyond)-line therapy following platinum-based chemotherapy, based on data drawn from many second-line therapy trials.”17
References:
- NCCN Clinical Practice Guidelines in Oncology. NonSmall Cell Lung Cancer, versionn 5.2017. www.nccn.org/professionals/physician_gls/default.aspx. Published April 2017. Accessed October 24, 2017.
- Dang TO, Ogunniyi A, Barbee MS, Drilon A. Pembrolizumab for the treatment of PD-L1 positive advanced or metastatic non-small cell lung cancer.Expert Rev Anticancer Ther. 2016;16(1):13-20. doi: 10.1586/14737140.2016.1123626.
- Pembrolizumab (Keytruda) 5-10-2017 [news release]. Silver Spring, MD: FDA; May 24, 2017. www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm558048.htm. Accessed October 24, 2017.
- Langer CJ, Gadgeel SM, Borghaei H, et al. Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study.Lancet Oncol.2016;17(11):1497-1508. doi: 10.1016/S1470-2045(16)30498-3.
- FDA approves Merck’s KEYTRUDA® (pembrolizumab) as first-line combination therapy with pemetrexed and carboplatin for patients with metastatic nonsquamous non-small cell lung cancer (NSCLC), irrespective of PD-L1 expression [news release]. Kenilworth, NJ: Merck & CO, Inc; May 10, 2017. investors.merck.com/news/press-release-details/2017/FDA-Approves-Mercks-KEYTRUDA-pembrolizumab-as-First-Line-Combination-Therapy-with-Pemetrexed-and-Carboplatin-for-Patients-with-Metastatic-Nonsquamous-Non-Small-Cell-Lung-Cancer-NSCLC-Irrespective-of-PD-L1-Expression/default.aspx. Accessed October 24, 2017.
- Pai-Scherf L, Blumenthal GM, Li H, et al. FDA approval summary: pembrolizumab for treatment of metastatic non-small cell lung cancer: first-line therapy and beyond [published online August 23, 2017].Oncologist.2017. doi: 10.1634/theoncologist.2017-0078.
- Herbst RS, Baas P, Kim DW, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial.Lancet.2016;387(10027):1540-1550. doi: 10.1016/S0140-6736(15)01281-7.
- Reck M, Rodriguez-Abreu D, Robinson AG, et al; KEYNOTE-024 Investigators. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer.N Engl J Med.2016;375(19):1823-1833. doi: 10.1056/NEJMoa1606774.
- Scagliotti GV, Parikh P, von Pawel J, et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer.J Clin Oncol.2008;26(21):3543-3551. doi: 10.1200/JCO.2007.15.0375.
- Schuette WH, Groschel A, Sebastian M, et al. A randomized phase II study of pemetrexed in combination with cisplatin or carboplatin as first-line therapy for patients with locally advanced or metastatic non-small-cell lung cancer.Clin Lung Cancer.2013;14(3):215-223. doi: 10.1016/j.cllc.2012.10.001.
- Cohen MH, Gootenberg J, Keegan P, Pazdur R. FDA drug approval summary: bevacizumab (Avastin) plus carboplatin and paclitaxel as first-line treatment of advanced/metastatic recurrent nonsquamous non-small cell lung cancer.Oncologist.2007;12(6):713-718. doi: 10.1634/theoncologist.12-6-713.
- Sandler A, Gray R, Perry MC, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer.N Engl J Med.2006;355(24):2542-2550. doi: 10.1056/NEJMoa061884.
- Patel JD, Socinski MA, Garon EB, et al. PointBreak: a randomized phase III study of pemetrexed plus carboplatin and bevacizumab followed by maintenance pemetrexed and bevacizumab versus paclitaxel plus carboplatin and bevacizumab followed by maintenance bevacizumab in patients with stage IIIB or IV nonsquamous non-small-cell lung cancer.J Clin Oncol.2013;31(34):4349-4357. doi: 10.1200/JCO.2012.47.9626.
- Lauro S, Onesti CE, Righini R, Marchetti P. The use of bevacizumab in non-small cell lung cancer: an update [published correction appears in Anticancer Res. 2014;34(6):3232].Anticancer Res.2014;34(4):1537-1545.
- Postmus PE, Kerr KM, Oudkerk M, et al; ESMO Guidelines Committee. Early and locally advanced non-small-cell lung cancer (NSCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up.Ann Oncol.2017;28(suppl 4):iv1-iv21. doi: 10.1093/annonc/mdx222.
- Remon J, Besse B, Soria J-C. Successes and failures: what did we learn from recent first-line treatment immunotherapy trials in non-small cell lung cancer?BMC Med.2017;15(1):55. doi: 10.1186/s12916-017-0819-3.
- Sun J-M. PD-1/PD-L1 inhibitor therapy: transitioning from a second-line to a first-line therapy for non-small cell lung cancer.Precis Future Med.2017;1(1):3-9. doi: 10.23838/pfm.2017.00066.
- Evans T. Chemotherapy in advanced non-small cell lung cancer: optimal treatment approach for elderly and patients with poor performance status.Am J Hemat Oncol.2015;11(7):12-16.
- Zinner R, Visseren-Grul C, Spigel DR, Obasaju C. Pemetrexed clinical studies in performance status 2 patients with non-small cell lung cancer.Int J Oncol.2016;48(1):13-27. doi: 10.3892/ijo.2015.3219.
- de Castria TB, da Silva EM, Gois AF, Riera R. Cisplatin versus carboplatin in combination with third-generation drugs for advanced non-small cell lung cancer.Cochrane Database Syst Rev.2013(8):CD009256. doi: 10.1002/14651858.CD009256.pub2.
- Zukin M, Barrios CH, Pereira JR, et al. Randomized phase III trial of single-agent pemetrexed versus carboplatin and pemetrexed in patients with advanced non-small-cell lung cancer and Eastern Cooperative Oncology Group performance status of 2.J Clin Oncol.2013;31(23):2849-2853. doi: 10.1200/JCO.2012.48.1911.
- Blanco R, Maestu I, de la Torre MG, et al. A review of the management of elderly patients with non-small-cell lung cancer.Ann Oncol.2015;26(3):451-463. doi: 10.1093/annonc/mdu268.
- Paz-Ares LG, Zimmermann A, Ciuleanu T, et al. Meta-analysis examining impact of age on overall survival with pemetrexed for the treatment of advanced non-squamous non-small cell lung cancer.Lung Cancer.2017;104:45-51. doi: 10.1016/j.lungcan.2016.12.007.
- Leighl NB, Zatloukal P, Mezger J, et al. Efficacy and safety of bevacizumab-based therapy in elderly patients with advanced or recurrent nonsquamous non-small cell lung cancer in the phase III BO17704 study (AVAiL).J Thorac Oncol.2010;5(12):1970-1976. doi: 10.1097/JTO.0b013e3181f49c22.
- Langer CJ, Socinski MA, Patel JD, et al. Isolating the role of bevacizumab in elderly patients with previously untreated nonsquamous non-small cell lung cancer: secondary analyses of the ECOG 4599 and PointBreak trials.Am J Clin Oncol.2016;39(5):441-447. doi: 10.1097/COC.0000000000000163.
- Zhu J, Sharma DB, Gray SW, et al. Carboplatin and paclitaxel with vs without bevacizumab in older patients with advanced non-small cell lung cancer.JAMA.2012;307(15):1593-1601. doi:10.1001/jama.2012.454.
- Kutluk Cenik B, Sun H, Gerber DE. Impact of renal function on treatment options and outcomes in advanced non-small cell lung cancer.Lung Cancer.2013;80(3):326-332. doi: 10.1016/j.lungcan.2013.02.011.
- Rossi A, Maione P, Santabarbara G, et al. The safety of second-line treatment options for non-small cell lung cancer.Expert Opin Drug Saf.2017;16(4):471-479. doi: 10.1080/14740338.2017.1297795.
- Borghaei H, Paz-Ares L, Horn L, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer.N Engl J Med.2015;373(17):1627-1639. DOI: 10.1056/NEJMoa1507643.
- Rittmeyer A, Barlesi F, Waterkamp D, et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial.Lancet.2017;389(10066:255-265. doi: 10.1016/S0140-6736(16)32517-X.
- Santini FC, Rudin CM. Atezolizumab for the treatment of non-small cell lung cancer.Expert Rev Clin Pharmacol.2017;10(9):935-945. doi: 10.1080/17512433.2017.1356717.
- Fossella FV, DeVore R, Kerr RN, et al. Randomized phase III trial of docetaxel versus vinorelbine or ifosfamide in patients with advanced non-small-cell lung cancer previously treated with platinum-containing chemotherapy regimens. the TAX 320 Non-Small Cell Lung Cancer Study Group.J Clin Oncol.2000;18(12):2354-2362.
- Garon EB, Ciuleanu TE, Arrieta O, et al. Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial.Lancet.2014;384(9944):665-673. doi: 10.1016/S0140-6736(14)60845-X.
- Arrieta O, Zatarain-Barron ZL, Cardona AF, et al. Ramucirumab in the treatment of non-small cell lung cancer.Expert Opin Drug Saf.2017;16(5):637-644. doi: 10.1080/14740338.2017.1313226.
- Hanna N, Shepherd FA, Fossella FV, et al. Randomized phase III trial of pemetrexed versus docetaxel in patients with non-small-cell lung cancer previously treated with chemotherapy.J Clin Oncol.2004;22(9):1589-1597. doi: 10.1200/JCO.2004.08.163.
- Brulé S, Wheatley-Price P. Second line and maintenance therapy for advanced non-small cell lung cancer without driver mutation: an evolving paradigm.Int J Cancer Clin Res.2016;3(4):1-7.
- Paz-Ares L, de Marinis F, Dediu M, et al. Maintenance therapy with pemetrexed plus best supportive care versus placebo plus best supportive care after induction therapy with pemetrexed plus cisplatin for advanced non-squamous non-small-cell lung cancer (PARAMOUNT): a double-blind, phase 3, randomised controlled trial.Lancet Oncol.2012;13(3):247-255. doi: 10.1016/S1470-2045(12)70063-3.
- Paz-Ares LG, de Marinis F, Dediu M, et al. PARAMOUNT: final overall survival results of the phase III study of maintenance pemetrexed versus placebo immediately after induction treatment with pemetrexed plus cisplatin for advanced nonsquamous non-small-cell lung cancer.J Clin Oncol.2013;31(23):2895-2902. doi: 10.1200/JCO.2012.47.1102.
- Belani CP, Brodowicz T, Ciuleanu TE, et al. Quality of life in patients with advanced non-small-cell lung cancer given maintenance treatment with pemetrexed versus placebo (H3E-MC-JMEN): results from a randomised, double-blind, phase 3 study.Lancet Oncol.2012;13(3):292-299. doi: 10.1016/S1470-2045(11)70339-4.
- Lopez-Chavez A, Young T, Fages S, et al. Bevacizumab maintenance in patients with advanced non-small-cell lung cancer, clinical patterns, and outcomes in the Eastern Cooperative Oncology Group 4599 Study: results of an exploratory analysis.J Thorac Oncol.2012;7(11):1707-1712. doi: 10.1097/JTO.0b013e318265b500.
- Barlesi F, Scherpereel A, Rittmeyer A, et al. Randomized phase III trial of maintenance bevacizumab with or without pemetrexed after first-line induction with bevacizumab, cisplatin, and pemetrexed in advanced nonsquamous non-small-cell lung cancer: AVAPERL (MO22089).J Clin Oncol.2013;31(24):3004-3011. doi: 10.1200/JCO.2012.42.3749.
- Barlesi F, Scherpereel A, Gorbunova V, et al. Maintenance bevacizumab-pemetrexed after first-line cisplatin-pemetrexed-bevacizumab for advanced nonsquamous nonsmall-cell lung cancer: updated survival analysis of the AVAPERL (MO22089) randomized phase III trial.Ann Oncol.2014;25(5):1044-1052. doi: 10.1093/annonc/mdu098.
- Perol M, Chouaid C, Perol D, et al. Randomized, phase III study of gemcitabine or erlotinib maintenance therapy versus observation, with predefined second-line treatment, after cisplatin-gemcitabine induction chemotherapy in advanced non-small-cell lung cancer.J Clin Oncol.2012;30(28):3516-3524. doi: 10.1200/JCO.2011.39.9782.
- Ciuleanu T, Brodowicz T, Zielinski C, et al. Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: a randomised, double-blind, phase 3 study.Lancet.2009;374(9699):1432-1440. doi: 10.1016/S0140-6736(09)61497-5.
- Gentzler RD, Patel JD. Maintenance treatment after induction therapy in non-small cell lung cancer: latest evidence and clinical implications.Ther Adv Med Oncol.2014;6(1):4-15. doi: 10.1177/1758834013510589.
- Van Schil PE, Hendriks JM, Hertoghs M, et al. Current surgical treatment of non-small-cell lung cancer.Expert Rev Anticancer Ther.2011;11(10):1577-1585. doi: 10.1586/era.11.142.
- Paul S, Andrews WG, Nasar A, et al. Outcomes of lobectomy in patients with severely compromised lung function (predicted postoperative diffusing capacity of the lung for carbon monoxide % ≤ 40%).Ann Am Thorac Soc.2013;10(6):616-621. doi: 10.1513/AnnalsATS.201305-117OC.
- Falcoz PE, Puyraveau M, Thomas P-A, et al. Video-assisted thoracoscopic surgery versus open lobectomy for primary non-small-cell lung cancer: a propensity-matched analysis of outcome from the European Society of Thoracic Surgeons database.Eur J Cardiothorac Surg. 2016;49(2):602-609. doi: 10.1093/ejcts/ezv154.
- Shibamoto Y, Hashizume C, Baba F, et al. Stereotactic body radiotherapy using a radiobiology-based regimen for stage I non-small-cell lung cancer: five-year mature results.J Thorac Oncol.2015;10(6):960-964. doi: 10.1097/JTO.0000000000000525.
- Verstegen NE, Oosterhuis JW, Palma DA, et al. Stage I-II non-small-cell lung cancer treated using either stereotactic ablative radiotherapy (SABR) or lobectomy by video-assisted thoracoscopic surgery (VATS): outcomes of a propensity score-matched analysis.Ann Oncol.2013;24(6):1543-1548. doi: 10.1093/annonc/mdt026.
- Rossi D. What can we save for the first line treatment of NSCLC in 2016?Clin Oncol.2016;1:1176.

Powell Reviews Updated IO/TKI Data and AE Management in Endometrial Cancer
April 18th 2024During a Case-Based Roundtable® event, Matthew A. Powell, MD, discussed the case of a patient with advanced endometrial cancer treated with lenvatinib plus pembrolizumab who experienced grade 2 treatment-related hypertension.
Read More
Savona Discusses First-Line JAK Inhibition for Patients With Myelofibrosis at Risk of Anemia
April 17th 2024During a Case-Based Roundtable® event, Michael Savona, MD, and participants discussed the case of a patient with myelofibrosis and moderate anemia receiving JAK inhibitor therapy.
Read More
Biomarker Testing Paves the Way for Better Targeted Therapies in NSCLC
April 16th 2024At a live virtual event, Edward S. Kim, MD, MBA, discussed the evolving landscape of biomarker testing before making treatment decisions for patients with early-stage non–small cell lung cancer (NSCLC).
Read More