Current and Emerging Treatment Options in Chronic Lymphocytic Leukemia
CLL is a malignant disease characterized by progressive accumulation of immature, immunophenotypically distinct lymphocytes in the blood, bone marrow, and lymphatic tissues. Although the clinical course of CLL can be unpredictable, this disease is typically slow-progressing and occurs most often in patients ≥65 years of age. As such, treatment, if indicated, is typically conservative. Current CLL treatments are not considered to be curative; however, long-term remission can be achieved in some patients using allogenic stem cell transplantation.

Chronic lymphocytic leukemia (CLL) is a malignant disease characterized by progressive accumulation of immature, immunophenotypically distinct (CD5- and CD23-positive) lymphocytes in the blood, bone marrow, and lymphatic tissues.1Although the clinical course of CLL can be unpredictable, this disease is typically slow-progressing and occurs most often in patients ≥65 years of age.1As such, treatment, if indicated, is typically conservative.1Current CLL treatments are not considered to be curative; however, long-term remission can be achieved in some patients using allogenic stem cell transplantation.1This article focuses on the shifting treatment options observed in recent years, both in first-line and subsequent settings, and the emerging treatment strategies that are likely to continue to shape the therapeutic landscape for patients with CLL.
TREATMENT VARIABLES
Given the highly variable clinical presentation and progression of CLL, not all patients require treatment at the time of diagnosis. In fact, most patients undergo active surveillance for many years before treatment.2However, treatment is indicated for CLL that is classified as being at an advanced clinical stage; as progressive bulky disease; or has transformed to B-cell/Hodgkin lymphoma (ie, Richter syndrome).3In addition, patients with CLL are indicated for treatment if they have significant disease-related symptoms (ie, severe fatigue, night sweats, weight loss, fever without infection [B-type symptoms]), threatened end-organ function, progressive anemia, and progressive thrombocytopenia.3
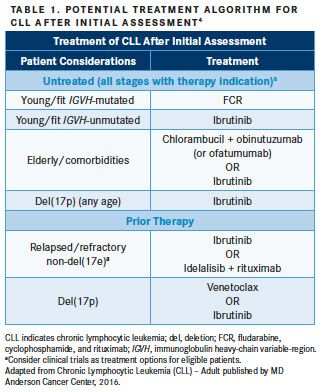
Treatment selection for CLL is based on distinct molecular profiles of the disease and takes into consideration the patient’s age and fitness level (TABLE 1).4Genetic aberrations that currently guide treatment decisions include p17 deletion (del[17p]) andTP53mutations, as well as immunoglobulin heavy-chain variable region gene (IGHV) mutational status.3,4In CLL, del(17p) andTP53mutations are associated with poor responses to chemotherapy-based regimens and are predictors of poor patient outcomes including shorter median progression-free survival (PFS) and median overall survival (OS).5,6Accordingly, this subgroup of patients with CLL requires specific therapeutic approaches.3IGHVmutational status has been demonstrated as a prognostic factor in CLL, as unmutatedIGHVis associated with poorer responses to chemotherapy and shorter median PFS compared with mutatedIGHV.3,6For example, recent data demonstrate long-term disease-free-survival in patients withIGHV-mutated CLL who received fludarabine, cyclophosphamide, and rituximab (FCR),7suggestingIGHVmutational status can be used as a biomarker of response, and these patients can potentially be cured when using FCR in the frontline setting. Although not incorporated into the guidelines to direct treatment decisions, the National Comprehensive Cancer Network recommends IGHV mutation testing based on reproducibility and availability.3However, leading oncology treatment centers, including The University of Texas MD Anderson Cancer Center and Mayo Clinic, currently stratify patients according toIGHVmutational status to guide treatment decisions.2,4
Recent years have seen significant changes to the therapeutic landscape for CLL treatment. The historical standard first-line therapy of chlorambucil was replaced with more effective treatment regimens including chemoimmunotherapy, combining standard chemotherapy and immunotherapy with monoclonal antibodies targeting CD20 (eg, rituximab, obinutuzumab, and ofataumumab) and targeted therapy for Bruton tyrosine kinase (BTK) with the oral small-molecule inhibitor ibrutinib.3Similarly, in patients with relapsed or refractory (R/R) CLL, small-molecule inhibitors including ibrutinib, idelalisib (a PI3K inhibitor), and venetoclax (a B-cell lymphoma 2 inhibitor) have become standards of care.3These treatments are not likely to be the last in the shifting paradigm of CLL treatment; current ongoing and planned studies in both treatment-naïve and treatment-experienced patients with CLL will no doubt bring further changes to the first-line, second-line, and subsequent treatment settings for this disease.
FIRST-LINE TREATMENT OPTIONS IN CLL
Combined Chemotherapy and Immunotherapy With Anti-CD20 Monoclonal Antibodies
The chemoimmunotherapy regimen FCR is the standard of care for patients with CLL without del(17p) orTP53mutation who are under the age of 65, according to a guideline based on the groundbreaking phase III trial CLL8 (the German CLL Study Group) that evaluated 817 physically fit, previously untreated patients with CLL.3,6The addition of rituximab to fludarabine and cyclophosphamide over 6 courses of treatment significantly improved the objective response rate ([ORR], 90% vs 80%;P<.0001) and complete response rate (44% vs 22%;P<.0001) in these patients.6Additionally, at 3 years, median PFS (65% vs 45%; HR, 0.56; 95% CI, 0.46-0.69;P<.001) and OS (87% vs 83%; HR, 0.76; 95% CI, 0.49-0.92;P= .012) were also significantly improved.6The survival benefits of FCR versus fludarabine and cyclophosphamide in this patient population was further confirmed in updated study results demonstrating a 5-year OS of 80.9% versus 69.2%, respectively (HR, 0.63; 95% CI, 0.47-0.84;P= .02).8In this study, the strongest predictors of shorter median PFS and shorter median OS wereTP53mutation, del(17p), and unmutatedIGHV.8,9Additionally, patients with mutated IGHV achieved a significantly longer median PFS with FCR compared with fludarabine and cyclophosphamide (PFS, median not reached [NR] vs 41.9 months; HR, 0.47; 95% CI, 0.33-0.68;P<.001).8As such, this study clearly validated the benefit of anti-CD20 monoclonal antibody use in combination with chemotherapy for the treatment of patients with CLL and specific mutations (without del[17p]/TP53).10
Bendamustine plus rituximab (BR) is another preferred first-line regimen for the treatment of patients under the age of 65 with CLL lacking del(17p) orTP53mutations.3This patient population was examined in CLL10, a phase III, international, open-label randomized trial that compared BR treatment with FCR.11In this study, similar ORR (95% vs 96%) and OS rates (3 years, 91% vs 92%; HR, 1.034; 95% CI, 0.620-1.724) were observed between treatment groups.11The primary endpoint was PFS to demonstrate noninferiority of BR to FCR.11The median PFS was significantly shorter for BR versus FCR (41.7 vs 55.2 months; HR, 1.634; 90.4% CI, 1.308-2.064;P= .0003) (Table 1).11BR treatment resulted in fewer complete responses (CRs), although the difference was not statistically significant (31% vs 40%), and more patients treated with BR had minimal residual disease. In elderly patients (>65 years), median PFS was similar between treatment groups (BR, 48.5 months; FCR, median NR); however, FCR produced more incidences of neutropenia, leukocytopenia, infections, and secondary neoplasms than did BR in this patient population (≥65 years).11
Other anti-CD20 monoclonal antibodies indicated for the treatment of CLL in the frontline setting are obinutuzumab and ofatumumab; both are recommended as first-line therapy in combination with chlorambucil in elderly patients and those with significant comorbidities who do not have del(17p) orTP53mutations.3In the CLL11 study, obinutuzumab plus chlorambucil showed significant improvement in ORR and PFS over chlorambucil alone and over rituximab plus chlorambucil. Median PFS was 26.7, 11.1, and 16.3 months for the obinutuzumab plus chlorambucil, chlorambucil alone, and the rituximab plus chlorambucil groups, respectively (P <.001),12 while ORRs were 77.3%, 31.4%, and 65.7%, respectively (P<.001).12On the basis of this study, obinutuzumab plus chlorambucil is considered the standard of care for elderly patients and for those with significant comorbidities.3
Ofatumumab, originally approved for fludarabine- and alemtuzumab-refractory CLL, recently received frontline indication in CLL based on the findings of the COMPLEMENT study, an open-label, phase III study in treatment-naïve patients (n = 447) who were not eligible for fludarabine-based therapy due to age or comorbidities.13The results of this study demonstrated that ofatumumab plus chlorambucil compared with chlorambucil alone significantly improved ORR (82% vs 69%; odds ratio [OR], 2.16; 95% CI, 1.36-3.42;P= .001) and median PFS (22.4 vs 13.1 months; HR, 0.57; 95% CI, 0.45-0.72;P<.0001).13
Inhibition of BTK With Small Molecules
Interest in targeting BTK, a key player in the B-cell antigen receptor survival pathway, resulted in the approval of ibrutinib for the treatment of R/R patients with CLL. In 2014, ibrutinib also became available as first-line therapy for both del(17p) and non-del(17p) CLL.14Currently, ibrutinib is the only small-molecule targeted therapy that is approved by the FDA for first-line treatment of CLL.3
The phase III RESONATE study demonstrated significant ORR, PFS, and OS benefits with ibrutinib compared with ofatumumab in previously treated patients with advanced or high-risk CLL, both with and without del(17p); a 57% reduction in risk of death was observed.15Significant improvements in hematologic function (improved platelet and neutrophil counts, improved hemoglobin levels) were observed in patients with cytopenia treated with ibrutinib compared with ofatumumab (79% vs 43%;P<.0001).16Additionally, ibrutinib treatment resulted in clinically meaningful improvements in patient-reported outcomes measured by the Functional Assessment of Chronic Illness Therapy-Fatigue measurement system.16
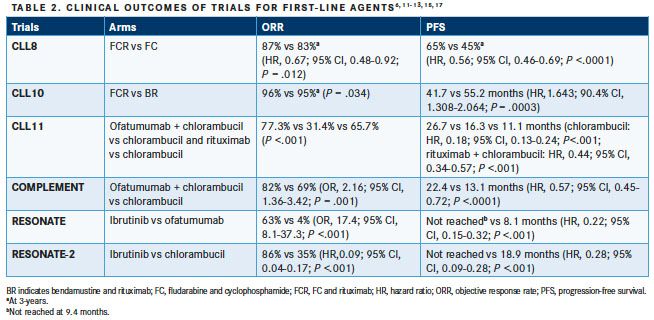
The RESONATE-2 study supported the use of ibrutinib in the frontline setting for CLL, demonstrating improved ORR and median PFS compared with chlorambucil in treatment-naïve, elderly (≥65 years) patients without del(17p).17Although this open-label, phase III trial excluded patients with known del(17p) CLL,171 study demonstrated ibrutinib efficacy in patients with del(17p) and TP53 mutations including both previously untreated patients and those with R/R CLL; ORRs of 97% and 80% were observed with ibrutinib treatment for treatment-naïve patients and those with R/R CLL, respectively, after a median 2-year follow-up for all patients.18 Of note, an integrated analysis of the results from multiple phase III studies in CLL (RESONATE, RESONATE-2, and HELIOS) showed that IGHV mutational status does not predict response to ibrutinib.19TABLE 2 lists clinical trial outcomes for first-line agents for the treatment of CLL.6,11-13,15,17
More recently, follow-up data from studies assessing ibrutinib in treatment-naïve and those with R/R CLL (PCYC-1102 and -1103 studies) demonstrated sustained efficacy of single-agent ibrutinib at 5 years; the ORR in treatment-naïve and treatment-experienced patients was 87% and 89%, respectively, with CR rates improving over time.20 Increased tolerability was also observed with long-term use, as treatment-limiting adverse events (AEs) decreased from years 1 to 5.20
Based on these data, ibrutinib is recommended as first-line therapy for patients with CLL without del(17p) or TP53 mutations who are either frail, elderly (≥65 years), or who are younger (<65 years) but with significant comorbidities, and for patients with CLL with del(17p) or TP53 mutations.3
Both chemoimmunotherapy and ibrutinib treatment are preferred regimens used in the frontline setting for CLL without del(17p) or TP53 mutations.3A recent cross-trial comparison of multiple phase III CLL studies (ie, CLL8, CLL10, CLL11, COMPLEMENT, RESONATE-2) directly compared the outcomes of ibrutinib and several chemoimmunotherapy regimens.21Results demonstrated that ibrutinib resulted in longer PFS than chemoimmunotherapy regimens among the entire sample population and in high-risk patients (ie, those with advanced and bulky disease, unmutated IGHV, del[11q]).21The OS achieved by ibrutinib was longer than chemoimmunotherapy in older, less-fit subgroups (eg, patients with presence of comorbidities, poor Eastern Cooperative Oncology Group [ECOG] performance status, or high Cumulative Illness Rating Scale [CIRS] scores), but similar to the OS achieved by chemoimmunotherapies FCR and BR in younger, more fit patient populations (eg, those with less comorbidities, good ECOG performance status, and low CIRS scores).21In addition, the overall safety profile of ibrutinib was more favorable than that of chemoimmunotherapy regimens, with reduced frequency of grade 3 AEs including infections and neutropenia.21The results of this study suggest that the benefits of ibrutinib use outweigh its risks, and ibrutinib use in the frontline setting may spare some patients toxic chemotherapy while providing improved responses.21Further, a recent pilot study in CLL demonstrated that BTK inhibition was effectively maintained over 3 cycles of ibrutinib therapy even when dosage reductions were employed after the first 28-day cycle of therapy (from 420 mg/day to 140 mg/day), potentially decreasing patient costs and unwanted AEs.22
TREATMENTS FOR R/R CLL
In addition to inhibition of BTK with ibrutinib, other preferred regimens for the treatment of patients with R/R CLL are idelasib or venetoclax in combinations with rituximab.3 The availability of these agents has significantly improved the outcome of patients with R/R CLL, including those with high-risk features (del[17]p), TP53 mutations, unmutated IHGV), extending the PFS and OS of these patients and improving their response rates.
Idelalisib Plus Rituximab
Idelalisib is an oral PI3K-delta inhibitor. PI3K plays a key role in the B-cell antigen receptor survival pathway. As such, its inhibition leads to increased apoptosis.23Approval was based on results from a phase III trial demonstrating that idelalisib plus rituximab compared with rituximab alone resulted in significantly longer median PFS (NR vs 5.5 months; HR, 0.15; 95% CI, 0.08-0.28;P<.001) and median OS (92% vs 80% at 12 months; HR, 0.28; 95% CI, 0.09-0.86;P= .02) and improved ORR (77% vs 15%; OR, 19.6; 95% CI, 9.6-39.9) in patients with R/R CLL.24,25
Venetoclax Plus Rituximab
Venetoclax is a B-cell lymphoma 2 inhibitor that was shown to promote apoptosis in CLL cells.26An ORR of 79.4% was observed in a phase II study of single-agent venetoclax in patients with R/R CLL with del(17p).27Additionally, the median PFS and OS were estimated to be 72% and 86.7%, respectively, at 12 months with single-agent venetoclax treatment.27
Recent study results from the phase III MURANO study in patients with R/R CLL demonstrated significantly better outcomes with venetoclax plus rituximab than with BR. After nearly 2 years, estimated PFS rate for the venetoclax plus rituximab group was 84.9%, compared with 36.3% of the BR group (HR, 0.17; 95% CI, 0.11-0.25;P<.0001).28On the basis of these data, venetoclax received FDA approval in June 2018 for the treatment of patients with CLL with or without del(17p) who had undergone at least 1 previous therapy.29
MAINTENANCE THERAPY
Treatment with lenalidomide can be considered for maintenance therapy for patients with high-risk CLL after first-line therapy or for patients who responded (CR or partial response [PR]) after therapy for R/R disease.3 The latter is supported by data from the CONTINUUM trial, which demonstrated a longer median PFS with lenalidomide than placebo (33.9 vs 9.2 months; HR, 0.40; 95% CI, 0.29-0.55;P<.001), although no differences in median OS were observed (HR, 0.96; 95% CI, 0.63-1.48; P = .86).30
Similarly, ofatumumab is a recommended maintenance therapy for patients with CLL who responded (CR or PR) after treatment for R/R disease.3PROLONG, a phase III, open-label, randomized trial, investigated ofatumumab versus observation and demonstrated an improved median PFS (29.4 vs 15.2 months; HR, 0.50; 95% CI, 0.38-0.66;P<.0001) in patients with CLL who were in remission after second-line therapy.31
EMERGING TREATMENT STRATEGIES: ONGOING COMBINATION TRIALS
Despite recent progress to improve outcomes for patients with CLL, including the recent changes in the therapeutic landscape to first-line and subsequent use of new chemoimmunotherapy and targeted therapies, other treatment strategies are still needed for some patients who do not respond.2In addition, the advent of new biologic therapies brings with it new challenges, such as agent-specific toxicities, that need to be managed. As such, new treatment strategies are emerging and include novel treatment schedules and fresh combinations of small-molecule inhibitors, chemotherapy, and immunomodulatory agents (TABLE 3).32-34
CAPTIVATE: Ibrutinib Lead-In Followed by Ibrutinib Plus Venetoclax
The combination of ibrutinib plus venetoclax is supported by early studies that demonstrated increased antitumor activity when used sequentially; to this end, BTK inhibition with ibrutinib has been shown to enhance CLL cell killing with venetoclax.35This concept is currently being tested in the ongoing phase II CAPTIVATE study in treatment-naïve patients with CLL; in this trial, patients receive ibrutinib monotherapy for 3 cycles followed by ibrutinib plus venetoclax for at least 12 cycles. Early data suggest this treatment regimen is safe and effective, with no dose-limiting toxicities or occurrence of clinical tumor lysis syndrome and an ORR of 100% (CR and PR; 14 patients).32,33
GA-101: Ibrutinib, Fludarabine, Cyclophosphamide, and Obinutuzumab
An ongoing phase II study (GA-101) is investigating the combination of ibrutinib, fludarabine, cyclophosphamide, and obinutuzumab as first-line treatment for CLL with mutated IGHV without del(17p).34The estimated completion date for this study is March 2022.
CONCLUSIONS
As published findings continue to demonstrate the benefit, safety, and tolerability of BTK inhibitors, both as monotherapies and in combination with other agents, the therapeutic outlook for CLL is potentially promising.
References:
- Chronic lymphocytic leukemia treatment (PDQ) - health professional version. National Cancer Institute website. https://www.cancer.gov/types/leukemia/hp/cll-treatment-pdq. Updated February 7, 2018. Accessed October 14, 2018.
- Strati P, Jain N, O’Brien S. Chronic lymphocytic leukemia: diagnosis and treatment. Mayo Clin Proc. 2018;93(5):651-664. doi: 10.1016/j.mayocp.2018.03.002.
- National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology (NCCN Guidelines): Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma. Version 2.2019. NCCN website. www.nccn.org/professionals/physician_gls/pdf/cll.pdf. Published October 5, 2018. Accessed October 11, 2018.
- Chronic Lymphocytic Leukemia (CLL): adult. MD Anderson Cancer Center website. https://www.mdanderson.org/documents/for-physicians/algorithms/cancer-treatment/ca-treatment-cll-web-algorithm.pdf. Published December 13, 2016. Accessed October 13, 2018.
- Döhner H, Stilgenbauer S, Benner A, et al. Genomic aberrations and survival in chronic lymphocytic leukemia. N Engl J Med. 2000;343(26):1910-1916. doi: 10.1056/NEJM200012283432602.
- Hallek M, Fischer K, Fingerle-Rowson G, et al. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Lancet. 2010;376(9747):1164-1174.
- Thompson PA, Tam CS, O’Brien SM, et al. Fludarabine, cyclophosphamide, and rituximab treatment achieves long-term disease-free survival in IGHV-mutated chronic lymphocytic leukemia. Blood. 2016;127(3):303-309. doi: 10.1182/blood-2015-09-667675.
- Fischer K, Bahlo J, Fink AM, et al. Long-term remissions after FCR chemoimmunotherapy in previously untreated patients with CLL: updated results of the CLL8 trial. Blood. 2016;127(2):208-215. doi: 10.1182/blood-2015-06-651125.
- Woyach JA, Ruppert AS, Heerema NA, et al. Chemoimmunotherapy with fludarabine and rituximab produces extended overall survival and progression-free survival in chronic lymphocytic leukemia: long-term follow-up of CALGB study 9712. J Clin Oncol. 2011;29(10):1349-1355. doi: 10.1200/JCO.2010.31.1811.
- Kipps TJ, Stevenson FK, Wu CJ, et al. Chronic lymphocytic leukaemia [published correction appears in Nat Rev Dis Primers. 2017;3:17008. doi:10.1038/nrdp.2017.8]. Nat Rev Dis Primers. 2017;3:16096. doi: 10.1038/nrdp.2016.96.
- Eichhorst B, Fink AM, Bahlo J, et al. International Group of Investigators; German CLL Study Group. First-line chemoimmunotherapy with bendamustine and rituximab versus fludarabine, cyclophosphamide, and rituximab in patients with advanced chronic lymphocytic leukaemia (CLL10): an international, open-label, randomised, phase 3, non-inferiority trial. Lancet Oncol. 2016;17(7):928-942. doi: 10.1016/S1470-2045(16)30051-1.
- Goede V, Fischer K, Busch R, et al. Obinutuzumab plus chlorambucil in patients with CLL and coexisting conditions. N Engl J Med. 2014;370(12):1101-1110. doi: 10.1056/NEJMoa1313984.
- Hillmen P, Robak T, Janssens A, et al; COMPLEMENT 1 Study Investigators. Chlorambucil plus ofatumumab versus chlorambucil alone in previously untreated patients with chronic lymphocytic leukaemia (COMPLEMENT 1): a randomised, multicentre, open-label phase 3 trial. Lancet. 2015;385(9980):1873-1883. doi: 10.1016/S0140-6736(15)60027-7.
- Imbruvica approval package. Food and Drug Administration website. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2014/205552s001.pdf. Published July 28, 2014. Accessed October 12, 2018.
- Byrd JC, Brown JR, O’Brien S, et al. RESONATE Investigators. Ibrutinib versus ofatumumab in previously treated chronic lymphoid leukemia. N Engl J Med. 2014;371(3):213-223. doi: 10.1056/NEJMoa1400376.
- Barrientos JC, O’Brien S, Brown JR, et al. Improvement in parameters of hematologic and immunologic function and patient well-being in the phase III RESONATE study of ibrutinib versus ofatumumab in patients with previously treated chronic lymphocytic leukemia/small lymphocytic lymphoma [published online August 18, 2018]. Clin Lymphoma Myeloma Leuk. doi: 10.1016/j.clml.2018.08.007.
- Burger JA, Tedeschi A, Barr PM, et al; RESONATE-2 Investigators. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N Engl J Med. 2015;373(25):2425-2437. doi: 10.1056/NEJMoa1509388.
- Farooqui MZ, Valdez J, Martyr S, et al. Ibrutinib for previously untreated and relapsed or refractory chronic lymphocytic leukaemia with TP53 aberrations: a phase 2, single-arm trial. Lancet Oncol. 2015;16(2):167-176. doi: 10.1016/S1470-2045(14)71182-9.
- Kipps TJ, Fraser G, Coutre S, et al. Unmutated IGHV is not an adverse predictor of outcomes to therapy with ibrutinib in patients with chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) [ACCR abstract CT158]. Cancer Res 2017;77(13 suppl; abstr CT158). doi: 10.1158/1538-7445.AM2017-CT158.
- O’Brien S, Furman RR, Courte S, et al. Single-agent ibrutinib in treatment-naïve and relapsed/refractory chronic lymphocytic leukemia: a 5-year experience. Blood. 2018;131(17):1910-1919. doi: 10.1182/blood-2017-10-810044.
- Robak T, Burger JA, Tedeschi A, et al. Single-agent ibrutinib versus chemoimmunotherapy regimens for treatment-naïve patients with chronic lymphocytic leukemia: a cross-trial comparison of phase 3 studies. A J Hematol. 2018;93(11):1402-1410. doi: 10.1002/ajg.25259.
- Chen LS, Bose P, Cruz ND, et al. A pilot study of lower doses of ibrutinib in patients with chronic lymphocytic leukemia [published online September 25, 2018]. Blood. doi: 10.1182/blood-2018-06-860593.
- Ringshausen I, Schneller F, Bogner C, et al. Constitutively activated phosphatidylinositol-3 kinase (PI-3K) is involved in the defect of apoptosis in B-CLL: association with protein kinase C-delta. Blood. 2002;100(10):3741-3748.
- Furman RR, Sharman JP, Coutre SE, et al. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N Engl J Med. 2014;370(11):997-1007. doi: 10.1056/NEJMoa1315226.
- Sharman JP, Coutre S, Furman RR, et al. Second interim analysis of a phase 3 study of idelalisib (Zydelig) plus rituximab (R) for relapsed chronic lymphocytic leukemia (CLL): efficacy analysis in patient subpopulations with del(17p) and other adverse prognostic factors. Blood. 2014;124(21):(abstr 330).
- Anderson MA, Deng J, Seymour JF, et al. The BCL2 selective inhibitor venetoclax induces rapid onset apoptosis of CLL cells in patients via a TP-53 independent mechanism. Blood. 2016;127(25):3215-3224. doi: 10.1182/blood-2016-01-688796.
- Stilgenbauer S, Eichhorst B, Schetelig J, et al. Venetoclax in relapsed or refractory chronic lymphocytic leukaemia with 17p deletion: a multicentre, open-label, phase 2 study. Lancet Oncol. 2016;17(6):768-778. doi: 10.1016/S1470-2045(16)30019-5.
- Seymour JF, Kipps TJ, Eichhorst BF, et al. Venetoclax-rituximab in relapsed or refractory chronic lymphocytic leukemia. N Engl J Med. 2018;378(12):1107-1120. doi: 10.1056/NEJMoa1713976.
- Venetoclax supplemental approval letter. FDA website. accessdata.fda.gov/drugsatfda_docs/appletter/2018/208573Orig1s004s005ltr.pdf. Published June 8, 2018. Accessed October 14, 2018.
- Chanan-Khan AA, Zaritskey A, Egyed M, et al. Lenalidomide maintenance therapy in previously treated chronic lymphocytic leukaemia (CONTINUUM): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Haematol. 2017;4(11):534-543. doi: 10.1016/S2352-3026(17)30168-0.
- van Oers MH, Kuliczkowski K, Smolej L, et al; PROLONG Study Investigators. Ofatumumab maintenance versus observation in relapsed chronic lymphocytic leukaemia (PROLONG): an open-label, multicentre, randomized phase 3 study. Lancet Oncol. 2015;16(13):1370-1379. doi: 10.1016/S1470-2045(15)00143-6.
- Wierda WG, Siddiqi T, Flinn I, et al. Phase 2 CAPTIVATE results of ibrutinib (ibr) plus venetoclax (ven) in first-line chronic lymphocytic leukemia (CLL). Presented at: 2018 American Society of Clinical Oncology Annual Meeting; June 1-5, 2018; Chicago, IL. Abstract 7502.
- Ibrutinib plus venetoclax in subjects with treatment-naive chronic lymphocytic leukemia/small lymphocytic lymphoma (CAPTIVATE). clinicaltrials.gov/ct2/show/NCT02910583. Updated September 26, 2108. Accessed October 15, 2018.
- Ibrutinib, fludarabine phosphate, cyclophosphamide, and obinutuzumab in treating patients with chronic lymphocytic leukemia (CLL). clinicaltrials.gov/ct2/show/NCT2629809. Updated October 5, 2108. Accessed October 15, 2018.
- Deng J, Isik E, Fernandes SM, et al. Bruton’s tyrosine kinase inhibition increases BCL-2 dependence and enhances sensitivity to venetoclax in chronic lymphocytic leukemia. Leukemia.2017;31(10):2075-2084. doi: 10.1038/leu.2017.32.
Novel Approaches Focus on Limiting Toxicity in Older Patients With ALL
April 22nd 2024The major challenges for clinicians treating older patients with acute lymphoblastic leukemia surround the emergence of resistance to existing therapies and the toxicities associated with current chemotherapies.
Read More
Ornstein Advises on Starting Dose and Management of Lenvatinib in RCC
April 21st 2024During a Case-Based Roundtable® event, Moshe Ornstein, MD, MA, provided guidance on dosing and toxicity concerns in a patient treated with lenvatinib plus pembrolizumab for advanced renal cell carcinoma.
Read More