The Current and Future Treatment Landscape of Cutaneous Squamous Cell Carcinoma
Although it is the second most common form of skin cancer in the United States, cutaneous squamous cell carcinoma can often be effectively treated with a variety of surgical modalities, such as standard excision, Mohs micrographic surgery, curettage and electrodessication, and cryosurgery.
1,2A subset of patients with high-risk tumors, such as those who experience local recurrence and nodal metastases, has driven development within the therapeutic landscape that has resulted in the recent FDA approval of the first agent for the treatment of cSCC.3,4Moreover, development continues on other investigational agents, which, if approved, would give patients more therapeutic options and also potentially decrease the large financial burden of this common cancer.
Surgical Treatment
Standard excision using “bread loaf” histopathologic sectioning requires a margin of skin that appears to be clinically normal around the tumor and surrounding erythema, to ensure complete removal of the cSCC with histologically negative margins.5The National Comprehensive Cancer Network guidelines recommend standard excision, using clinical margins of 4 mm to 6 mm as a primary treatment for localized, low-risk cSCC.2An expert work group agrees with the use of standard excision with a 4 mm to 6 mm margin of uninvolved skin around the biopsy site and tumor, extending down to mid-subcutaneous adipose tissue for histologic margin assessment in patients with low-risk primary cSCC.5
Although standard excision may be considered for highrisk tumors, complete margin assessment is recommended prior to selecting a treatment modality.5If standard excision is performed on high-risk tumors, clinicians should consider the use of a linear repair, skin graft, or healing by second intention.5Importantly, closure should be delayed until confirmation of negative histologic margins, especially in cases of repair that require significant tissue rearrangement.5
A systematic review and pooled analysis of 12 studies reported an overall local recurrence of 5.4% during followup after surgical excision.6These studies used inconsistent surgical margins, ranging from 2 mm to >10 mm, with some decisions regarding excision margins based on clinical diagnosis and surgeon preference.6Overall, authors reported a pooled average estimate of 8.8% for the frequency of incomplete surgical excisions.6
Mohs Micrographic Surgery
The use of chemosurgery was first described for the removal of recurrent or difficult-to-treat cutaneous tumors in the 1940s.7This method was updated approximately 30 years later to eliminate the use of zinc chloride fixative paste (which contributed to patient discomfort), and to improve conservation of tissue by mitigating fixation beyond surgical margins and decreasing the amount of time required for the procedure.8This technique, referred to as microscopically controlled excision, involved injection with 1% lidocaine with epinephrine into the area of suspected tumor, which was then curetted. An excision of tissue approximately 3 mm to 5 mm thick was taken from the tumor bed, extending an additional 1 mm to 3 mm around the periphery of the wound. The tissue was briefly fixed in formaldehyde solution, and sections were mounted and stained for examination.8This technique was successful. Of the 102 lesions treated, only 3 recurrences were reported during a follow-up of 3 to 5 years for 76 tumors, and 5 to 8 years for 26 tumors. These recurrences were reported within 12 months, retreated, and were followed without reports of additional recurrence.8
Following these reports, the use of MMS expanded, and appropriate-use criteria for MMS were published in 2012.9A systematic review reported that for primary cSCC treated with MMS, the 5-year local recurrence rate was 3.1%, whereas the 5-year local recurrence rates with C&E, surgical excision, and radiation therapy were 3.7%, 8.1%, and 10.0%, respectively.10Upon consideration of high-risk characteristics, the cure rate with MMS was 74.8% for tumors ≥2 cm and 67.4% for poorly differentiated tumors, compared with 58.3% and 46.4%, respectively, with non-Mohs modalities.10The 5-year rates for locally recurrent cSCC were reported to be 10.0% for cSCC treated with MMS and 23.3% for cSCC treated with surgical excision.10MMS has been recommended for the treatment of high-risk cSCC.5
Curettage and Electrodessication
C&E is regularly used for the treatment of low-risk cSCC and can be considered for the treatment of small, low-risk, primary cSCC, as well.5A pooled analysis demonstrated a recurrence rate of 1.7% following treatment with C&E.6A study of 257 basal cell carcinomas (BCCs) revealed no major cosmetic adverse events (AEs) following C&E.11In some cases, depressed scars on the nose or tissue contraction on the lips were reported.11
Nonsurgical Treatment
cSCC is often successfully treated with surgical modalities; however, if surgical therapy is not feasible, low-risk tumors may be treated with nonsurgical modalities. Radiation therapy may be considered for low-risk tumors, because the rate of cure may be diminished.5Cryosurgery is an option for low risk cSCC if other therapies are contraindicated or impractical. Topical therapies, including imiquimod, 5-fluorouracil, and photodynamic therapy (PDT), are not evidence-based recommendations. Additionally, due to lack of data, no official recommendations have been made with respect to the use of laser therapies or electronic surface brachytherapy.5
Radiation Therapy
Primary radiation therapy can be used for patients for whom surgery is contraindicated, not feasible, or not preferred. Different types of radiotherapy have been used to treat cSCC, including external electron beam radiation, superficial radiation therapy, and isotope-based brachytherapy. Radiation therapy has been shown to result in good tumor control and cosmesis in select patients with cSCC.12However, disease-specific survival at 4 years was shown to be only 60% in patients with cSCC, compared with 100% in patients with BCC following radiation therapy.13Following surgical treatment, patients with primary cSCC and perineural invasion or those at high risk for metastasis may be considered for adjuvant radiation therapy.14
Topical Therapy
Because only case reports or small case series have been reported regarding the treatment of cSCC with topical imiquimod, it is not currently recommended for cSCC.15,165-fluorouracil has been used both alone and in combination as a treatment for cSCC, but it has been associated with erythema, erosions, and crust that can last for ≥1 month, which can lead to decreased treatment adherence and diminished effectiveness.5Imiquimod has also been shown to lead to erythema, edema, erosions, ulceration, and crust. This can vary among patients, leading to difficulties with dosing decisions. When imiquimod is used on larger surface areas, systemic symptoms may be reported, such as headache, fatigue, myalgia, and influenza-like symptoms.5
Energy Devices
The use of PDT has demonstrated benefits in high-risk patients with invasive cSCC, including solid organ transplant recipients, when used in combination with curettage or surgery; however, the specific contribution of PDT to the observed outcomes is unclear.17,18
Management of Metastatic Disease
Patients with cSCC have been reported to have a risk for metastasis of 3.7%.3Recommendations for therapy are based on the extent of disease; the primary choices consist of surgical resection, possible lymph node dissection, and the use of adjuvant radiation therapy with or without concurrent systemic therapy.5Multidisciplinary decision-making is recommended for metastatic cSCC because of the rarity and complexity of disease. Palliative and supportive care should be offered to patients with advanced disease, to improve symptom management and quality of life.5
Chemotherapy has demonstrated some activity in patients with distant metastatic cSCC. In a study of patients treated with cisplatin-based chemotherapy, 28% achieved complete remission and 40% achieved partial remission following the use of chemotherapy alone. Moreover, all patients who received surgery following cisplatin-based chemotherapy (n = 5) achieved complete remission, with only 1 patient relapsing. Additionally, 7 of 8 patients who received radiation therapy following cisplatin-based chemotherapy achieved complete remission, with no relapses reported.19A study investigating the combined effects of interferon alfa, retinoic acid, and cisplatin demonstrated response rates of 67% in patients with locally advanced disease and 17% in those with metastatic disease, yielding an overall response rate (ORR) of 34%.20
EGFR Inhibitors
EGFR inhibitors have also been investigated for the treatment of patients with advanced unresectable cSCC. In a study of 36 patients treated with cetuximab, the disease control rate was 69%; 2 patients achieved complete responses and 8 achieved partial responses.21The most frequently reported AE was a grade 1 to 2 acne-like rash in 87% of the per-protocol population.21 Grade 3 or 4 serious adverse events (SAEs) were reported in 61% of patients; only 10% of the events were related to cetuximab.21A total of 4 patients experienced an SAE and discontinued treatment, whereas other patients experiencing grade 3 or 4 SAEs continued cetuximab without a dose reduction.21
Results from a phase II study of 16 patients treated with panitumumab demonstrated a best ORR of 31%, including 3 partial responses, 2 complete responses, and 6 patients achieving stable disease.22Of the patients who experienced complete responses, 1 relapsed with distant metastatic disease at 16 months after the last panitumumab dose; the other patient received 7 panitumumab infusions and exhibited no evidence of disease at 24 months following the last dose.22No hypersensitivity reactions related to panitumumab were reported at the first infusion.22
Immunotherapy
Immunocompromised patients, particularly those who are solid organ transplant recipients, have a risk for cSCC, which is increased by 198-fold among heart and lung recipients, 121-fold among kidney recipients, and 32-fold among liver recipients.23These figures suggest that immune surveillance may be critical for the prevention of cSCC in this population. A case report of patients with BCC and cSCC who were treated with an antibody directed against the PD-1 immune checkpoint receptor suggests that these cancers are sensitive to PD-1 blockade.24
Cemiplimab, a human monoclonal antibody that targets PD-1, has been investigated as a treatment for patients with metastatic or locally advanced cSCC.25In expansion cohorts of a phase I study investigating cemiplimab in patients with metastatic or locally advanced cSCC, 50% of patients achieved a response.25
Phase II results in the metastatic-disease cohort reported a response in 47% of patients, with the duration of response exceeding 6 months in 57% of patients, and 82% continuing to exhibit a response with cemiplimab treatment at data cutoff.25Frequently reported AEs in the phase II portion of the study included fatigue, diarrhea, nausea, constipation, and rash. Treatment discontinuation due to AEs was reported in 7% of patients.25
Cemiplimab was recently approved by the FDA for patients with metastatic or locally advanced cSCC who are not candidates for curative surgery or radiation.4This approval was based on results from the open-label R2810-ONC-1423 study, which included patients with various advanced solid tumors, and the open-label R2810-ONC-1540 study, which included patients with metastatic or locally advanced cSCC regardless of prior treatment.4Of the 108 patients with advanced disease (metastatic, n = 75; locally advanced, n = 33), the ORR was 47%.4Patients with metastatic or locally advanced disease had ORRs of 47% and 49%, respectively.4Overall, 61% of responses were durable for ≥6 months.4The most common AEs reported in the 534 patients from both trials were rash, fatigue, and diarrhea.4Immune-mediated adverse reactions including pneumonitis, colitis, hepatitis, nephritis, adrenal insufficiency, diabetes mellitus, hypo- and hyperthyroidism, and infusion reactions were also reported.4
Future Therapies Several additional therapeutic options are under investigation in clinical trials, including topical creams, chemotherapy, targeted therapy, and immunotherapy (TABLE).26-29
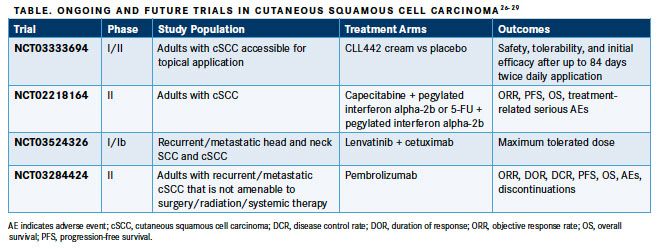
A first-in-human, proof-of-concept, phase I/II study has been designed to assess the safety, tolerability, and initial efficacy of CLL442 cutaneous cream compared with placebo following ≤84 days of twice-daily application on a single cSCC lesion.26
The combination of capecitabine or 5-fluorouracil with pegylated interferon alpha-2b is under investigation in a phase II study of patients with cSCC.27The primary outcome is the ORR per participant after 9 weeks, with secondary outcomes of 1-year progression-free survival, 1-year overall survival, and the occurrence of treatment-related SAEs.27
A phase I study was designed to determine the maximum tolerated dose of lenvatinib when combined with cetuximab in patients with recurrent or metastatic head and neck cSCC.28The safety and efficacy of pembrolizumab is under investigation in a phase II study of adult participants with recurrent or metastatic cSCC who are not eligible for surgery, radiation, and systemic therapies.29
Because of its success in human papillomavirus (HPV) associated head and neck SCC, HPV has been suggested as a promising target for the treatment of cSCC.30MicroRNAs (miRNAs) have been shown to suppress the expression of target genes and have been associated with both the onset and progression of various cancers, implicating their potential as targets in cancer therapies.30 miRNA-1, miRNA-34a, and miRNA-124/214 have been shown to be deregulated in cSCC.31These miRNAs target important molecules of cell proliferation, which are often upregulated in cancer cells; therefore, promoting the overexpression of these miRNAs may be an option for therapeutic development.30
Conclusions
Several recommended treatment strategies are available that are effective for most patients with cSCC. However, there are fewer recommended options for patients with recurrent or metastatic disease. Cemiplimab recently received FDA approval as the first systemic treatment for patients with recurrent or metastatic disease who are not candidates for curative surgery or radiation therapy. The need remains for additional therapies in this high-risk group to improve quality of life and survival and to facilitate the reduction in the large financial burden associated with this common cancer.
References:
- Rogers HW, Weinstock MA, Feldman SR, Coldiron BM. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the U.S. population, 2012.JAMA Dermatol.2015;151(10):1081-1086. doi: 10.1001/jamadermatol.2015.1187.
- National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology. Squamous Cell Skin Cancer. NCCN website. https://www.nccn.org/professionals/ physician_gls/pdf/squamous.pdf. Updated October 23, 2018. Accessed October 25, 2018.
- Schmults CD, Karia PS, Carter JB, et al. Factors predictive of recurrence and death from cutaneous squamous cell carcinoma: a 10-year, single-institution cohort study.JAMA Dermatol. 2013;149(5):541-547. doi: 10.1001/jamadermatol.2013.2139.
- US Food and Drug Administration. FDA approves cemiplimab-rwlc for metastatic or locally advanced cutaneous squamous cell carcinoma. FDA website. www.fda.gov/ Drugs/InformationOnDrugs/ApprovedDrugs/ucm622251.htm. Updated October 2, 2018. Accessed October 25, 2018.
- Alam M, Ratner D. Cutaneous squamous-cell carcinoma.N Engl J Med. 2001;344(13):975-983.
- Lansbury L, Bath-Hextall F, Perkins W, et al. Interventions for non-metastatic squamous cell carcinoma of the skin: systematic review and pooled analysis of observational studies.BMJ. 2013;347:f6153. doi: 10.1136/bmj.f6153.
- Mohs FE. Chemosurgical treatment of cancer of the nose; a microscopically controlled method.Arch Surg.1946;53:327-344.
- Tromovitch TA, Stegeman SJ. Microscopically controlled excision of skin tumors.Arch Dermatol.1974;110(2):231-232.
- Connolly SM, Baker DR, Coldiron BM, et al; Ad Hoc Task Force. AAD/ACMS/ ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery.J Am Acad Dermatol.2012;67(4):531-550. doi: 10.1016/j.jaad.2012.06.009.
- Rowe DE, Carroll RJ, Day CL Jr. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip: implications for treatment modality selection.J Am Acad Dermatol.1992;26(6):976-990.
- Rodriguez-Vigil T, Vázquez-López F, Perez-Oliva N. Recurrence rates of primary basal cell carcinoma in facial risk areas treated with curettage and electrodesiccation.J Am Acad Dermatol.2007;56(1):91-95.
- Mendenhall WM, Amdur RJ, Hinerman RW, et al. Radiotherapy for cutaneous squamous and basal cell carcinomas of the head and neck.Laryngoscope. 2009;119(10):1994-1999. doi: 10.1002/lary.20608.
- Kwan W, Wilson D, Moravan V. Radiotherapy for locally advanced basal cell and squamous cell carcinomas of the skin.Int J Radiat Oncol Biol Phys.2004;60(2):406-411.
- Jennings L, Schmults CD. Management of high-risk cutaneous squamous cell carcinoma.J Clin Aesthet Dermatol.2010;3(4):39-48.
- Tiodorovic-Zivkovic D, Zalaudek I, Longo C, et al. Successful treatment of two invasive squamous cell carcinomas with topical 5% imiquimod cream in elderly patients.Eur J Dermatol.2012;22(4):579-580. doi: 10.1684/ejd.2012.1771.
- Dirschka T, Schmitz L, Bartha Á. Clinical and histological resolution of invasive squamous cell carcinoma by topical imiquimod 3.75%: a case report.Eur J Dermatol.2016;26(4):408-409. doi: 10.1684/ejd.2016.2788.
- Jambusaria-Pahlajani A, Ortman S, Schmults CD, Liang C. Sequential curettage, 5-fluorouracil, and photodynamic therapy for field cancerization of the scalp and face in solid organ transplant recipients.Dermatol Surg.2016;42(1 suppl):S66-S72. doi: 10.1097/DSS.0000000000000589.
- Wang Y, Yang Y, Yang Y, Lu Y. Surgery combined with topical photodynamic therapy for the treatment of squamous cell carcinoma of the lip.Photodiagnosis Photodyn Ther.2016;14:170-172. doi: 10.1016/j.pdpdt.2016.04.008.
- Guthrie TH Jr, Porubsky ES, Luxenberg MN, et al. Cisplatin-based chemotherapy in advanced basal and squamous cell carcinomas of the skin: results in 28 patients including 13 patients receiving multimodality therapy.J Clin Oncol.1990;8(2):342-346.
- Shin DM, Glisson BS, Khuri FR, et al. Phase II and biologic study of interferon alfa, retinoic acid, and cisplatin in advanced squamous skin cancer.J Clin Oncol.2002;20(2):364-370.
- Maubec E, Petrow P, Scheer-Senyarich I, et al. Phase II study of cetuximab as firstline single-drug therapy in patients with unresectable squamous cell carcinoma of the skin.J Clin Oncol.2011;29(25):3419-3426. doi: 10.1200/JCO.2010.34.1735.
- Foote MC, McGrath M, Guminski A, et al. Phase II study of single-agent panitumumab in patients with incurable cutaneous squamous cell carcinoma.Ann Oncol.2014;25(10):2047-2052. doi: 10.1093/annonc/mdu368.
- Krynitz B, Edgren G, Lindelöf B, et al. Risk of skin cancer and other malignancies in kidney, liver, heart and lung transplant recipients 1970 to 2008--a Swedish populationbased study.Int J Cancer.2013;132(6):1429-1438. doi: 10.1002/ijc.27765.
- Falchook GS, Leidner R, Stankevich E, et al. Responses of metastatic basal cell and cutaneous squamous cell carcinomas to anti-PD1 monoclonal antibody REGN2810.J Immunother Cancer.2016;4:70. doi: 10.1186/s40425-016-0176-3.
- Migden MR, Rischin D, Schmults CD, et al. PD-1 blockade with cemiplimab in advanced cutaneous squamous-cell carcinoma.N Engl J Med.2018;379(4):341-351. doi: 10.1056/NEJMoa1805131.
- Safety, tolerability, and efficacy study of CLL442 in patients with cutaneous squamous cell carcinoma in situ (SCCis). clinicaltrials.gov/ct2/show/NCT03333694. Published November 7, 2017. Updated October 24, 2018. Accessed October 29, 2018.
- Capecitabine or 5-FU with pegylated interferon alpha-2b in unresectable/metastatic cutaneous squamous cell carcinoma. clinicaltrials.gov/ct2/show/NCT02218164. Published July 20, 2018.Updated July 20, 2018. Accessed October 29, 2018.
- Testing lenvatinib and cetuximab in patients with advanced head and neck squamous cell carcinoma and cutaneous squamous cell carcinoma. clinicaltrials.gov/ct2/show/ NCT03524326. Published May 14, 2018. Updated September 26, 2018. Accessed October 29, 2018.
- Study of pembrolizumab (MK-3475) in adults with recurrent or metastatic cutaneous squamous cell carcinoma (R/M cSCC) (MK-3475-629/KEYNOTE-629). clinicaltrials.gov/ct2/show/NCT03284424. Published September 15, 2017. Updated September 10, 2018. Accessed October 29, 2018.
- Yanagi T, Kitamura S, Hata H. Novel therapeutic targets in cutaneous squamous cell carcinoma.Front Oncol.2018;8:79. doi: 10.3389/fonc.2018.00079.
- Yu X, Li Z. The role of miRNAs in cutaneous squamous cell carcinoma.J Cell Mol Med.2016;20(1):3-9. doi: 10.1111/jcmm.12649.
Savona Discusses First-Line JAK Inhibition for Patients With Myelofibrosis at Risk of Anemia
April 17th 2024During a Case-Based Roundtable® event, Michael Savona, MD, and participants discussed the case of a patient with myelofibrosis and moderate anemia receiving JAK inhibitor therapy.
Read More
Biomarker Testing Paves the Way for Better Targeted Therapies in NSCLC
April 16th 2024At a live virtual event, Edward S. Kim, MD, MBA, discussed the evolving landscape of biomarker testing before making treatment decisions for patients with early-stage non–small cell lung cancer (NSCLC).
Read More
Creating Solutions for a 'Continual State of Transition' in Cancer Care
April 15th 2024In a Peers & Perspectives in Oncology feature article, we focus on the importance of the transition-of-care process for patients with cancer as they move from the inpatient to outpatient setting, as well as between lines of therapy with comments from Marc J. Braunstein, MD, PhD, and Michael Shusterman, MD.
Read More
