The Rationale for Adjuvant Ovarian Suppression in Patients With Advanced Breast Cancer Following Chemotherapy
This article focuses specifically on the role of ovarian suppression in young premenopausal women. It reviews methods of ovarian suppression, provides evidence from recent pivotal clinical trials, outlines guidelines for its use, and discusses best practices and practical management applications.
1Premenopausal women are at an increased risk of developing ABC and have a worse prognosis than older women.2In addition to having factors that put them at high risk for aggressive breast cancer subtypes, young premenopausal women (typically aged ≤40 years) are at an increased risk for long-term toxicities (ie, ovarian failure) associated with currently available treatments.2,3Therefore, they require complex therapeutic regimens.
This article focuses specifically on the role of ovarian suppression in young premenopausal women. It reviews methods of ovarian suppression, provides evidence from recent pivotal clinical trials, outlines guidelines for its use, and discusses best practices and practical management applications.
THERAPEUTIC OVERVIEW
Breast cancer is the most common cancer diagnosed in women in the United States, with an estimated 268,600 new cases of invasive breast cancer diagnoses in 2019.4Approximately 12% of women will receive a diagnosis of breast cancer at some point in their lives.4Nearly 42,000 women are estimated to die from breast cancer in 2019. Although breast cancer is also the second most common cause of cancer-related death among women, more women are surviving now than in past years.4In fact, more than 3.1 million women are breast cancer survivors in the United States.4
Cancer stage at diagnosis is a strong prognostic factor for survival and helps guide treatment decisions.5,6Breast cancer staging is used to determine the extent of cancer spread, if any. Staging involves assessing the size and grade of the tumor, the extent of lymph node involvement, whether metastasis has occurred, whether the tumor is positive for hormone receptors (HRs)including estrogen receptors (ERs) and progesterone receptors—and HER2 status.6Breast cancer stages range from 0 (carcinoma in situ) to IV.6
Goals of Therapy
ABC includes both LABC and metastatic (or stage IV) breast cancer.1LABC is typically considered inoperable due to local spread into the chest wall and invasion of nearby lymph nodes.7Metastatic breast cancer is cancer that has spread to distant sites or organs (eg, lungs, liver, or bones).6Despite the availability of treatments, once breast cancer has reached either of these stages, it is considered incurable.1Thus, the goal of therapy is to extend survival and improve patient quality of life (QoL).1,8
Hormones, especially estrogen, play a key role in the development, staging, and treatment of patients with ABC.1,6,9Studies have demonstrated an association between estrogen and an increased risk of breast cancer, although the exact mechanism for its role in cancer development remains unclear.9
Compared with postmenopausal women, premenopausal women are at an increased risk for developing ABC.2These women are more likely to have high-risk features, such as a higher cancer stage at diagnosis with higher tumor grades, larger tumor sizes, the presence of local spread to auxiliary lymph nodes, and metastases to distant sites.2Their breast cancer is also more likely to be an aggressive subtype, such as HER2-overexpressing breast cancer (HER2+) or triple-negative breast cancer.2These factors result in poorer clinical outcomes. Moreover, the risk of recurrence is 1.5 times higher for young women (aged <40 years) compared with older women (aged ≥40 years), putting the former at an increased risk of death from their disease.2
In premenopausal women, the primary source of estrogen is the ovaries, with a small amount produced through peripheral aromatization of androgens.10By blocking ovarian function, a process commonly referred to as ovarian suppression or ablation, the ability of estrogen to promote cancer cell growth is hindered.11This treatment strategy is commonly used in patients with hormone-sensitive breast cancer (eg, HR-positive [HR+]).11Approximately 75% of all breast cancers are ER-positive (ER+).8,12
Methods of Ovarian Suppression for Advanced Breast Cancer
In general, when discussing strategies for ABC treatment, all of the following should be taken into account: HR and HER2 status, prior therapies and toxicities, tumor burden and site of metastasis, disease-free interval, biological age and menopause status, performance status, comorbidities, whether the patient requires rapid disease or symptom control, and any socioeconomic and psychological patient factors.1Ovarian suppression is a recommended part of the treatment protocol for young high-risk premenopausal women.1
An overview of the therapeutic strategies for ovarian suppression in women with ABC is provided inTABLE 1.10,11,13

Surgery
Oophorectomy, surgical removal of the ovaries, is one of the oldest strategies used for ovarian suppression.14This procedure was first used in 1896 by George Beatson, MD, who subsequently observed tumor suppression and prolonged survival in a patient with recurrent breast cancer.10Although oophorectomy consistently and rapidly diminishes circulating estrogen levels, it comes with a potential increased risk for morbidity and mortality, as patients require surgery and hospitalization.10It is also an irreversible process.10Not only is ovarian suppression via surgical removal of the ovaries beneficial for improving breast cancer outcomes, but this procedure also concurrently lowers the possibility of ovarian cancer in patients who may be at risk, especially those with theBRAC1andBRAC2genetic mutations and hereditary breast and ovarian cancer syndrome.10,15
Radiotherapy
Ovarian irradiation, or radiotherapy, irreversibly stops ovarian function through localized application of radiation to the ovaries.11,15This method of ovarian suppression came into use as an adjuvant treatment for breast cancer more than half a century ago.10This technique of ovarian suppression is inexpensive and can be performed in an outpatient setting; however, its use is associated with the potential risk of long-term toxicities.10Importantly, ovarian irradiation has been shown to improve progression-free survival (PFS) but provides little to no benefit on overall survival (OS) in premenopausal or perimenopausal women with ABC.16Additionally, radiotherapy does not produce a consistent reduction in estrogen production, and compared with oophorectomy, the decrease in estrogen levels is much slower.10
Pharmacotherapy
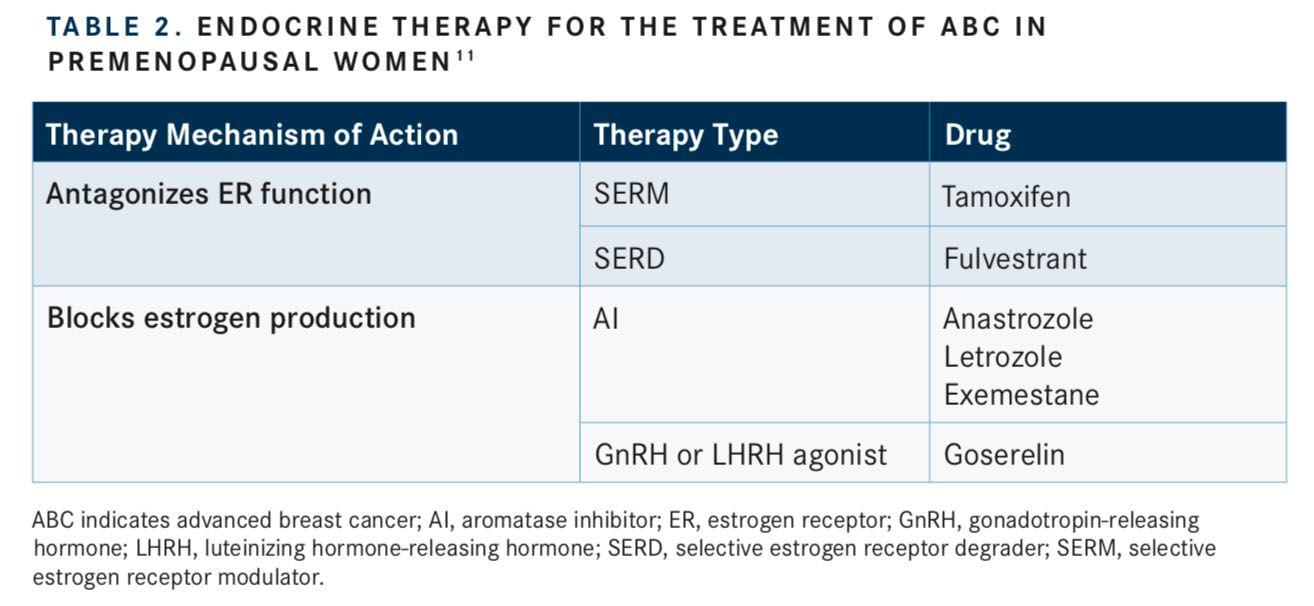
Suppression of ovarian function can also be achieved via administration of pharmacological agents.17There are 2 main types of pharmacotherapy (often referred to as hormone or endocrine therapy) that are used for treating ABC: compounds that directly antagonize ER and compounds that decrease endogenous estrogen production (TABLE 2).11
Surgery
Selective estrogen receptor modulators (SERMs) are designed to compete with estrogen for binding to the ERs, thereby modulating ER activity and subsequent downstream signaling.8These compounds modulate ER signaling differently depending on the tissue type, giving them the name “selective modulators.”8Tamoxifen, a SERM approved by the FDA in 1977, has antagonistic effects on ER activity in the breast, but can promote ER signaling in the uterus, heart, and bones.8,13
Similarly, selective estrogen receptor degraders (SERDs) inhibit ER activity and downstream signaling but through a different mechanism than SERMs. These agents bind and induce ER degradation.8Compared with SERMs, SERDs work purely as estrogen antagonists.11Fulvestrant was originally approved to treat ER+ metastatic breast cancer for patients who had prior progression on endocrine therapy, but it is now also available for the treatment of ER+ ABC.8
Aromatase inhibitors (AIs) block estrogen production.11AIs that are FDA approved for the treatment of breast cancer include anastrozole and letrozole.11These agents are primarily used in postmenopausal women, as they are not generally considered effective against the high levels of aromatase produced by premenopausal women.11However, recent studies have shown a benefit of AI use in premenopausal women with ABC when given with a drug that suppresses ovarian function.18
Other compounds that decrease endogenous estrogen production are agonists of gonadotropin-releasing hormone (GnRH) or of luteinizing hormone-releasing hormone (LHRH).10GnRH agonists act by downregulating GnRH receptors in the pituitary gland, resulting in decreased release of luteinizing hormone and follicle-stimulating hormone, which in turn, leads to inhibition of estrogen production in the ovaries.15As such, GnRH agonists are increasingly used in the treatment of ABC in women with functioning ovaries (ie, premenopausal and perimenopausal women).10LHRH analogues include goserelin, triptorelin, and leuprolide; however, goserelin is the only LHRH analogue approved by the FDA for the treatment of ABC in premenopausal women.10Of note, goserelin is contraindicated in pregnant women unless the agent is being used for palliation of ABC; pregnant women should be notified of the potential harm to the fetus.19The adverse events (AEs) that were observed with goserelin use in at least 20% of patients enrolled in clinical trials for breast cancer included hot flashes and sweating, headaches, acne, emotional lability and depression, decreased libido, vaginitis, breast atrophy, seborrhea, and peripheral edema.19Initiation of goserelin therapy may also induce tumor flaring,19in which transient increases occur in estrogen levels in women with breast cancer (and in testosterone levels in men with prostate cancer). This can result in worsening of symptoms in the first few weeks of therapy.19
The use of GnRH agonists for pharmacologic suppression of ovarian function, compared with surgery or radiotherapy, may be more expensive and require continued intervention (injections every month), but this approach is reversible and has fewer risks and toxicities associated with its use.15
Chemotherapy
Ovarian failure may be an unintended consequence of systemic cytotoxic chemotherapy.10Chemotherapy is recommended as a neoadjuvant treatment to control distant metastases in patients with ER+, HER2 ABC, but it should be reserved for individuals with rapid clinical progression or life-threatening metastases, or those who need quick symptom relief.1The toxic effects (ie, loss of ovarian function, fertility concerns) of chemotherapy depend on the specific agent used, the dose administered, and patient-specific factors including age and performance status.10Chemotherapy is generally useful, but given this devastating long-term AE, attempts have been made to protect ovarian function when chemotherapy is indicated. Of note, ovarian protection has been demonstrated when goserelin is concurrently administered with cytotoxic chemotherapy.20
ADJUVANT OVARIAN SUPPRESSION IN PREMENOPAUSAL WOMEN WITH ADVANCED BREAST CANCER
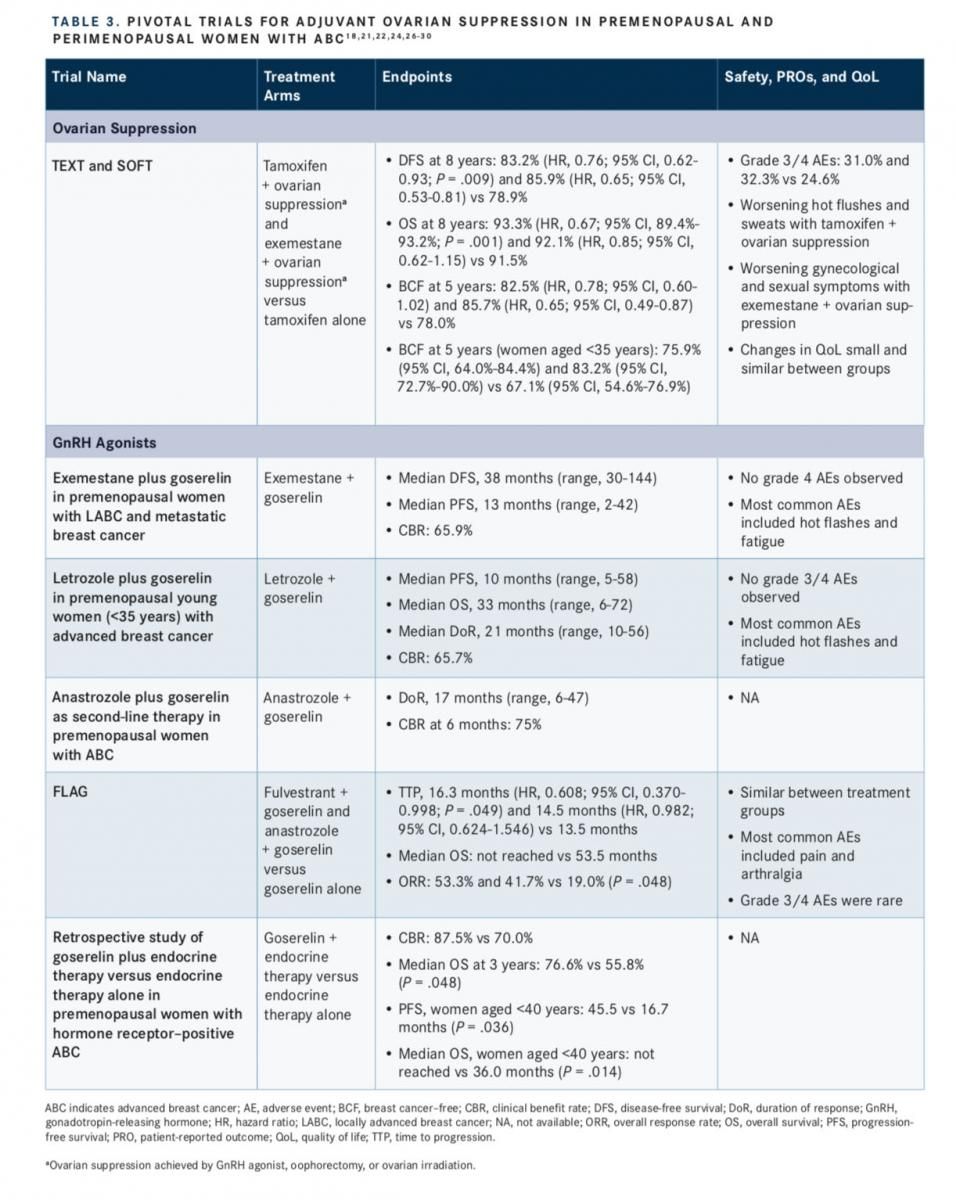
The benefits of ovarian suppression in premenopausal women with breast cancer were evaluated by the International Breast Cancer Study Group in the Tamoxifen and EXemestane Trial (TEXT) and the Suppression of Ovarian Function Trial (SOFT).21From these studies, further analysis was performed to determine the benefit of ovarian suppression specifically in young women (aged <35 years).22Both studies were designed to evaluate the role of AIs and ovarian suppression as adjuvant treatment for premenopausal women with endo- crine-sensitive breast cancer, with TEXT focusing on patients for whom ovarian suppression was indicated from the start of adjuvant therapy, and SOFT focusing on patients who remained premenopausal after (neo)adjuvant chemotherapy or for whom tamoxifen alone following surgery was a practical therapy choice.21
Patients included in the trials were premenopausal with early invasive breast cancer that had been resected and were proven to be HR+ via histology.21In addition, the tumor had to have been confined to the breast and spread to the axillary lymph nodes only, with no other metastases detected.21Between the 2 studies, 5738 patients were enrolled for an intention-to-treat population of 5707, of which 582 patients were aged <35 years at the time of randomization.21,22For TEXT, patients were randomized 1:1 to receive either ex-emestane or tamoxifen, combined with ovarian suppression via the GnRH agonist triptorelin.21SOFT was designed with 3 treatment arms: tamoxifen alone, tamoxifen plus ovarian suppression, and exemestane plus ovarian suppression. Patients were randomized 1:1:1. For patients randomized for ovarian suppression, this could be achieved via GnRH agonist, oophorectomy, or ovarian irradiation.21
Recently published results from the 8-year follow-up of these studies demonstrated that the combination of tamoxifen and ovarian suppression, compared with tamoxifen therapy alone, improved the rates of disease-free survival (DFS; 83.2% vs 78.9%; hazard ratio, 0.76; 95% CI, 0.62-0.93;P= .009) and OS (93.3% vs 91.5%; hazard ratio, 0.67; 95% CI, 89.4%-93.2%;P= .001).18Similarly, DFS was improved with exemestane plus ovarian suppression compared with tamoxifen alone (85.9% vs 78.9%; hazard ratio, 0.65; 95% CI, 0.53-0.81), as was OS (92.1% vs 91.5%; hazard ratio, 0.85; 95% CI, 0.62-1.15).18
These results were consistent with those observed at 5 years, which also demonstrated that patients who underwent chemotherapy, but remained premenopausal, were free from breast cancer longer if they received the treat- ment regimen of ovarian suppression and either tamoxifen (82.5%; hazard ratio, 0.78; 95% CI, 0.60-1.02) or exemestane (85.7%; hazard ratio, 0.65; 95% CI, 0.49-0.87) versus tamoxifen alone (78.0%), with the greatest improvement observed in the exemestane plus ovarian suppression group.23Similar results were observed in the cohort of young women (aged <35 years), in whom a longer median breast cancerfree interval at 5 years was observed with tamoxifen plus ovarian suppression (75.9%; 95% CI, 64.0%-84.4%) or exemestane plus ovarian suppression (83.2%; 95% CI, 72.7%-90.0%) compared with tamoxifen alone (67.1%; 95% CI, 54.6%-76.9%).22
In addition, pooled safety data from the TEXT and SOFT 8-year follow-up trials were published, demonstrating similar AEs among treatment groups. Grade 3/4 AEs occurred in 31.0%, 32.3%, and 24.6% of patients in the tamoxifen plus ovarian suppression group, exemestane plus ovarian sup- pression group, and tamoxifen group, respectively.18
Patient-reported outcomes and QoL measures were evaluated at baseline and multiple times during the 3- to 6-year time frame of the studies.24Patients receiving tamoxifen plus ovarian suppression suffered from increased worsening of hot flashes and sweats from baseline to 6 months compared with patients receiving exemestane plus ovarian suppression, although these symptoms significantly improved from 6 months to 5 years (P <.0001).24However, those who were administered exemestane plus ovarian suppression experienced greater negative gynecological and sexual symptoms (eg, more vaginal dryness, loss of sexual interest, decreased arousal) that persisted over 5 years compared with tamoxifen plus ovarian suppression.24This patient group also experienced more bone or joint pain, which was more pronounced from baseline to 6 months among the treatment groups.24
Overall, minor changes in global QoL measurements from baseline to the end of the study were observed for each treatment group; these changes were similar among groups.24
In addition to TEXT and SOFT, a recently published meta-analysis of 29 randomized controlled trials demonstrated the effects of ovarian suppression in premenopausal women with breast cancer.25Improved DFS (hazard ratio, 0.84; 95% CI, 0.73-0.97; P = .02) and OS (hazard ratio, 0.78; 95% CI, 0.66-0.94; P = .008) were observed in women aged ≤40 years who underwent ovarian suppression.25As this analysis was specific to young women, no significant differences in either DFS (hazard ratio, 0.91; 95% CI, 0.80-1.03; P = .15) or OS (hazard ratio, 0.88; 95% CI, 0.75-1.02; P = .10) were found in women aged >40 years.25
Evaluation of Ovarian Suppression With a GnRH Agonist Combined With Endocrine Therapy in Premenopausal Women With Advanced Breast Cancer
The efficacy and tolerability of exemestane plus ovarian suppression with a GnRH agonist, goserelin, was evaluated in a phase II trial of premenopausal women with either LABC or metastatic breast cancer.26This treatment regimen was observed to be effective, with median DFS and PFS of 38 months (range, 3-144 months) and 13 months (range, 2-42 months), respectively; 65.9% of patients received clinical benefit from the treatment.26 Similar to other studies of ovarian suppression and AI therapy, the most common AEs included hot flashes and fatigue. No grade 4 AEs were reported.26
The combination of goserelin plus letrozole was also evaluated in premenopausal young women (aged <35 years) with ABC.27Results demonstrated a clinical benefit rate of 65.7%, a median duration of response of 21 months (range,10-56 months), a median PFS of approximately 10 months (range, 5-58 months), and a median OS of 33 months (range, 6-72 months), all while being well tolerated. No grade 3/4 toxicities were observed, and the most common AEs were similar to those seen in other studies, including hot flashes and fatigue.27
Improved clinical responses were also observed in a study of goserelin plus anastrozole as second-line endocrine therapy in premenopausal women with ABC.28Investigators observed a clinical benefit rate of 75% at 6 months and median duration of response of 17 months (range, 6-47 months).28
Evidence is lacking regarding which endocrine therapy in combination with ovarian suppression is best for premenopausal women who have been pretreated with or are resistant to tamoxifen. A phase II trial in premenopausal women with HR+, HER2 metastatic breast cancer, who had undergone previous tamoxifen therapy, were enrolled for researchers to evaluate the safety and efficacy of fulvestrant or anastrozole, combined with goserelin, compared with goserelin alone.29The median time to progression for patients receiving fulvestrant plus goserelin was 16.3 months (hazard ratio, 0.608; 95% CI, 0.370-0.998) and for those receiving anastrozole plus goserelin it was 14.5 months (hazard ratio, 0.982; 95% CI, 0.624-1.546), compared with goserelin alone (13.5 months; 95% CI, 10.3-16.8), but only the increase observed in the fulvestrant plus goserelin group was statistically significant (P= .049).29The median OS was 53.5 months for goserelin alone but was not reached in the other treatment groups during the follow-up period of the study.29The overall response rate was significantly improved in the fulvestrant plus goserelin group and the anastrozole plus goserelin group compared with goserelin alone (53.3% and 41.7% vs 19.0%; P = .048).29Safety was similar among treatment groups. The most commonly reported AEs across treatment groups included pain and arthralgia, and the observation of grade 3/4 AEs was rare.29
A retrospective study in premenopausal women with HR+ ABC evaluated the role of adding goserelin to other endocrine therapies. The treatment group included 40 patients who, in addition to being premenopausal with HR+ ABC, had received goserelin plus endocrine therapy. The control group included 40 patients who received only endocrine therapy who were matched for age, receptor status, and tumor stage.30Patients receiving goserelin plus endocrine therapy had greater clinical benefit compared with patients receiving just endocrine therapy (87.5% vs 70.0%), which translated into extended median OS out to 3 years (76.6% vs 55.8%;P= .048).30In patients aged <40 years, goserelin plus endocrine therapy significantly increased PFS (45.5 vs 16.7 months;P= .036) and OS (median not reached vs 36 months;P= .014) compared with endocrine therapy alone.30TABLE 3lists pivotal trials for adjuvant ovarian suppression in premenopausal and perimenopausal women with
ABC.18,21,22,24,26-30
ADJUVANT OVARIAN SUPPRESION IN BREAST CANCER: BEST PRACTICES AND PRACTICAL IMPLICATIONS
Clinical Guidelines and Identifying Candidates for Adjuvant Ovarian Suppression
Based on the results of the TEXT and SOFT studies, the National Comprehensive Cancer Network (NCCN) Breast Cancer Guidelines were updated in 2015 to include the adjuvant endocrine therapy option of ovarian suppression/ablation plus an AI for 5 years. This therapy regimen was recommended for women who are premenopausal, have HR+ breast cancer, and who are at a higher risk of recurrence based on factors such as age, tumor grade, and lymph node involvement.31
In 2017, the NCCN guidelines were updated to include recommendations for the treatment of women with breast cancer who are premenopausal at diagnosis. The NCCN recommended 5 years of treatment with either tamoxifen with or without ovarian suppression, or ovarian suppression plus an AI. Each of these treatment regimens have a category 1 recommendation.32
The American Society of Clinical Oncology (ASCO) convened in 2015 to discuss the role of ovarian suppression in the treatment of premenopausal women with ER+ breast cancer, and more specifically, to determine if ovarian suppression should be provided concurrently with tamoxifen or an AI, and if so, to identify the patient population that would most benefit.33The ASCO panel recommended that patients with high-risk stage II or III breast cancer should receive ovarian suppression in addition to adjuvant endocrine therapy rather than adjuvant chemotherapy.33This treatment regimen may also be considered in patients with stage I and II breast cancers who have a high risk of recurrence.33
Similarly, the European School of Oncology/European Society of Medical Oncology International Consensus Guidelines for Advanced Breast Cancer recommend ovarian suppression/ablation combined with additional endocrine therapy for premenopausal women with ER+, HER2- ABC.1
The International Consensus Guidelines for Breast Cancer in Young Women, guidelines endorsed by the European Society of Breast Specialists, focus specifically on the care and management of breast cancer in young women (defined as aged <40 years), given the complexity of treatment that may be required due to fertility, genetics, and psychosocial concerns.3These guidelines recommend that patients with HR+ disease undergo adjuvant endocrine therapy with tamoxifen for at least 5 years. The panel states that the use of a GnRH agonist combined with tamoxifen and/or chemotherapy is controversial, but it should be considered in the absence of chemotherapy or tamoxifen for this patient population.3
Most recently, the 2019 NCCN guidelines for breast cancer noted that available evidence suggests that among premenopausal women with HR+ early breast cancer, adjuvant exemestane plus ovarian suppression significantly reduces recurrence compared with tamoxifen plus ovarian suppression.34
Increasing Knowledge About the Role of Adjuvant Ovarian Suppression in the Management of Breast Cancer
Although consensus on the best first-line therapy option for premenopausal women with ABC has not been reached, the role of ovarian suppression in these patients is becoming clearer. Based on society guidelines, ovarian suppression can be considered a practical, useful, therapeutic option and is the preferred choice (plus endocrine therapy) for premenopausal women with high-risk disease.1To maintain better disease management, however, providers need access to up-to-date guidelines and therapeutic approaches.17Increasing knowledge is likely to improve disease control, reduce AEs, and ultimately enhance the patient’s QoL.17
In addition, increasing patient education about ovarian suppression and the different ovarian suppression methods can help to manage their expectations regarding the benefits and risks associated with treatment, including potential AEs. This improved understanding, along with giving patients a role in choosing their treatment modality, can result in enhanced patient adherence to complex treatments,35as it has been shown that early discontinuation of or noncompliance with adjuvant therapy is associated with a significant increase in mortality in patients with breast cancer.36Survival at 10 years was 80.7% and 73.6%, respectively (P<.001), in women who continued therapy or discontinued their therapy early.36Similarly, the estimated survival at 10 years was 81.7% in women who adhered to their endocrine therapy treatment and 77.8% in those who did not (P <.001).36The rate of nonadherence for pharmacological ovarian suppression with goserelin or triptorelin may be as high as 21.9% by 4 years.23
A recent study surveyed premenopausal women with breast cancer and showed that, although all patients had received goserelin based on study inclusion, nonpharmaco-logical ovarian suppression methods (eg, oophorectomy or ovarian irradiation) were rarely even discussed during the course of cancer treatment.37Interestingly, of the women who received goserelin for ovarian suppression, 53.8% indicated that they would have preferred a nonpharmacological approach had the option been provided; the reasons they gave included time, hospital visits, injections, and concerns about disease recurrence.37
For patients, successful control of AEs is central to maintaining a good QoL, as AEs that are intolerable are a key factor in reduced patient adherence or therapy discontinuation.35Therefore, providers should discuss with patients the potential AEs of the chosen treatment prior to its initiation. A conversation regarding strategies for managing these AEs, which can include pharmacological approaches and nonpharmacological coping tactics, is also suggested.35For example, symptoms of hot flashes, a common AE of estrogen deficiency, can be lessened with modifications such as dressing in layers and avoiding symptom-provoking activities (eg, eating spicy food).38If patients know what to expect and have the means available for managing AEs, they are more likely to continue treatment.35
CONCLUSIONS
Although ABC is considered an incurable disease, many current available treatment options can help alleviate symptoms and increase the length of survival. Young premenopausal women are at a higher risk for developing ABC and require treatment regimens that take into account individual fertility concerns and the high risk of disease recurrence. Methods of ovarian suppression, compared with endocrine therapy alone, have been shown to significantly improve clinical outcomes for this patient population, including extending DFS, OS, and breast cancerfree intervals. As such, ovarian suppression combined with endocrine therapy has found an important place among many well-known and well-regarded clinical guidelines, specifically in the adjuvant setting for premenopausal women. In this setting, ovarian suppression can play a foundational role in reducing recurrence.
Because the AEs associated with GnRH agonists can be a burden for patients, it is essential that providers take the time with patients to discuss expectations and the options available to mitigate these AEs. Increased knowledge and education regarding the benefits and drawbacks of ovarian suppression are essential not only to improving adherence and disease control, but also to enhancing QoL.
References:
- Cardoso F, Costa A, Senkus E, et al. 3rd ESO-ESMO International Consensus Guidelines for Advanced Breast Cancer (ABC 3). Ann Oncol. 2017;28(12):3111. doi: 10.1093/annonc/mdx036.
- Bardia A, Hurvitz S. Targeted therapy for premenopausal women 12 Ovarian Suppression in Patients With Advanced Breast Cancer Evolving Paradigms with HR+, HER2 advanced breast cancer: focus on special consid- erations and latest advances. Clin Cancer Res. 2018;24(21):5206- 5218. doi: 10.1158/1078-0432.CCR-18-0162.
- Partridge AH, Pagani O, Abulkhair O, et al. First International Con- sensus Guidelines for Breast Cancer in Young Women (BCY1). Breast. 2014;23(3):209-220. doi: 10.1016/j.breast.2014.03.011.
- How common is breast cancer? American Cancer Society website. cancer.org/cancer/breast-cancer/about/how-common-is-breast- cancer.html. Updated January 8, 2019. Accessed January 20, 2019. 5. Cancer stat facts: female breast cancer. National Cancer Institute website. seer.cancer.gov/statfacts/html/breast.html. Accessed October 19, 2018.
- Cancer stat facts: female breast cancer. National Cancer Institute website. seer.cancer.gov/statfacts/html/breast.html. Accessed October 19, 2018.
- Breast cancer stages. American Cancer Society website. cancer. org/cancer/breast-cancer/understanding-a-breast-cancer-diagno- sis/stages-of-breast-cancer.html. Updated December 20, 2017. Accessed October 3, 2018.
- Simos D, Clemons M, Ginsburg OM, Jacobs C. Definition and con- sequences of locally advanced breast cancer. Curr Opin Support Pal- liat Care. 2014;8(1):33-38. doi: 10.1097/SPC.0000000000000020. 8. Patel HK, Bihani T. Selective estrogen receptor modulators (SERMs) and selective estrogen receptor degraders (SERDs) in can- cer treatment. Pharmacol Ther. 2018;186:1-24. doi: 10.1016/j.phar- mthera.2017.12.012.
- Patel HK, Bihani T. Selective estrogen receptor modulators (SERMs) and selective estrogen receptor degraders (SERDs) in can- cer treatment. Pharmacol Ther. 2018;186:1-24. doi: 10.1016/j.phar- mthera.2017.12.012.
- Yue W, Yager JD, Wang JP, et al. Estrogen receptor-dependent and independent mechanisms of breast cancer carcinogenesis. Steroids. 2013;78(2):161-170. doi: 10.1016/j.steroids.2012.11.001. 10. Prowell TM, Davidson NE. What is the role of ovarian ablation in the management of primary and metastatic breast cancer today? Oncologist. 2004;9(5):507-517. doi: 10.1634/theoncologist. 9-5-507.
- Prowell TM, Davidson NE. What is the role of ovarian ablation in the management of primary and metastatic breast cancer today? Oncologist. 2004;9(5):507-517. doi: 10.1634/theoncologist. 9-5-507.
- National Cancer Institute. Hormone therapy for breast cancer. NCI website. cancer.gov/types/breast/breast-hormone-therapy-fact- sheet. Updated February 14, 2017. Accessed October 23, 2018. 12. Perou CM, Sorlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature. 2000;406(6797):747-752. doi: 10.1038/35021093.
- Perou CM, Sorlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature. 2000;406(6797):747-752. doi: 10.1038/35021093.
- McDonald Wade S 3rd, Hackney MH, Khatcheressian J, Lyckholm LJ. Ovarian suppression in the management of premenopausal breast cancer: methods and efficacy in adjuvant and metastatic settings. Oncology. 2008;75(3-4):192-202. doi: 10.1159/000163059.
- Ponzone R, Biglia N, Jacomuzzi ME, et al. Antihormones in the prevention and treatment of breast cancer. Ann N Y Acad Sci. 2006;1089:143-158. doi: 10.1196/annals.1386.037. 15. American Cancer Society. Ovarian cancer risk factors. ACS web- site. cancer.org/cancer/ovarian-cancer/causes-risks-prevention/risk- factors.html. Updated April 11, 2018. Accessed October 28, 2018.
- American Cancer Society. Ovarian cancer risk factors. ACS web- site. cancer.org/cancer/ovarian-cancer/causes-risks-prevention/risk- factors.html. Updated April 11, 2018. Accessed October 28, 2018. 16. Asiri MA, Tunio MA, Abdulmoniem R. Is radiation-induced ovar- ian ablation in breast cancer an obsolete procedure? results of a meta-analysis. Breast Cancer (Dove Med Press). 2016;8:109-116. doi: 10.2147/BCTT.S94617.
- Asiri MA, Tunio MA, Abdulmoniem R. Is radiation-induced ovar- ian ablation in breast cancer an obsolete procedure? results of a meta-analysis. Breast Cancer (Dove Med Press). 2016;8:109-116. doi: 10.2147/BCTT.S94617.
- Tancredi R, Furlanetto J, Loibl S. Endocrine therapy in premeno- pausal hormone receptor positive/human epidermal growth receptor 2 negative metastatic breast cancer: between guidelines and literature. Oncologist. 2018;23(8):974-981. doi: 10.1634/theoncolo- gist.2018-0077.
- Francis PA, Pagani O, Fleming GF, et al; SOFT and TEXT Investi- gators and the International Breast Cancer Study Group. Tailoring adjuvant endocrine therapy for premenopausal breast cancer. N Engl J Med. 2018;379(2):122-137. doi: 10.1056/NEJMoa1803164.
- Zoladex [prescribing information]. Lake Forest, IL: AstraZeneca; 2017. documents.tersera.com/zoladex-us/3.6mg_MagnumPI.pdf. Accessed October 29, 2018.
- Moore HC, Unger JM, Phillips KA, et al; POEMS/S0230 Investiga- tors. Goserelin for ovarian protection during breast-cancer adjuvant chemotherapy. N Engl J Med. 2015;372(10):923-932. doi: 10.1056/ NEJMoa1413204.
- Regan MM, Pagani O, Fleming GF, et al; GroupSOFT and TEXT Investigators. Adjuvant treatment of premenopausal women with endocrine-responsive early breast cancer: design of the TEXT and SOFT trials. Breast. 2013;22(6):1094-1100. doi: 10.1016/j. breast.2013.08.009.
- Saha P, Regan MM, Pagani O, et al; SOFT; TEXT Investigators; International Breast Cancer Study Group. Treatment efficacy, adher- ence, and quality of life among women younger than 35 years in the International Breast Cancer Study Group TEXT and SOFT adjuvant endocrine therapy trials. J Clin Oncol. 2017;35(27):3113-3122. doi: 10.1200/JCO.2016.72.0946.
- Francis PA, Regan MM, Fleming GF, et al; SOFT Investigators; In- ternational Breast Cancer Study Group. Adjuvant ovarian suppression in premenopausal breast cancer. N Engl J Med. 2015;372(5):436-446. doi: 10.1056/NEJMoa1412379.
- Bernhard J, Luo W, Ribi K, et al. Patient-reported outcomes with adjuvant exemestane versus tamoxifen in premenopausal women with early breast cancer undergoing ovarian suppression (TEXT and SOFT): a combined analysis of two phase 3 randomised trials. Lancet Oncol. 2015;16(7):848-858. doi: 10.1016/S1470- 2045(15)00049-2.
- Zhang P, Li CZ, Jiao GM, et al. Effects of ovarian ablation or sup- pression in premenopausal breast cancer: a meta-analysis of random- ized controlled trials. Eur J Surg Oncol. 2017;43(7):1161-1172. doi: 10.1016/j.ejso.2016.11.011.
- Wang J, Xu B, Yuan P, et al. Phase II trial of goserelin and ex- emestane combination therapy in premenopausal women with lo- cally advanced or metastatic breast cancer. Medicine (Baltimore). 2015;94(26):e1006. doi: 10.1097/MD.0000000000001006.
- Liu X, Qu H, Cao W, et al. Efficacy of combined therapy of gosere- lin and letrozole on very young women with advanced breast cancer as first-line endocrine therapy. Endocr J. 2013;60(6):819-828. doi: 10.1507/endocrj.EJ12-0434.
- Forward DP, Cheung KL, Jackson L, Robertson JF. Clinical and en- docrine data for goserelin plus anastrozole as second-line endocrine therapy for premenopausal advanced breast cancer. Br J Cancer. 2004;90(3):590-594. doi: 10.1038/sj.bjc.6601557.
- Kim JY, Im SA, Jung KH, et al; Breast Cancer Committee of Korean Cancer Study Group (KCSG). Fulvestrant plus goserelin versus anastrozole plus goserelin versus goserelin alone for hormone receptor-positive, HER2-negative tamoxifen-pretreated premenopausal women with recurrent or metastatic breast cancer (KCSG BR10-04): a multicentre, open-label, three-arm, randomised phase II trial (FLAG study). Eur J Cancer. 2018;103:127-136. doi: 10.1016/j.ejca.2018.08.004.
- Wu S, Li Q, Zhu Y, et al. Role of goserelin in combination with endocrine therapy for the treatment of advanced breast cancer in premenopausal women positive for hormone receptor: a retro- spective matched case-control study. Cancer Biother Radiopharm. 2013;28(10):697-702. doi: 10.1089/cbr.2012.1436.
- Gradishar WJ, Anderson BO, Balassanian R, et al. NCCN Guidelines Insights: Breast Cancer, version 1.2016. J Natl Compr Canc Netw. 2015;13(12):1475-1485. doi: 10.6004/ jnccn.2015.0176.
- Gradishar WJ, Anderson BO, Balassanian R, et al. NCCN Guide- lines Insights: Breast Cancer, version 1.2017. J Natl Compr Canc Netw. 2017;15(4):433-451. doi: 10.6004/jnccn.2017.0044.
- Burstein HJ, Lacchetti C, Anderson H, et al. Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: American Society of Clinical Oncology clinical practice guideline update on ovarian suppression. J Clin Onc. 2016;34(14):1689-1701. doi: 10.1200/JCO.2015.65.9573.
- NCCN Clinical Practice Guidelines in Oncology. Breast Cancer, version 1.2019. National Comprehensive Cancer Network website. nccn.org/professionals/physician_gls/pdf/breast.pdf. Published March 14, 2019. Accessed March 27, 2019.
- Monnier A. Clinical management of adverse events in adjuvant therapy for hormone-responsive early breast cancer. Ann Oncol. 2007;18(8 suppl):viii36-viii44. doi: 10.1093/annonc/mdm264. 36. Hershman DL, Shao T, Kushi LH, et al. Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat. 2011;126(2):529-537. doi: 10.1007/s10549- 010-1132-4.
- Hershman DL, Shao T, Kushi LH, et al. Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat. 2011;126(2):529-537. doi: 10.1007/s10549- 010-1132-4.
- Hsieh AH, Kichenadasse G, Vatandoust S, et al. Goserelin toxici- ties and preferences for ovarian suppression method in pre-meno- pausal women with breast cancer. Intern Med J. 2016;46(10):1153- 1159. doi: 10.1111/imj.13169.
- Leon-Ferre RA, Majithia N, Loprinzi CL. Management of hot flashes in women with breast cancer receiving ovarian function suppression. Cancer Treat Rev. 2017;52:82-90. doi: 10.1016/j. ctrv.2016.11.012.
Breast Cancer Leans into the Decade of Antibody-Drug Conjugates, Experts Discuss
September 25th 2020In season 1, episode 3 of Targeted Talks, the importance of precision medicine in breast cancer, and how that vitally differs in community oncology compared with academic settings, is the topic of discussion.
Listen
Powell Reviews Updated IO/TKI Data and AE Management in Endometrial Cancer
April 18th 2024During a Case-Based Roundtable® event, Matthew A. Powell, MD, discussed the case of a patient with advanced endometrial cancer treated with lenvatinib plus pembrolizumab who experienced grade 2 treatment-related hypertension.
Read More
