Adoptive Immunotherapy Makes Inroads in Head and Neck Cancers, But Challenges Remain
Adoptive immunotherapy has shown promising outcomes in early-stage trials for many cancers, but successful approaches in head and neck cancers will require addressing several challenges that are inherent in many solid tumors, such as creating the right homing mechanisms, achieving adequate T-cell infiltration into the tumor, and finding an antigen that will not produce off-tumor effects.

Adoptive immunotherapy has shown promising outcomes in early-stage trials for many cancers, but successful approaches in head and neck cancers will require addressing several challenges that are inherent in many solid tumors, such as creating the right homing mechanisms, achieving adequate T-cell infiltration into the tumor, and finding an antigen that will not produce off-tumor effects.
Early data on adoptive immunotherapy in head and neck cancers have shown just modest responses. However, they provide proof-of-concept that such immunotherapy can be effective, and ongoing research on optimizing adoptive immunotherapy in hematologic malignancies will help guide research for head and neck cancer, said Ezra Cohen, MD, associate director, Moores Cancer Center, and professor of medicine, University of California, San Diego, in an interview withTargeted Therapies in Oncology™.
“The success has not been as dramatic as we’ve seen in leukemia with CD19 chimeric antigen receptors [CARs] or multiple myeloma with B-cell maturation antigen CARs, but the fact that it’s been shown to work in some patients demonstrates that at least there’s a principle that appears to be true,” said Cohen. “The question becomes, how do we turn that from an unusual response to an expected response?”
TYPES OF ADOPTIVE T-CELL THERAPY
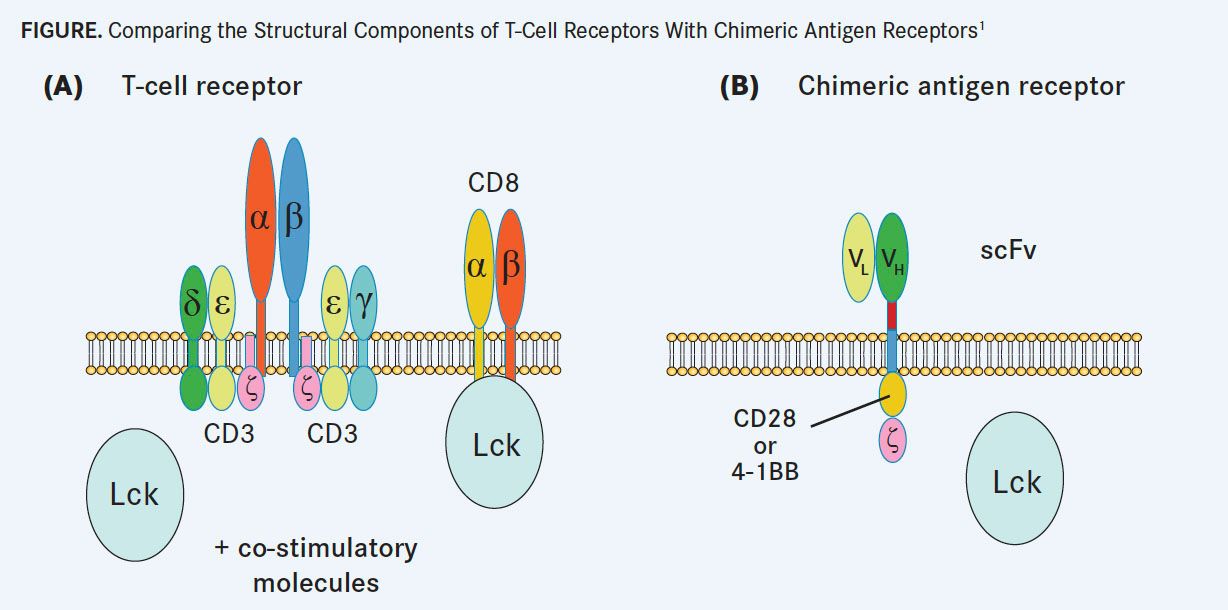
Adoptive T-cell therapy for cancer involves harvesting T cells from the patient’s blood or tumor, stimulating them to grow and expand in an in vitro culture system, and reinfusing the expanded cells back into the patient with the goal of mediating tumor destruction. Development of T-cell receptors (TCRs) involves introduction of a tumor antigenspecific TCR to genetically modify T cells. These modified T cells use a complex assembly of proximal signaling molecules to achieve highly sensitive recognition of abnormal intracellular antigens, which enables them to initiate a potent, specific immune response against viruses and transformed cancer cells. By contrast, CARs are hybrid receptors formed by linking an extracellular, tumor-specific antibody fragment, a stalk-like region, a transmembrane region, and intracellular signaling domains derived from proximal T-cell signaling machinery (FIGURE).1
“The biggest difference is that CARs depend on a surface-expressed receptor or protein that can be detected by the CAR,” said Cohen. “It has to be something expressed on the surface of the cancer cell. The TCRs depend on the cancer cell essentially processing a protein and presenting it as a peptide in the context of that cell’s major histocompatibility complex.”
TCR APPROACHES FOR HEAD AND NECK CANCER
TCR approaches are appealing for head and neck cancers, particularly those that are virus-related, because they contain antigens that are clearly “nonhuman,” according to Cohen. A phase I study2 administered ex vivo expanded autologous T cells directed toward the Epstein-Barr virus (EBV) to 22 patients with metastatic EBV-positive nasopharyngeal cancer. Expansion of EBV-specific T cells was successful in 16 patients, who demonstrated a median overall survival (OS) of 523 days. By contrast, the patients who did not have successful T-cell expansion had a median OS of 220 days.
Early research also suggests that adoptive cell therapy could work synergistically with chemotherapy by enhancing the antitumor effect and improve immune function, which is often compromised from chemotherapies frequently used for head and neck cancer. A retrospective study3showed that patients with head and neck squamous cell cancer (HNSCC) who received adoptive cell therapy after radical resection and induction chemotherapy had significantly longer progression-free survival (PFS) and OS than patients who received induction chemotherapy alone (PFS, 56 vs 40 months; OS, 58 vs 45 months). Additionally, the adoptive cell therapy, which involved expansion and profiling of cytokine-induced killer cells (CIKs) from peripheral blood mononuclear cells, increased the cytotoxicity of the CIKs and restored the systemic immunosuppression that occurred after the chemotherapy. The authors also noted that restoring the systemic immunosuppression that occurs with many HNSCC chemotherapy regimens can be particularly valuable for reducing infections and improving patients’ quality of life during treatment.
Recent preclinical research also suggests that TCRs increase tumor sensitivity to chemotherapy and exhibit cytotoxicity to chemotherapy-resistant tumor cells. In an in vitro study,4adding cytomegalovirus (CMV) pp65 antigen specific cytotoxic T-lymphocytes (CTLs) 5-fluorouracil and cisplatin (CDDP) chemotherapy demonstrated cytotoxicity against oral squamous cell cancer (OSCC) cell lines overexpressing CMVpp65 antigen, and the cytotoxicity of CMVpp65-CTL was the same with CDDP-resistant and -nonresistant OSCC cells. Further, the chemotherapeutic agents sensitized the OSCC cells to CMVpp65-CTL and did not inhibit cytotoxicity or the interferon-γ release response. From these data, the authors concluded that combining TCRs with chemotherapy could be particularly promising for patients who have CDDP-resistant tumors or who cannot tolerate high doses of CDDP.
Viruses as Targets for TCR Therapy
The rapidly increasing incidence of human papillomavirus (HPV)-positive HNSCC over the past decade has also encouraged recent research efforts toward developing effective HPV-targeted TCRs. Preliminary data from a recently completed phase I/II trial of patients with various metastatic, HPV16-positive, HLA-A*02:01-positive cancers (including oropharyngeal, cervical, anal, and vaginal) showed that administration of a nonmyeloablative conditioning regimen of cyclophosphamide and fludarabine, a single infusion of T cells genetically engineered to express a TCR targeting an HLA-A*02:01-restricted epitope of E6 (E6 TCR T cells), and systemic high-dose aldesleukin yielded partial responses in 2 of 12 patients (both with anal cancer).5 The responders had high levels of E6 TCR T-cell memory 1 month after treatment, and 7% of E6 TCR T cells were detected 10 months after treatment in a resected tumor of the patient who had a 6-month response. By contrast, tumor tissue from a nonresponder had no detectable levels of E6 TCR T cells 3 months after treatment. The researchers plan to recruit up to 18 patients for the dose-escalation portion of the study and enroll at least 41 patients for the phase II portion, provided that a clinical response is observed in at least 2 patients of the first 21 patients in the phase II portion. The primary outcome measures for the study are maximum tolerated dose, objective tumor response rate, and duration of response.
Although Cohen stated that development of adoptive immunotherapy approaches will be more challenging for virus-negative cancers, he suggested that identifying other common neoantigens, such as PIK3CA and HRAS mutations with HNSCC, and the peptides they produce, may be instrumental for developing TCRs that target these mutations. He also noted that finding targets may be somewhat easier for thyroid cancers because they tend to be more homogeneous in their genetic alterations than HNSCCs. “More than 50% of papillary thyroid cancers have BRAF mutations, so that could be a good antigen for a T-cell receptor,” said Cohen.
Using Biotechnology to Improve Identification of Target Peptides for TCR
In partnership with investigators at MD Anderson Cancer Center, researchers at Immatics US, Inc, developed IMA201 using their proprietary ACTengine approach, which engineers a patient’s T cells to express an exogenous TCR targeted to a tumor site identified by their XPRESIDENT target discovery platform. The XPRESIDENT platform improves the identification of target peptides for TCR development by analyzing peptides obtained from samples of tumor tissue and corresponding normal tissue through a combination of quantitative transcriptomics and quantitative HLA peptidomics to identify HLA peptides that are present on cells in tumor tissue and absent on cells in normal tissue.6
An open-label, first-in-human, dose-escalating phase I trial (NCT03247309) was initiated in September 2017 to investigate the safety, tolerability, and clinical activity of IMA201 in patients with HNSCC or nonsmall cell lung cancer (NSCLC) with an HLA-A*02:01 phenotype who have no further options for treatment or who cannot tolerate current treatments (TABLE).7The primary objective of the study is to assess safety and to identify the maximum tolerated dose. Secondary endpoints include overall response rate (assessed using RECIST criteria and Immune- Related Response Criteria), PFS, and OS. The authors also hope to assess several translational objectives, including the persistence and functionality of IMA201 in vivo, correlative biomarkers for clinical success, and the target expression levels in the tissue.
CAR T-CELL THERAPY
CAR T-cell therapies have shown remarkable responses in hematologic malignancies, including CAR T cells targeting CD19 in relapsed acute lymphoblastic leukemia (ALL),8multiple myeloma,9and diffuse large B-cell lymphoma or follicular lymphoma.10However, development of CAR T-cell therapies for HNSCC and other solid tumors has been challenging, according to Cohen.
“A T cell has to traffic to the cancer and overcome an immunosuppressive microenvironment to kill that tumor cell, even if it’s an engineered T cell through a chimeric antigen receptor or TCR,” he said. “For solid tumors, those T cells may not necessarily have the right homing mechanisms, and part of the education of a T cell is that it learns to home to specific tissue.” He also pointed out that the density of solid tumors and the fibrous tissue that makes up many head and neck cancers can prevent T cells from infiltrating the tumor.
Intratumoral Approach With a Multitargeted CAR T-Cell Therapy
To address the challenges of CAR T-cell homing and off-tumor effects, a research group at King’s College in London developed a pan-ErbBtargeted CAR T-cell therapy that is injected intratumorally, rather than systemically, for HNSCC. The T4 immunotherapy the team used in a dose-escalation phase I study11involved T cells that were retrovirally transduced to co-express T1E28ζ (a CAR coupling a promiscuous ErbB ligand derived from EGF and TGFα to a fused CD28+CD3ζ endodomain) and 4αβ (a chimeric cytokine receptor containing the interleukin (IL)-4Rα ectodomain coupled to the IL-2Rβ endodomain). The T4 immunotherapy was successfully generated from a 130-mL blood draw, even though 90% of the patients were lymphopenic, and patients were treated in cohorts of 1 x 107, 3 x 107, and 10 × 107 T4-positive T cells. The overall disease control rate was 44%, and all 3 patients who received the highest dose had stable disease. Treatment-related adverse events were grade 2 or lower (the most common being tumor swelling, pain, and fatigue), and no dose-limiting toxicities were observed.
The trial is still ongoing, with the study authors planning to investigate the safety and efficacy of higher doses as well as combinations with other immunooncology agents. However, lead author Sophie Papa, PhD, MBBS, MRCP, said that this mechanism of T-cell therapy could also be a promising approach to a regionally directed therapy, such as treatment in the thoracic cavity for mesothelioma or in the abdominal peritoneal cavity for ovarian cancer.
“This is, essentially, nontoxic personalized therapy for patients with a very lethal form of cancer that appears to be controlling disease in the majority of patients,” said Papa, medical oncologist, Clinical Academic Group, Department of Research Oncology, King’s College London, in an earlier interview with Targeted Therapies in Oncology™.12“The future for this is to continue to dose-escalate because we are starting to see signs of more activity in patients in the higher dose level.”
Minimizing On-Target, Off-Tumor Effects
Another challenge for CAR T-cell therapy in HNSCC is finding an appropriate antigen to target because many of the antigens on the surface of the tumors are also present on normal cells throughout the body. This can lead to on-target, off-tumor effects that are a common concern when developing CAR T-cell therapies, as was demonstrated when systemically delivered carcinoembryonic antigentargeted CAR T-cell therapy for colorectal cancer led to life-threatening colitis in several patients.13
“The most highly expressed protein in head and neck cancer is EGFR, but EGFR is expressed at high levels in the liver, skin, and gut,” said Cohen. “It’d be very difficult to get a CAR against EGFR because it would be too toxic.”
Tyrosine kinaselike orphan receptor (ROR1) has been identified as a promising target for CAR T-cell therapy because the receptor is produced in solid and hematologic malignancies but not in normal cells, according to Cohen. Additionally, ROR1 expression has been shown to enhance tumor cell growth and survival and promote epithelial–mesenchymal transition and metastasis,14,15and high ROR1 expression has been associated with shorter overall survival in triple-negative breast cancer (TNBC), lung adenocarcinoma, and ovarian cancer.16-19Although the work by Cohen and his colleagues, funded by the California Institute for Regenerative Medicine, is still in the preclinical stages, he anticipates that his ROR1-directed CAR T-cell therapy will be tested in human trials within the next 12 to 18 months. An ongoing phase I clinical trial (NCT02706392) sponsored by the Fred Hutchinson Cancer Research Center in Seattle is investigating a different ROR1-targeted CAR T-cell therapy in patients with advanced, treatment-refractory, ROR1-positive chronic lymphocytic leukemia, mantle cell lymphoma, ALL, NSCLC, or TNBC.
Cohen also noted that the incidences of cytokine release syndrome observed with CAR T-cell therapy for hematologic malignancies may be a concern for HNSCC and other solid tumors, but he was optimistic that these will become less common as researchers learn how to predict them. “We’ve learned to anticipate those in hematologic malignancies,” said Cohen. “By the time we’re treating solid tumors in head and neck cancer with CARs, hopefully that won’t be as much of a concern.”
Addressing Resistance Mechanisms
Once effective CAR T-cell therapies are developed for head and neck cancer, resistance will likely be the next major concern, according to Cohen.
“We’d love to have that as a real-time concern,” he said. “The reality is that for cancer to become resistant to a therapy, that therapy has to work in the first place. [Resistance] is almost certainly going to happen, and it’s going to happen through loss of expression of that receptor, specific clones that eventually grow out, and highly immunosuppressive microenvironments that develop.”
Cohen noted that the research on resistance mechanisms to CAR T-cell therapies in hematologic malignancies will help guide research in solid tumors. While CD19-targeted CAR T-cell therapy has been shown to yield a 90% response rate in patients with relapsed ALL, it also led to CD19 epitope loss in 10% to 20% of patients.8Further research has shown that disease relapse was associated with the development of alternative splicing variants of the CD19 protein that were still functional in promoting B-cell proliferation.20Thus, researchers are currently working on designing approaches that tackle these resistance mechanisms, such as designing new antibodies that recognize alternative epitopes in the CD19 domain, forcing the cell to produce a full-length CD19, or co-expression of CARs.
“If you become more sophisticated with your engineering and begin to coexpress CARs or have more than 1 target, you can begin to get around that initial resistance and hopefully eliminate every cancer cell so that resistance never develops,” said Cohen.
Although adoptive immunotherapy approaches are still in the infancy stages for head and neck cancer, Cohen was optimistic that they would soon play a role in treatment, possibly in conjunction with checkpoint inhibitors, for patients with chemotherapy-refractory disease and eventually in the first-line recurrent metastatic setting.
“I have no doubt that we will see T-cell engineering in head and neck cancer later this year or early next year,” said Cohen. “We’ll begin to see the first CARs and more TCRs. We’ll find a way to make them work in solid tumors, and I think they’ll eventually work quite well. It’s just a matter of getting there.”
References:
- Harris DT, Kranz DM. Adoptive T cell therapies: a comparison of T cell receptors and chimeric antigen receptors.Trends Pharmacol Sci.2016;37(3):220-230. doi: 10.1016/j.tips.2015.11.004
- Smith C, Tsang J, Beagley L, et al. Effective treatment of metastatic forms of Epstein-Barr virusassociated nasopharyngeal carcinoma with a novel adenovirus- based adoptive immunotherapy.Cancer Res.2012;72(5):1116-1125. doi: 10.1158/0008-5472.CAN-11-3399.
- Jiang P, Zhang Y, Archibald SJ, Wang H. Adoptive cell transfer after chemotherapy enhances survival in patients with resectable HNSCC.Int Immunopharmacol.2015;28(1):208-214. doi: 10.1016/j.intimp.2015.05.042.
- Nishio-Nagai M, Suzuki S, Yoshikawa K, et al. Adoptive immunotherapy combined with FP treatment for head and neck cancer: an in vitro study.Int J Oncol.2017;51(5):1471-1481. doi: 10.3892/ijo.2017.4142.
- Hinrichs CS, Doran SL, Stevanovic S, et al. A phase I/II clinical trial of E6 T-cell receptor gene therapy for human papillomavirus (HPV)-associated epithelial cancers.J Clin Oncol.2017;35(suppl; abstr 3009). meetinglibrary.asco.org/record/152695/ abstract.
- XPRESIDENT. Immatics US, Inc, website. immatics.com/x-president.html. Accessed March 26, 2018.
- Blumenschein GR, Bourgogne A, Reinhardt C, et al. Phase I trial evaluating genetically modified autologous T cells (ACTengine IMA201) expressing a T-cell receptor recognizing a cancer/germline antigen in patients with squamous NSCLC or HNSCC.J Clin Oncol.2018;36(suppl 5S; abstr TPS78). meetinglibrary.asco.org/ record/156705/abstract.
- Maude SL, Frey N, Shaw PA, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia.N Engl J Med.2014;371(16):1507-1517. doi: 10.1056/NEJMoa1407222.
- Garfall AL, Maus MV, Hwang WT, et al. Chimeric antigen receptor T cells against CD19 for multiple myeloma.N Engl J Med.2015;373(11):1040-1047. doi: 10.1056/ NEJMoa1504542.
- Schuster SJ, Svoboda J, Chong EA, et al. Chimeric antigen receptor T cells in refractory B-cell lymphomas.N Engl J Med.2017;377(26):2545-2554. doi: 10.1056/ NEJMoa1708566.
- Papa S, Adami A, Metoudi M, et al. T4 immunotherapy of head and neck squamous cell carcinoma using pan-ErbB targeted CAR T-cells.Cancer Res.2017;77(suppl 13):abstr CT118. doi: 10.1158/1538-7445.AM2017-CT118.
- Early trial results show potential for T-cell therapy in head and neck cancer. Targeted Oncology website. targetedonc.com/news/early-trial-results-show-potentialfor- car-tcell-therapy-in-head-and-neck-cancer. Published April 20, 2017. Accessed March 26, 2018.
- Parkhurst MR, Yang JC, Langan RC, et al. T cells targeting carcinoembryonic antigen can mediate regression of metastatic colorectal cancer but induce severe transient colitis.Mol Ther.2011;19(3):620-626. doi: 10.1038/mt.2010.272.
- Zhang S, Chen L, Cui B, et al. ROR1 is expressed in human breast cancer and associated with enhanced tumor-cell growth.PloS One.2012;7(3):e31127. doi: 10.1371/journal.pone.0031127.
- Cui B, Zhang S, Chen L, et al. Targeting ROR1 inhibits epithelialmesenchymal transition and metastasis.Cancer Res.2013;73(12):3649-3660. doi: 10.1158/0008-5472.CAN-12-3832.
- Chien HP, Ueng SH, Chen SC, et al. Expression of ROR1 has prognostic significance in triple negative breast cancer.Virchows Arch.2016;468(5):589-595. doi: 10.1007/s00428-016-1911-3.
- Zhang H, Qiu J, Ye C, et al. ROR1 expression correlated with poor clinical outcome in human ovarian cancer.Sci Rep.2014;4:5811. doi: 10.1038/srep05811.
- Liu Y, Yang H, Chen T, et al. Silencing of receptor tyrosine kinase ROR1 inhibits tumor-cell proliferation via PI3K/AKT/mTOR signaling pathway in lung adenocarcinoma.PloS One.2015;10(5):e0127092. doi: 10.1371/journal.pone.0127092.
- Karachaliou N, Gimenez-Capitan A, Drozdowskyj A, et al. ROR1 as a novel therapeutic target for EGFR-mutant non-small-cell lung cancer patients with the EGFR T790M mutation.Transl Lung Cancer Res.2014;3(3):122-130. doi: 10.3978/j. issn.2218-6751.2014.03.02.
- Sotillo E, Barrett DM, Black KL, et al. Convergence of acquired mutations and alternative splicing of CD19 enables resistance to CART-19 immunotherapy.Cancer Discov.2015;5(12):1282-1295. doi: 10.1158/2159-8290.CD-15-1020.
Biomarker Testing Paves the Way for Better Targeted Therapies in NSCLC
April 16th 2024At a live virtual event, Edward S. Kim, MD, MBA, discussed the evolving landscape of biomarker testing before making treatment decisions for patients with early-stage non–small cell lung cancer (NSCLC).
Read More
Creating Solutions for a 'Continual State of Transition' in Cancer Care
April 15th 2024In a Peers & Perspectives in Oncology feature article, we focus on the importance of the transition-of-care process for patients with cancer as they move from the inpatient to outpatient setting, as well as between lines of therapy with comments from Marc J. Braunstein, MD, PhD, and Michael Shusterman, MD.
Read More