Induction Chemotherapy Improves Symptom Burden in Oropharynx Carcinoma
Patients with nonmetastic squamous cell carcinoma of the oropharynx and unknown primaries treated with induction chemotherapy followed by chemoradiotherapy had fewer symptom burdens, including need of a feeding tube, due to a significant response in primary and nodal sites.
Frank E. Mott, MD

Frank E. Mott, MD
Patients with nonmetastatic squamous cell carcinoma (SCC) of the oropharynx and unknown primaries treated with induction chemotherapy (IC) followed by chemoradiotherapy had fewer symptom burdens, including need of a feeding tube, due to a significant response in primary and nodal sites.
“IC was well tolerated with reasonable response rates,” wrote the study investigators led by Frank E. Mott, MD, associate professor in the Department of Thoracic/Head and Neck Medical Oncology of the Division of Cancer Medicine at The University of Texas MD Anderson Cancer Center in Houston, who presented their data in a poster session during the American Head and Neck Society 2019 Annual Meeting. “Based on patient-reported outcomes, the percentage of patients experiencing dysphagia, xerostomia, mucositis, pain, and distress improved from baseline following completion of IC.”
According to the investigators, the use of IC in oropharyngeal SCC remains controversial, mostly due to conflicting data and the lack of consistency in previous trials. The effects of its use prior to locoregional therapy for the purposes of tumor volume reduction do not have a known overall survival (OS) benefit.
Using the Oropharynx Program database at MD Anderson, the investigators performed a retrospective study of patients treated from 2014 to 2018. Of 684 patients >18 years enrolled in the database, 125 had received IC followed by either chemoradiotherapy or radiation therapy alone.
Patient characteristics showed that most were men (88%), white (87%), and positive for human papilloma virus (72%). The majority of patients who received IC had advanced disease, defined as nodal stage N2b or greater, and either tumor stage T3 (20%) or T4 (35.2%).
The IC regimen of choice was docetaxel, cisplatin, and fluorouracil in 47 patients (37.6%); paclitaxel, carboplatin, and cetuximab (Erbitux) in 39 (31.2%); cisplatin and docetaxel in 15 (12%); carboplatin and docetaxel in 10 (8%); carboplatin and docetaxel in 9 (7.2%); and carboplatin, cetuximab, and docetaxel in 5 (4%).
The most common reasons for IC were bulky nodal disease ≥N2b, bulky primary disease ≥T3, and low-neck disease. Other reasons included radiotherapy delay due to dental extractions, symptom relief, organ or voice preservation, or treatment on a clinical trial.
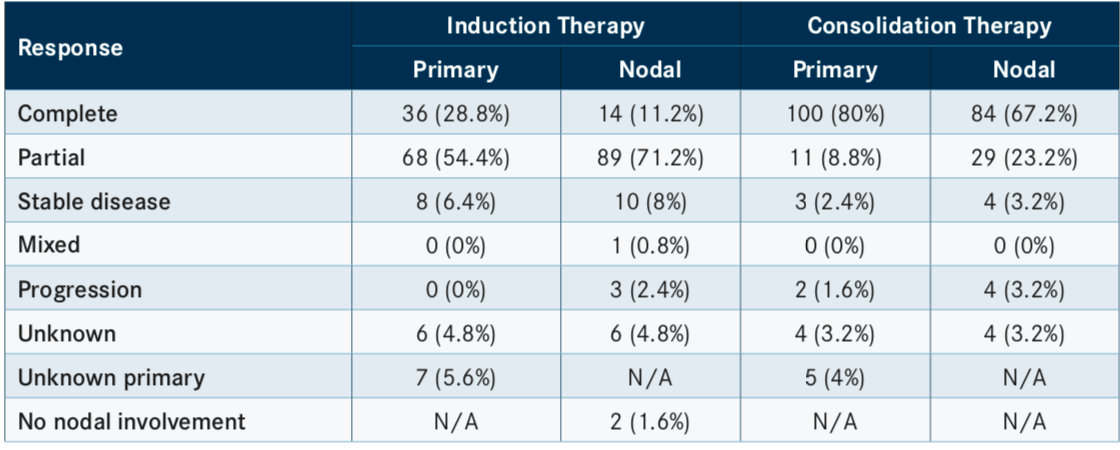
The overall response to IC therapy was 88%, with 9 patients (7.2%) achieving a complete response (CR) in both primary and nodal sites or an unknown primary with a CR. Patients achieving a CR in the primary site and partial response (PR) in the nodal sites made up 20.8% of patients. Conversely, 4% of patients had a PR in the primary and CR in nodal sites. PR was achieved in the primary and nodal sites, nodal sites in an unknown primary tumor, or in a primary tumor with no nodal involvement in 50.4% of patients. Finally, there were 5.6% of patients with stable disease and a PR at 1 site each.
At the median follow-up of 22 months (range, 3-84), 103 patients (82.4%) were alive. The median time to relapse in 32 patients (25.6%) was 11 months, and the median time to death in 23 was 19 months; 5 deaths in the cohort were due to disease progression.
Compared with patients receiving chemoradiotherapy or radiation alone, less than half of those who had IC required a feeding tube during their treatment course (55.9% vs 45.6%). Lymphedema consultation within 6 months of chemoradiotherapy occurred in 13.6% of patients receiving IC.
Patient-reported outcomes were assessed at baseline, the end of IC, and the end of chemoradiotherapy/radiotherapy using the MD Anderson Symptom Inventory to determine the severity of disease symptoms. Data are available for 100 patients (80%).
Many symptoms were temporarily improved by treatment with IC, including patient-reported distress and pain. At baseline, 42% of patients reported xerostomia, which decreased to 36% following IC and increased again to 56% at the end of chemoradiotherapy; dysphagia was reported in 51%, 29%, and 57% of patients and mucositis in 31%, 24%, and 56%, respectively.
In total, 100 patients (80%) achieved a CR in the primary tumor site and 84 (67.2%) had a CR in nodal sites following consolidation response. Progression in primary and nodal sites was observed in 2 (1.6%) and 4 (3.2%) patients, respectively. Unknown responses in both primary and nodal sites were noted in 4 patients each (TABLE).
Future investigation of the same database will assess efficacy outcomes, such as OS and progression-free survival.
“Although IC does not have an established survival advantage, it does positively impact patient symptom burden,” Mott and colleagues wrote. “IC may be considered more frequently for patients presenting with a high symptom burden due to bulky primary and/or nodal disease.”
Reference:
Mott F, Sacks R, Gillison M, et al. Induction chemotherapy in oropharyngeal carcinoma. Poster presented at: American Head and Neck Society 2019 Annual Meeting; May 1-2, 2019; Austin, TX. Abstract D234. bit.ly/2WUGXg3.

Advances in Subsequent Therapies Shake Up Sequencing of ccRCC Treatment
April 25th 2024With the approval of belzutifan and other newer data for treating patients with recurrent renal cell carcinoma, the state of subsequent therapies is advancing beyond the reuse of frontline options with impacts on duration of response and quality of life.
Read More
Novel Approaches Focus on Limiting Toxicity in Older Patients With ALL
April 22nd 2024The major challenges for clinicians treating older patients with acute lymphoblastic leukemia surround the emergence of resistance to existing therapies and the toxicities associated with current chemotherapies.
Read More
Ornstein Advises on Starting Dose and Management of Lenvatinib in RCC
April 21st 2024During a Case-Based Roundtable® event, Moshe Ornstein, MD, MA, provided guidance on dosing and toxicity concerns in a patient treated with lenvatinib plus pembrolizumab for advanced renal cell carcinoma.
Read More