Monitoring of ctDNA in Patients With Bladder Cancer Has Potential to Assist Therapy Selection
By testing circulating tumor DNA at critical time points throughout neoadjuvant chemotherapy and cystectomy, clinicians may be able to better predict which patients with advanced bladder cancer would likely benefit from additional systemic therapies, according to results of a study published in the <em>Journal of Clinical Oncology</em>.
Emil Christensen, PhD

Emil Christensen, PhD
By testing circulating tumor DNA (ctDNA) at critical time points throughout neoadjuvant chemotherapy and cystectomy,
clinicians may be able to better predict which patients with advanced bladder cancer would likely benefit from additional systemic therapies, according to results of a study published in theJournal of Clinical Oncology.
“We have found ctDNA testing in patients with bladder cancer who undergo chemotherapy and cystectomy to be highly sensitive [100%] and specific [98%] for early risk stratification of patients, prediction of treatment response, and early detection of metastatic relapse,” said the study investigators, led by Emil Christensen, PhD, of Aarhus University Hospital in Denmark. “CtDNA biomarkers are superior to tumorcentric biomarkers for predicting treatment efficacy.”
Investigators on the study enrolled 99 patients with muscleinvasive bladder cancer (MIBC) who were receiving neoadjuvant chemotherapy to undergo plasma sample collection starting at diagnosis and throughout disease surveillance. In total, 68 patients fulfilled the inclusion criteria and were used in the analysis.
A previously validated ctDNA analysis pipeline was applied to 656 plasma samples. The patient-specific assays were designed to detect 16 somatic mutations. Multiplex polymerase chain reaction next-generation sequencing was performed using the QIAamp Circulating Nucleic Acid Kit on plasma cell-free DNA. Samples were considered ctDNA positive if ≥2 variants were detected with a confidence score above 0.97.
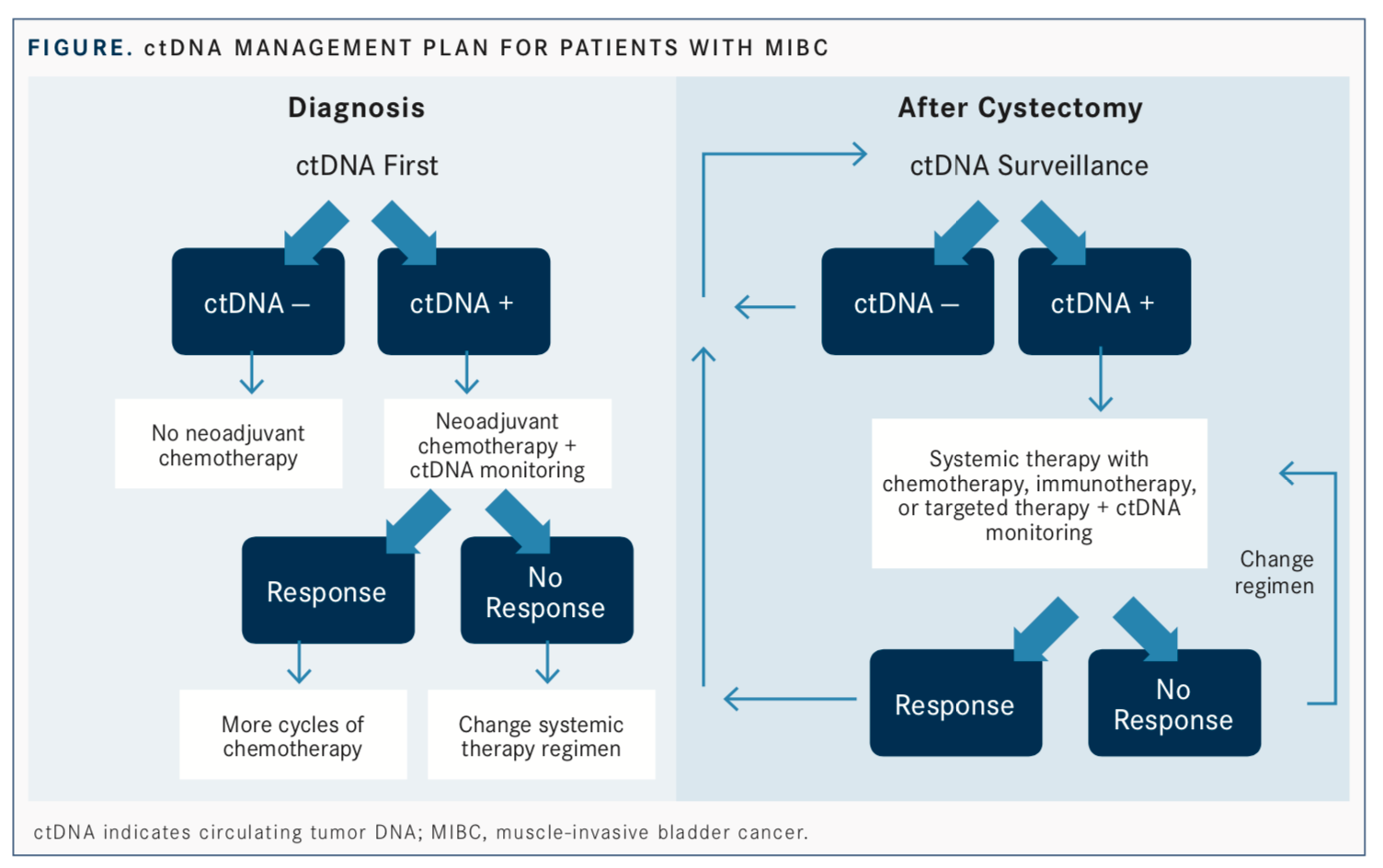
Investigators determined that sampling was of highest significance at 3 time points: at diagnosis before chemotherapy, after chemotherapy but before cystectomy, and during disease surveillance.
The detection of ctDNA positivity at diagnosis and before systemic intervention occurred in 24 patients, and the 12-month and overall recurrence rates in these patients were 42% and 46%, respectively, versus a recurrence rate of only 3% overall in those who were ctDNA negative (n = 35).
After chemotherapy, comparing patients who were ctDNA positive (n = 8) with those who were ctDNA negative (n = 55) yielded a 12-month recurrence rate of 75% versus 7% (P<.001) and an overall recurrence rate of 75% versus 11%, respectively (P<.001).
Christensen et al said that ctDNA status during surveillance was the most prognostically significant time point for sampling and was a stronger predictor of recurrence-free survival than any other predictive factor, such as lymph node status before cystectomy and pathologic downstaging (HR, 129.6; P<.001). The recurrence rates in patients who were ctDNA negative (n = 47) were 0% at both 12 months and for the duration of the study (P<.001). In patients with ctDNA positivity (n = 17), rates of 12-month and overall recurrence were 59% and 76%, respectively.
Predictive biomarkers of treatment response in the primary tumor were also assessed and showed a higher contribution of the trinucleotide mutational signature 5 in patients with responses to chemotherapy (P= .024); this was further associated with ERCC2mutational status, which also correlates with DNA damage response mechanisms. Upon further analysis, the correlation with chemotherapy responses in patients with ERCC2mutations was found to not reach statistical significance (P= .47). These findings reinforced ctDNA monitoring as the strongest predictor of metastatic spread and therapy response in patients with high-risk MIBC.
Patients with ctDNA negativity before chemotherapy, for example, may be eligible for cystectomy with neoadjuvant chemotherapy because their overall risk of disease recurrence is low.
Monitoring of ctDNA throughout chemotherapy may also help predict who would benefit from other therapeutic strategies. Clearance of ctDNA was associated with pathologic downstaging in 53% of patients who showed a response versus 0% with no clearance.
Finally, ctDNA detection after cystectomy is indicative of remnant disease. Patients who were found to have metastatic relapse based on ctDNA analysis had a median lead time of 96 days over conventional imaging techniques (range, 83 to 245;P= .023). This provides a potential “window of opportunity” for early intervention and, hopefully, improved efficacy, according to Christensen et al.
“On the basis of these findings, new paradigms for ctDNA-guided patient management should be investigated in future clinical trials,” the investigators concluded.TT
Reference:
Christensen E. Birkenkamp-Demtroder K, Sethi H, et al. Early detection of metastatic relapse and monitoring of therapeutic efficacy by ultra-deep sequencing of plasma cell-free DNA in patients with urothelial bladder carcinoma [published online May 6, 2019].J Clin Oncol. doi: 10.1200/JCO.18.02052.

FDA Approves Nogapendekin Alfa Inbakicept for BCG-Unresponsive NMIBC Carcinoma In Situ
April 22nd 2024Patients with Bacillus Calmette-Guérin-unresponsive non-muscle-invasive bladder cancer carcinoma in situ now have a new treatment option following the FDA’s approval of nogapendekin alfa.
Read More
Novel Approaches Focus on Limiting Toxicity in Older Patients With ALL
April 22nd 2024The major challenges for clinicians treating older patients with acute lymphoblastic leukemia surround the emergence of resistance to existing therapies and the toxicities associated with current chemotherapies.
Read More
Ornstein Advises on Starting Dose and Management of Lenvatinib in RCC
April 21st 2024During a Case-Based Roundtable® event, Moshe Ornstein, MD, MA, provided guidance on dosing and toxicity concerns in a patient treated with lenvatinib plus pembrolizumab for advanced renal cell carcinoma.
Read More
Rugo Surveys Peers on Treatment After Metastatic Relapse of HR+, HER2– Breast Cancer
April 20th 2024During a Case-Based Roundtable® event, Hope S. Rugo, MD, FASCO, moderated a discussion on treatment options for a patient whose breast cancer recurred several years after adjuvant therapy.
Read More