Patient With a PIK3CA-Positive Tumors Exhibits Hyperglycemia Associated With Alpelisib
The approval of alpelisib in May 2019 poses a challenge to oncologists and their practices with the emergence of hyperglycemia as an AE associated with PI3K inhibitors. Managing the patient effectively requires a multidisciplinary team approach that involves counseling up front, monitoring throughout the course of therapy, and input and feedback from the team of oncologists, advanced practice providers, pharmacists, and dietitians.
In may 2019, the fda approved the first PI3K inhibitor indicated for patients with hormone receptorpositive, HER2-negative, PIK3CA-mutated, advanced or metastatic breast cancer following progression on or after an endocrine-based regimen.
The approval was based on the phase III SOLAR-1 trial,1which demonstrated that in a subset of patients withPIK3CAmutations, the median progression-free survival (PFS) by local assessment was 11.0 months (95% CI, 7.5-14.5) for those who received the alpelisib (Piqray) plus fulvestrant combination compared with 5.7 months (95% CI, 3.7-7.4) for those who received placebo plus fulvestrant. Those results, assessed after a median follow-up of 20 months, translated into a 35% reduction in the risk of progression or death, with a hazard ratio of 0.65 in favor of alpelisib (95% CI, 0.50-0.85;P= .00065). There was no advantage to alpelisib regarding median PFS in patients without aPIK3CAmutation.
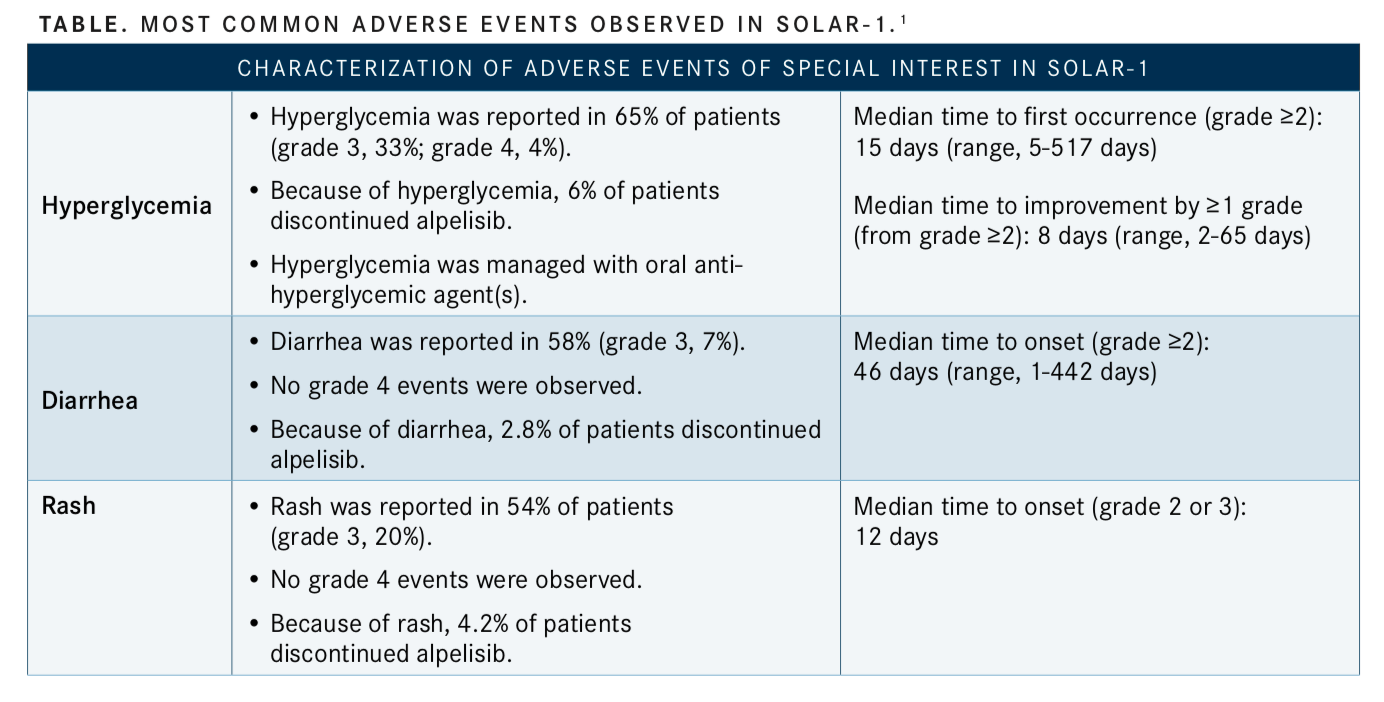
Common adverse effects (AEs) of alpelisib are hyperglycemia, increased creatinine, diarrhea, rash, decreased lymphocyte count, elevated liver enzymes, nausea, fatigue, low red blood cell count, increased lipase, decreased appetite, stomatitis, vomiting, weight loss, low calcium levels, prolonged activated partial thromboplastin time, and hair loss (TABLE).1
The investigators noted that an on-target effect of PI3K inhibition is the development of hyperglycemia. In the total cohort of SOLAR-1, 18 patients (6.3%) discontinued alpelisib treatment due to hyperglycemia. In the combination arm, 181 patients (63.7%) reported all-grade hyperglycemia. Ninety-three patients (32.7%) had grade 3 hyperglycemia, and 11 (3.9%) had grade 4 hyperglycemia. In the placebo arm, 28 (9.8%) patients had hyperglycemia, 1 (0.3%) had grade 3, and 1 (0.3%) had grade 4 hyperglycemia.2
In the alpelisib arm, 56% of patients were prediabetic at baseline (fasting plasma glucose [FPG], 5.6-7.0 mmol/L) and 4% were diabetic (FPG ≥7.0 mmol/L), based on FPG and glycated hemoglobin A1C (HbA1C) levels. Patients with uncontrolled diabetes were excluded from the trial. Increases in FPG and HbA1C were more pronounced in patients who were diabetic or pre-diabetic at baseline. Among patients who were prediabetic, 74% experienced hyperglycemia during the treatment period. The investigators reported that mean FPG values peaked within the first 2 weeks, then decreased toward baseline FPG levels.
Patients being treated with alpelisib have experienced severe hyperglycemia, and the safety of alpelisib in those with type 1 or uncontrolled type 2 diabetes is not known. The FDA advises healthcare professionals to obtain fasting glucose and HbA1C levels and optimize glycemic control.
The FDA also approved the companion diagnostic test, therascreen PIK3CA RGQ PCR Kit, to detect thePIK3CAmutation in a tissue and/ or liquid biopsy. Patients who are negative by the test using the liquid biopsy should undergo tumor biopsy forPIK3CAmutation testing.
Hematology-Oncology Associates of Central New York
Since 1982, Hematology-Oncology Associates of CNY (HOA), the private practice of John J. Gullo, MD, and Santo M. DiFino, MD, has been dedicated to caring for people with blood disorders and cancer. Gullo and DiFino’s vision is to serve the community by providing the highest level of quality care to patients dealing with cancer and blood disorders. They were soon joined by Jeffrey J. Kirshner, MD, and Anthony J. Scalzo, MD. These 4 founding partners have subsequently been joined by physicians who are also dedicated to the HOA mission. Gullo and DiFino are now retired.
The approval of alpelisib in May 2019 poses a challenge to oncologists and their practices with the emergence of hyperglycemia as an AE associated with PI3K inhibitors. Managing the patient effectively requires a multidisciplinary team approach that involves counseling up front, monitoring throughout the course of therapy, and input and feedback from the team of oncologists, advanced practice providers, pharmacists, and dietitians. Whenever possible, these and other toxicities are managed within the practice before referral is made to a specialist.
In general, our multidisciplinary approach includes extensive counseling of patients about their disease state and our plan of treatment, as well as the expected AEs and plans for minimizing and treating toxicities. This includes a formal teaching session at the onset of therapy with the advanced practice providers and at dispensing, when instruction is given by our pharmacist and nurse navigator, as well as counseling services throughout therapy.
A patient with breast cancer presented with aPIK3CAmutation and was considered a candidate for alpelisib. The patient fully understood the anticipated AEs and our management strategy upon initiation of therapy. It should be noted that this patient was not as heavily pretreated as the patients in SOLAR-1.
We considered adding metformin to the patient’s daily regimen of alpelisib 300 mg during week 2 of treatment; however, the patient’s baseline fasting blood sugar of 115 mg/dL doubled to 233 mg/dL by week 2.
Therefore, metformin was initiated at the recommended dose of 500 mg by mouth daily. Unfortunately, the patient developed a full body rash within 3 days. Because we were familiar with the AEs associated with alpelisib, rash and diarrhea, we were prepared to deal with this development. Alpelisib and metformin were held while the patient was treated with a 10-day corticosteroid taper.
When the rash cleared, we rechallenged the patient with a reduced dose of alpelisib at 250 mg daily and restarted metformin. We were not prepared for facial swelling and throat tightness. She also reported severe nausea and vomiting unrelieved by prochlorperazine (Compazine), lorazepam (Ativan), or ondansetron (Zofran). She also had stomach, joint, muscle, and low back pain.
Did alpelisib or metformin cause this second round of AEs? Both were new therapies for our patient, and these were anticipated complications of both. Having already exhausted multiple lines of breast cancer treatment, we were hopeful that we could treat her effectively for herPIK3CAmutation and provide her an additional year of progression-free survival. We needed a plan to treat her toxicities and keep her on therapy.
Both drugs were withheld a second time until her symptoms resolved. It was decided that she would take the alpelisib at home, then come to the office within 3 hours (Cmax 4 hours) for observation and possible treatment if she again reacted and needed additional support.
She had no reaction from the alpelisib. It was determined that her allergic reaction was caused by metformin. We then faced the challenge of how to treat her hyperglycemia, because metformin was no longer an option.
We were confronted by a number of treatment options: Do we use a sulfonylurea and risk hypoglycemia if she had additional nausea, vomiting, or diarrhea, which are expected during week 3 of treatment with alpelisib? Do we use a glucagonlike peptide1 agonist or dipeptidyl peptidase–4 agonist and risk additional nausea, vomiting, and diarrhea? Do we use a sodium-glucose cotransporter-2 inhibitor and risk further weight loss? She had already lost approximately 5 pounds in 3 weeks. As a result of these considerations, insulin became our primary choice, but we were not certain if our patient was motivated to administer daily injections of insulin and check her blood sugar levels 3 or 4 times daily.
As a team, we decided to start her on a low dose of basal insulin (0.1 units/kg = 6 units daily), based on her willingness to self-administer the injections and check her blood sugar level throughout the day. She called us every 2 or 3 days with her readings. Her basal insulin was slowly titrated up based on her elevated home blood sugar readings, with a goal of maintaining a fasting blood sugar less than 140 mg/dL. As we escalated her basal insulin to >30 units daily and continued to see her postprandial readings elevated, we added sliding scale insulin, a progressive increase of insulin in the premeal or nighttime insulin dose. Again, we treated conservatively, starting at a range of 6 to 12 units, but her blood sugar levels were still 250 to 350 mg/dL and, in some cases, 450 mg/dL.
We were in frequent contact with our patient, and counseled her regularly about diet. We referred her to our dietitian for additional counseling. She was not consuming excessive amounts of sugar, simple carbohydrates, or alcohol. In fact, her diet consisted mostly of lean protein and vegetables. So why were her blood sugar levels so high, and why were her bedtime readings well over 200 mg/dL? Even though we were not treating her at bedtime with short-acting insulin, her blood sugar had time to decrease overnight, although they were not at a goal fasting level of 80 to 100 mg/dL.
Was this all an on-target effect of the alpelisib, indicating that it was working, and did it correspond to the kinetics of the drug? She usually took her first dose of alpelisib around 7 am, and her highest blood sugar levels seemed to correspond to the Cmax of the drug around 11 am. Her lowest blood glucose levels were in the morning fasting state but also when the drug was reaching its minimum concentration.
We continued to follow our patient for additional AEs. She had no complaints of nausea, vomiting, diarrhea, or rash, so we increased her dose of alpelisib back to 300 mg. She did have some fatigue and thirst associated with her hyperglycemia.
As our patient was approaching potential insulin resistance at doses of 2 units/kg of total insulin daily (50 units of basal and 50 units of bolus sliding scale), we looked further into the mechanism of PI3K inhibition and its effect on glucose metabolism. We also sought help from the drug manufacturer and diabetes specialists in our area. Were we undoing the benefits of the alpelisib by treating her with so much exogenous insulin? Should we try a small dose of sulfonylurea to reduce her insulin requirement or use another agent completely and deal with those additional AEs?
We also knew that if her therapy with alpelisib was discontinued, she would have no need for the high doses of insulin or another hyperglycemia treatment, because this toxicity is reversible. We had already seen her blood sugars normalized when the dose was held, despite being on a 10-day course of corticosteroids.
Unfortunately, 3.5 months after initiating treatment with alpelisib, the patient progressed with worsening metastases in the liver. Therefore, in early August 2019, the alpelisib was discontinued. Within days, the patient’s glucose decreased despite stopping the insulin. Within a month, her glucose normalized.
The hyperglycemic effect of alpelisib appears to be unique to that targeted agent. We have not seen hyperglycemia as an AE with other targeted agents.
The advantage of working with a multidisciplinary team is the ability to comprehensively address all the patient’s medical issues, not only the malignancy but also the AEs of the treatment. Having the team within our practice facilitated communication and allowed appropriate changes to the treatment program to occur on a timely basis.
References
- André F, Ciruelos EM, Rubovszky G, et al. Alpelisib (ALP) + fulvestrant (FUL) for advanced breast cancer (ABC): results of the phase 3 SOLAR-1 trial. Presented at: European Society for Medical Oncology 2018 Congress; October 19-23; Munich, Germany. Abstract LBA3.
- Fasching PA. The advancing role of PI3K inhibitorsindustry satellite symposium: innovation in HR+, HER2-advanced breast cancer and the emerging role of biomarkers. Presented at: European Society for Medical Oncology Breast Cancer 2019 Annual Congress; May 2-4, 2019; Berlin, Germany.
Batalini Explores Role of UGT1A1 in Patients Treated With Sacituzumab Govitecan for HR+ MBC
April 22nd 2024During a Community Case Forum live event in partnership with The Arizona Clinical Oncology Society, Felipe Batalini, MD, discussed the TROPiCS-02 trial of sacituzumab govitecan and the impact of the UGT1A1 status on adverse event frequency.
Read More
Breast Cancer Leans into the Decade of Antibody-Drug Conjugates, Experts Discuss
September 25th 2020In season 1, episode 3 of Targeted Talks, the importance of precision medicine in breast cancer, and how that vitally differs in community oncology compared with academic settings, is the topic of discussion.
Listen
Ornstein Advises on Starting Dose and Management of Lenvatinib in RCC
April 21st 2024During a Case-Based Roundtable® event, Moshe Ornstein, MD, MA, provided guidance on dosing and toxicity concerns in a patient treated with lenvatinib plus pembrolizumab for advanced renal cell carcinoma.
Read More