Patients With Low-Risk Myeloma Could Prove Ideal for Daratumumab Therapy
The discussion about the pros and cons of a 4-drug regimen for patients with multiple myeloma was highlighted during the 2019 Debates and Didactics in Hematology and Oncology conference.
Ajay K. Nooka, MD, MPH

Ajay K. Nooka, MD, MPH
In the quest to improve the depth and duration of response for patients with newly diagnosed multiple myeloma, the quadruplet drug regimen daratumumab (Darzalex)/ bortezomib (Velcade)/thalidomide (Thalomid)/ dexamethasone (D-VTd) can now be considered versus the standard triplet regimen: bortezomib/thalidomide/dexamethasone (VTd). The discussion about the pros and cons of a 4-drug regimen in this disease setting was highlighted during the 2019 Debates and Didactics in Hematology and Oncology conference, a Winship Cancer Institute Annual Cancer Conference, held in Sea Island, Georgia.1
Ajay K. Nooka, MD, MPH, associate professor of medicine, was tasked with discussing the benefits of the 4-drug regimen, and Craig C. Hofmeister MD, MPH, associate professor of medicine, argued against its use. Both are oncologists at Winship Cancer Institute of Emory University in Atlanta, Georgia, and faculty members of Emory University School of Medicine.
Historically, treating multiple myeloma evolved from a 2-drug to a 3-drug regimen based on results from SWOG S0777,2 a phase III trial that evaluated the triplet combination (bortezomib/ lenalidomide (Revlimid)/dexamethasone; VRd) versus doublet (Rd) among patients with newly diagnosed myeloma. The endpoints of the trial were progression-free survival (PFS) and overall survival (OS). Median PFS was 13 months in favor of the VRd arm (HR, 0.712; 95% CI, 0.560-0.906;P= .0018), and the OS was 11 months in favor of the VRd arm (HR, 0.709; 95% CI, 0.524-0.959;P= .0250).2
“[From] the lessons that we learned in the past, going from a 2-drug to 3-drug regimen has allowed oncologists to embrace the 3-drug regimen,” Nooka said. “This makes my job much easier to convince you that a 4-drug regimen is the way to go.”
During his presentation, Nooka highlighted findings from the clinical trials CASSIOPEIA3(NCT02541383) and MAIA (MMY3008).4
Transplant-Eligible Patients
CASSIOPEIA examined the combination of daratumumab plus VTd versus VTd alone in 1085 previously untreated patients with multiple myeloma who were transplant eligible. Median follow-up was 18.8 months (range, 0.0-32.2). Findings for part 1 of the study demonstrated a 53% reduction in the risk of progression or death in the D-VTd arm (HR, 0.47; 95% CI, 0.33- 0.67;P<.0001).
“Median OS was not reached in either treatment arm, but at 15 months, we’re seeing some separation between the 2 arms of treatment,” Nooka said.
Nooka addressed safety concerns about adding a new agent to an established regimen: “The safety results are almost similar when the daratumumab arm is compared with the non-daratumumab arm.” Additionally, daratumumab did not have a significant impact on stem cell collection and transplantation.5
The clinical efficacy of daratumumab plus VRd (D-VRd) versus VRd alone is being evaluated in the randomized, open-label, parallel-assignment phase II GRIFFIN study.6In data presented at the 2018 American Society of Hematology Annual Meeting, D-VRd was shown to induce at least a very good partial response (VGPR) in all patients, with 63% (n = 16) achieving complete response (CR) or stringent CR at the end of consolidation therapy.
Transplant-Ineligible Patients
For transplant-ineligible patients, initial results from ALCYONE demonstrated that daratumumab plus the combination of bortezomib/ melphalan/prednisone (VMP) produced a 50% reduction in the risk of progression or death compared with VMP alone (HR, 0.50; 95% CI, 0.38-0.65;P<.001). The median PFS was 18.1 months in the VMP arm and not yet reached for those treated with the daratumumab regimen.7
The open-label, multicenter, phase III MAIA study followed 737 patients aged 45 to 90 years with newly diagnosed multiple myeloma who were ineligible for high-dose chemotherapy and autologous stem cell transplant (ASCT). Patients received either daratumumab/lenalidomide/dexamethasone (DRd) or Rd alone in 28-day cycles. The median follow-up was 28 months. Investigators reported that the median PFS for DRd had not been reached compared with 31.9 months for patients who received Rd alone. Moreover, in patients who received DRd, higher rates of complete response or better at 48% versus 25% were reported. The overall response rate was also higher with the triplet regimen, at 93% versus 81%, respectively.4Patients in the daratumumab group showed a 44% reduction in risk of disease progression or death versus lenalidomide/ dexamethasone alone (HR, 0.56; 95 CI, 0.43- 0.73;P<.0001).4FDA approval for this frontline indication was granted on June 27, 2019.8
“Adding a monoclonal antibody in the up-front setting results in deeper responses, but patients with high-risk myeloma, with a higher International Staging System [ISS] score or comorbidities such as impaired hepatic function, need further study,” Nooka concluded.
Value of Daratumumab
Hofmeister’s counterargument for the use of daratumumab in the upfront setting focused on its value, determined by OS and PFS benefit divided by adverse effects, administration and logistical challenges, and wholesale cost of daratumumab.
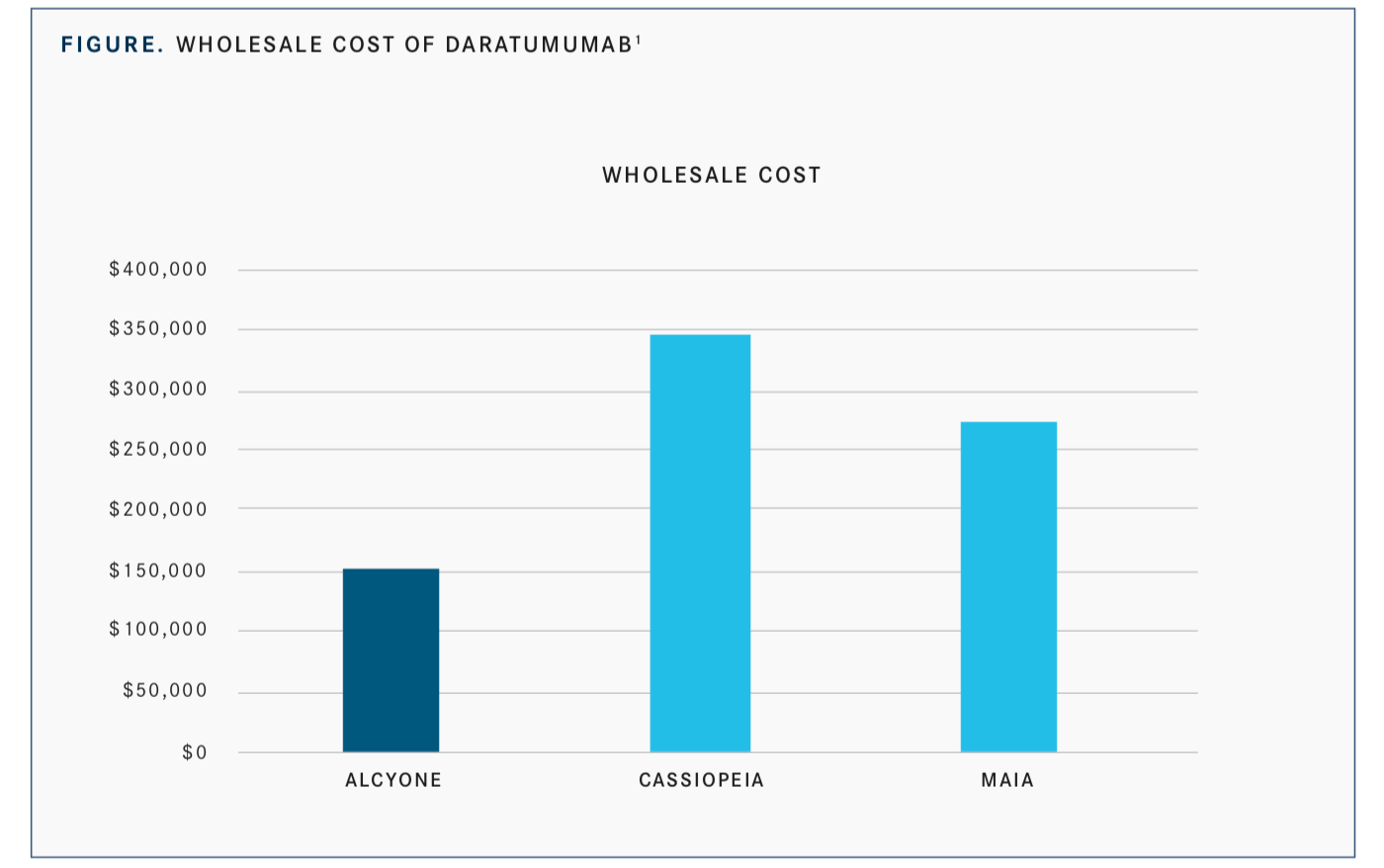
When the cost of daratumumab is factored into the 3 major trials involving the monoclonal antibody, MAIA, ALCYONE, and CASSIOPEIA, the additional cost is significant (FIGURE).1
“In general, across those 3 trials, you’re going to double your disease control with the addition of daratumumab,” Hofmeister said. “But there are also going to be challenges. You’re going to have more infections, you’re going to have weekly IV [intravenous] infusions, and there [are] going to be additional costs.”
When reviewing the trials relevant to the United Statesie, MAIA and CASSIOPEIA— Hofmeister noted that patients at high risk in both trials demonstrated no benefit. High-risk patients were defined as those with translocations at t(4;14), del(17p), or t(14/16) and an ISS score of 3. “Did these patients benefit in PFS? No, not at all,” he said. “And you are adding an additional cost with daratumumab.”
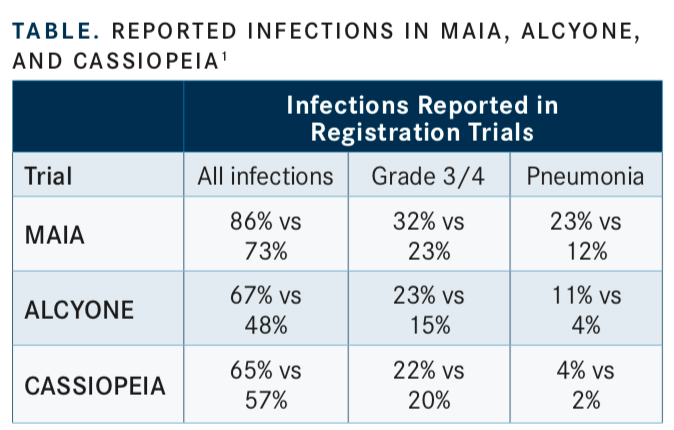
Hofmeister noted that adding the monoclonal antibody to the 3-drug regimen also comes with a medical cost, with increased infections observed during these major clinical trials. In MAIA, for all infections, patients in the daratumumab arm reported more infections (86% vs 73%). For ALCYONE, the rate of all infections with daratumumab was 67% versus without daratumumab (48%). Similarly, in CASSIOPEIA, the rate of all infections with daratumumab was 65% versus patients without daratumumab (57%) (TABLE).1
“You can expect that when giving daratumumab, your patients are going to have some significant infections, and, in fact, more infections than have been reported in the registration trials,” Hofmeister said.
When patients are stratified by high risk, standard risk, low risk, and unfit, Hofmeister suggested, daratumumab would fit for the low-risk and unfit patients. Low-risk patients are defined as having ISS 1 to 2 plus normal lactate dehydrogenase and no translocations for t(4;14), del(17p), or t(4;16). Similarly, unfit patients were those with an ISS of 1 to 2 and no translocations for t(14q32) or del(17p).
“In certain patients, especially those patients with low-risk myeloma, daratumumab could provide a benefit with a longer period of disease control, and that certainly can be useful,” he concluded.
References
- Nooka AJ, Hofmeister C. Should monoclonal antibodies be added to front-line myeloma treatment? Presented at: 2019 Debates and Didactics in Hematology and Oncology; July 24-28, 2019; Sea Island, GA.
- Durie BG, Hoering A, Abidi MH, et al. Bortezomib with lenalidomide and dexamethasone versus lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma without intent for immediate autologous stem-cell transplant (SWOG S0777): a randomised, open-label, phase 3 trial.Lancet. 2017;389(10068):519-527. doi: 10.1016/S0140- 6736(16)31594-X.
- Moreau P, Attal M, Hulin C, et al. Phase 3 randomized study of daratumumab (DARA) + bortezomib/thalidomide/dexamethasone (D-VTd) vs VTd in transplant-eligible (TE) newly diagnosed multiple myeloma (NDMM): CASSIOPEIA part 1 results.J Clin Oncol. 2019;37(suppl; abstr 8003). doi: 10.1200/JCO.2019.37.15_8003.
- Facon T, Kumar SK, Plesner T, et al. Phase 3 randomized study of daratumumab plus lenalidomide and dexamethasone (D-Rd) versus lenalidomide and dexamethasone (Rd) in patients with newly diagnosed multiple myeloma (NDMM) ineligible for transplant (MAIA).Blood. 2018;132(suppl 1;abstr LBA-2). doi: 10.1182/blood-2018-120737.
- Moreau P, Attal M, Hulin C, et al. Bortezomib, thalidomide, and dexamethasone with or without daratumumab before and after autologous stem-cell transplantation for newly diagnosed multiple myeloma (CASSIOPEIA): a randomised, open-label, phase 3 study.Lancet. 2019;394(10192):29-38. doi: 10.1016/S0140-6736(19)31240-1.
- Voorhees PM, Rodriguez C, Reeves B, et al. Efficacy and updated safety analysis of a safety run-in cohort from griffin, a phase 2 randomized study of daratumumab (Dara), bortezomib (V), lenalidomide (R), and dexamethasone (D; Dara-Vrd) vs. Vrd in patients (pts) with newly diagnosed (ND) multiple myeloma (MM) eligible for high-dose therapy (HDT) and autologous stem cell transplantation (ASCT).Blood. 2018;132(suppl 1;abstr 151). doi: 10.1182/blood-2018-151.
- Mateos MV, Dimopoulos MA, Cavo M, et al; ALCYONE Trial Investigators. Daratumumab plus bortezomib, melphalan, and prednisone for untreated myeloma.N Engl J Med. 2018;378(6):518-528. doi: 10.1056/ NEJMoa1714678.
- Janssen announces U.S. FDA approval of Darzalex (daratumumab) in combination with lenalidomide and dexamethasone for newly diagnosed patients with multiple myeloma who are transplant ineligible [news release]. Horsham, PA: Janssen Pharmaceutical Companies of Johnson & Johnson; June 27, 2019. prn.to/2ZSPDkN. Accessed August 23, 2019.

Biomarker Testing Paves the Way for Better Targeted Therapies in NSCLC
April 16th 2024At a live virtual event, Edward S. Kim, MD, MBA, discussed the evolving landscape of biomarker testing before making treatment decisions for patients with early-stage non–small cell lung cancer (NSCLC).
Read More