Roundtable Roundup: Lung Cancer Biomarkers
In a live virtual event, Yasir Y. Elamin, MD, discussed biomarker testing and management of non–small cell lung cancer (NSCLC) and a related patient case with participating physicians.
Yasir Y. Elamin, MD
Assistant Professor
Department of Thoracic/Head and Neck Medical Oncology
Division of Cancer Medicine
The University of Texas MD Anderson Cancer Center
Houston, TX

CASE SUMMARY
A 66-year-old man presented to his primary care physician complaining of visual disturbances, fatigue, and sporadic headaches. He had a medical history of hypertension managed with candesartan and hyperlipidemia managed with simvastatin in addition to a smoking history of 25 pack-years. A physical examination showed blood pressure level at 148/70 mm Hg and decreased breath sounds in the lower left lobe, but it showed an otherwise negative result for disease. Complete blood count and chemistry results were within normal limits.
A brain MRI scan demonstrated a 10-mm right parietal mass at the gray-white junction without significant vasogenic edema. A CT scan of the chest, abdomen, and pelvis revealed a 3.4-cm mass in the lower left lobe and several small liver metastases. Ultrasound-guided transthoracic needle biopsy result of the lung lesion showed grade 2 lung acinar adenocarcinoma. Staging was T2aN0M1c, and his ECOG performance status was 1. The patient had 90% PD-L1 expression on tumor cells (22C3 pharmDx test) and was anxious to start treatment.
CASE UPDATE
Molecular panel testing result: EGFR exon 19 deletion
CASE UPDATE
The patient received stereotactic radiosurgery while awaiting molecular studies. Osimertinib 80 mg once daily was initiated. He experienced a good partial response.

Roundtable Wrap-Upa
In this Case-Based Roundtable event on advanced metastatic NSCLC, Eamin covered important topics in the field, including the following:
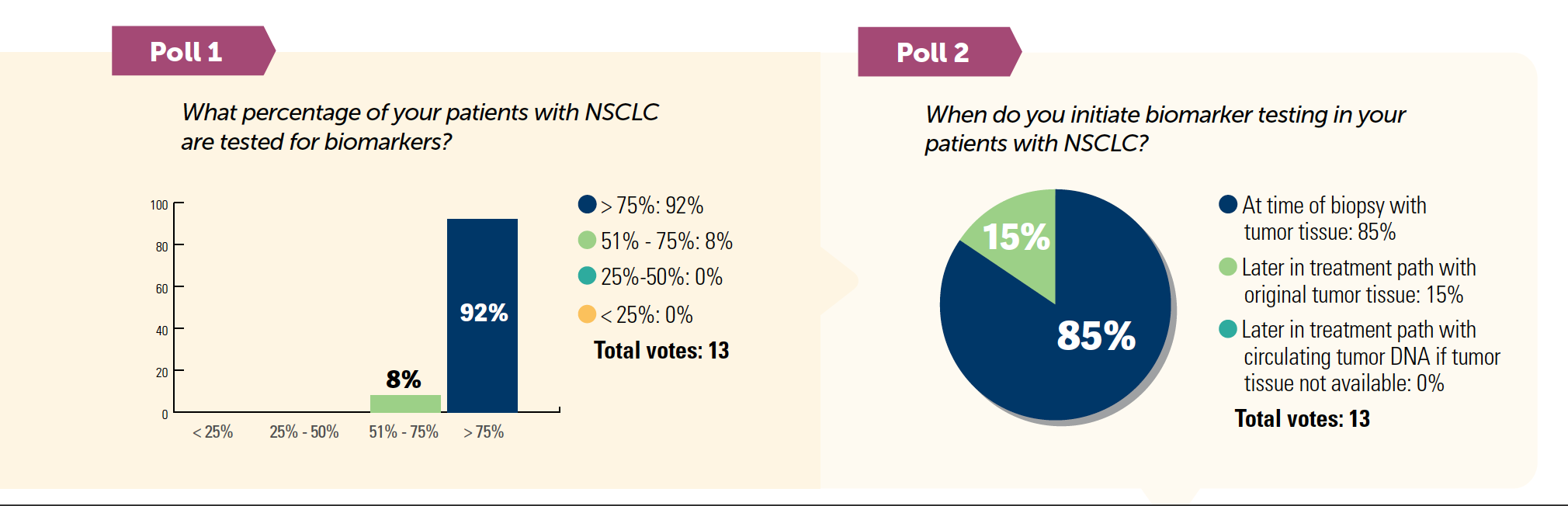
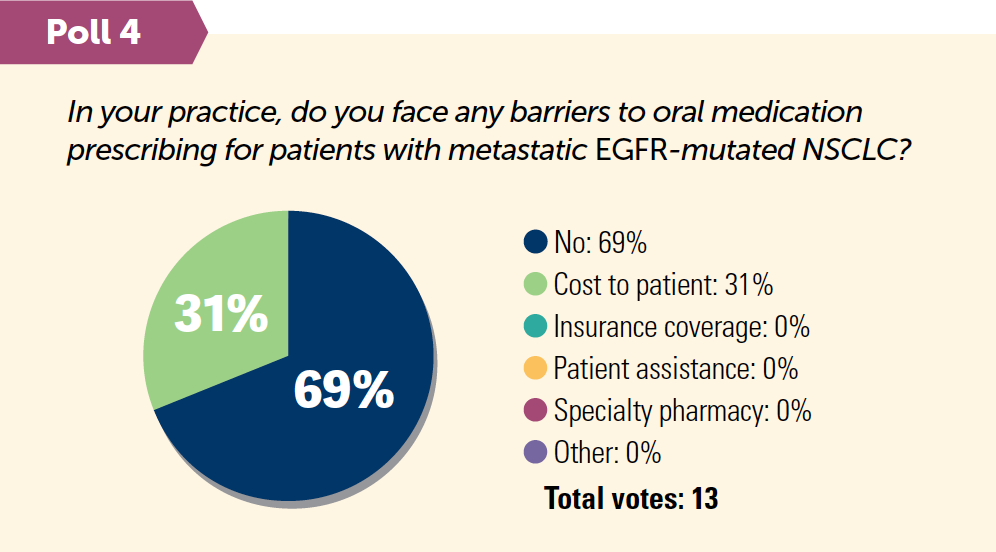
- Barriers to biomarker testing keep test rates down, especially for community practices. Access, awareness of current guidelines, cost, turnaround time, and quality or quantity of tissue are the main barriers for physicians.1,2
- According to the National Comprehensive Cancer Network (NCCN) guidelines, biomarker testing should be obtained before first-line therapy for EGFR, ALK, KRAS, ROS1, BRAF, NTRK1/2/3, MET, RET, and ERBB2 (HER2) genes.3
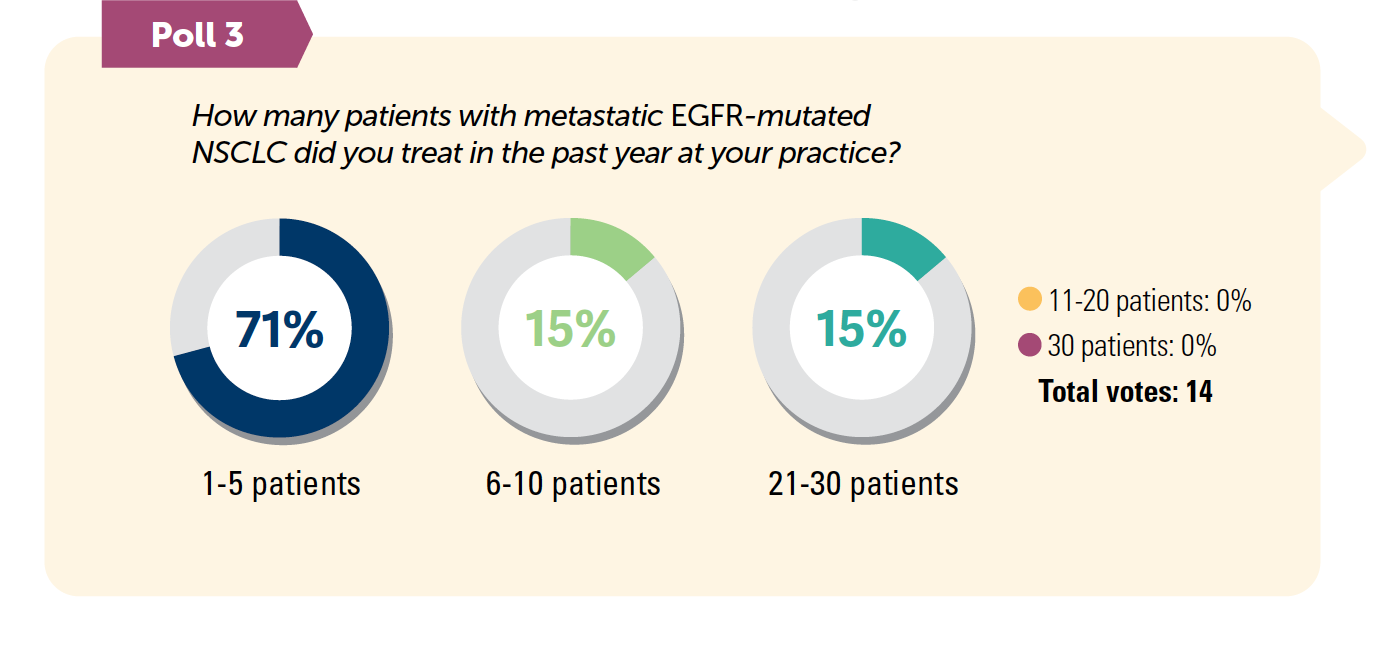
- Osimertinib (Tagrisso) is the NCCN’s preferred category 1 option for first-line therapy in patients with EGFR-mutated advanced NSCLC.3 Findings from the FLAURA trial (NCT02296125) demonstrated survival improvements, leading to osimertinib’s position as a preferred regimen.4
- After treatment with osimertinib, repeat or liquid biopsy is recommended to identify resistance mutations in the EGFR pathway such as EGFR C797X.5
- Findings from DESTINY-Lung02 (NCT04644237) led to the approval of fam-trastuzumab deruxtecan-nxki (Enhertu) as a treatment option for patients with HER2-mutated disease refractory to standard management.6
a This content was generated in tandem with artificial intelligence.
REFERENCES
Hess LM, Krein PM, Haldane D, Han Y, Sireci AN. Biomarker testing for patients with advanced/ metastatic nonsquamous NSCLC in the United States of America, 2015 to 2021. JTO Clin Res Rep. 2022;3(6):100336. doi:10.1016/j.jtocrr.2022.100336
2. Sadik H, Pritchard D, Keeling DM, et al. Impact of clinical practice gaps on the implementation of personalized medicine in advanced non-small-cell lung cancer. JCO Precis Oncol. 2022;6:e2200246. doi:10.1200/PO.22.00246
3. NCCN. Clinical Practice Guidelines in Oncology. Non-small cell lung cancer, version 3.2023. Accessed September 5, 2023. https://bit.ly/3P4vyU6
4. Ramalingam SS, Vansteenkiste J, Planchard D, et al; FLAURA Investigators. Overall survival with osimertinib in untreated, EGFR-mutated advanced NSCLC. N Engl J Med. 2020;382(1):41-50. doi:10.1056/NEJMoa1913662
5. Ramalingam SS, Zhang N, Yu J, et al. Real-world landscape of EGFR C797X mutation as a resistance mechanism to osimertinib in NSCLC. Presented at: International Association for the Study of Lung Cancer 2022 World Conference on Lung Cancer; August 6-9, 2022; Vienna, Austria. Abstract OA03.05.
6. FDA grants accelerated approval to fam-trastuzumab deruxtecan-nxki for HER2-mutant non-small cell lung cancer. FDA. August 16, 2022. Accessed September 5, 2023. https://bit.ly/3Eshzm2

Overcoming Barriers in Cancer Clinical Trials: A Path Forward for Better Patient Care
April 29th 2024Clinical trials play a pivotal role in developing effective therapies, yet their integration is challenged by issues such as insufficient reimbursement structures, misaligned incentives, physician burnout, and a complex regulatory environment.
Read More
Gholam Analyzes Treatment Outcomes for Advanced HCC in Child-Pugh B Population
April 28th 2024During a live Community Case Forum event in partnership with the Tennessee Oncology Practice Society, Pierre Gholam, MD, examined the current state of treatment for patients with hepatocellular carcinoma, looking in particular at what data is available for those with Child-Pugh B and C status who have poorer outcomes and have limited data from prospective clinical trials.
Read More