Moving the Bar in Pancreatic Cancer
Recent advancements in systemic therapies are changing the treatment landscape for patients with pancreatic cancer. New therapeutics and treatment strategies are emerging, meaning more patients than ever are candidates for curative procedures.
John L. Marshall, MD
Recent advancements in systemic therapies are changing the treatment landscape for patients with pancreatic cancer. New therapeutics and treatment strategies are emerging, meaning more patients than ever are candidates for curative procedures.
Pancreatic cancer is notoriously difficult to treat, and it involves multidisciplinary consultation with a variety of clinicians involved in multiple aspects of care. As new options emerge, there will be more opportunities to tailor care to the specific needs of each patient at each stage of treatment. Most recently, new systemic methodsincluding combination therapies, immunotherapies, and neoadjuvant approaches—are showing promise in treating this historically complicated disease.
According to George Kim, MD, of Orange Park Medical Center in Orange Park, Florida, there is “a lot of reason for optimism in the treatment of pancreatic cancer.”
Adjuvant Therapy
Surgical resection is the only curative technique available for patients with pancreatic cancer, but the chances of disease-free survival (DFS) are increased when surgery is followed with adjuvant treatment. Because of the high rates of recurrence associated with this disease, adjuvant therapy is required in all cases of surgically resected pancreat-ic cancer.
According to National Comprehensive Cancer Network (NCCN) guidelines, there is no defined standard for the administration of adjuvant therapy to patients with pancreatic cancer.1NCCN-recommended therapeutic options for adjuvant treatment include gemcitabine monotherapy, 5-fluorouracil (5-FU)/leucovorin, gemcitabine/capecitabine, continuous-infusion 5-FU, capecitabine monotherapy, and induction chemotherapy followed by chemoradiation or chemoradiation and subsequent chemotherapy.1But recent advancements have been made for adjuvant therapies for pancreatic cancer (TABLE 1).
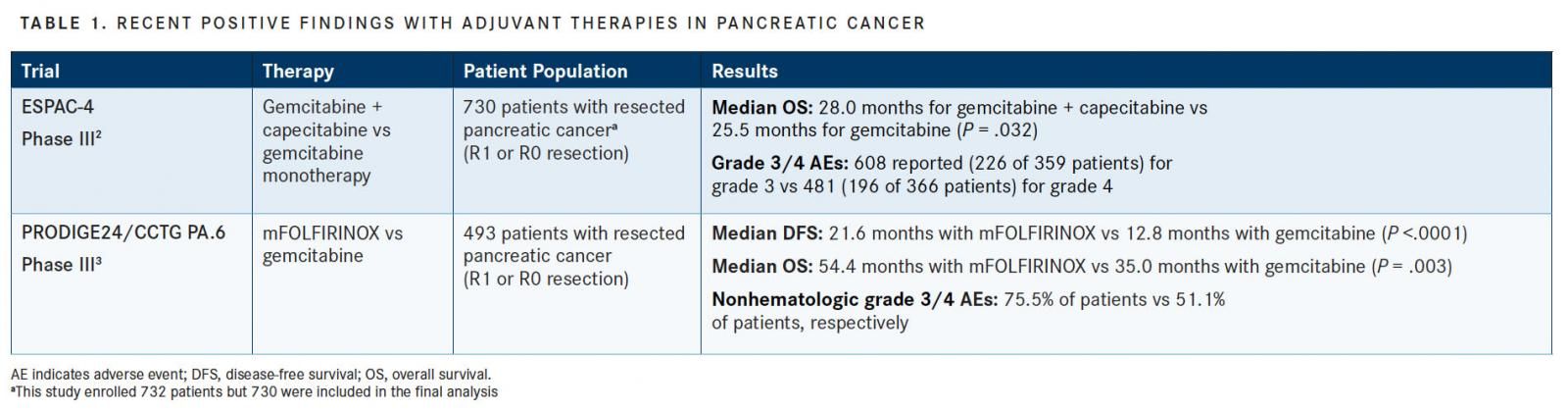
The recent open-label, multicenter, randomized phase III ESPAC-4 trial evaluated the efficacy of gemcitabine plus capecitabine versus gemcitabine monotherapy for the treatment of resected pancreatic cancer.2A total of 732 patients were treated across 92 hospitals, and the median overall survival (OS) of patients receiving gemcitabine plus capecitabine was 28.0 months, compared with 25.5 months for those receiving gemcitabine monotherapy (HR, 0.82; 95% CI, 0.68-0.98; P = .032). Adverse events (AEs) were experienced by 63% of the patients receiving the combination therapy, compared with 54% of the patients receiving monotherapy.2
Results from the PRODIGE24/CCTG PA.6 phase III trial were recently presented at the 2018 American Society of Clinical Oncology (ASCO) Annual Meeting.3The goal of this study was to evaluate adjuvant treatment with gemcitabine versus mFOLFIRINOX, a modified regimen of folinic acid, 5-FU, irinotecan, and oxaliplatin, in patients with resected pancreatic cancer. Treatment with mFOLFIRINOX facilitated greater median DFS (21.6 months with mFOLFIRINOX vs 12.8 months with gemcitabine; stratified HR, 0.58; 95% CI, 0.46-0.73;P<.0001), and the median OS was increased by nearly 20 months with mFOLFIRINOX treatment (54.4 vs 35.0 months; HR, 0.64; 95% CI, 0.48-0.86;P= .003).3
ASCO’s chief medical officer, Richard L. Schilsky, MD, remarked during the 2018 ASCO Annual Meeting, “This is probably the biggest advance we’ve seen in pancreatic cancer since the induction of gemcitabine.… This is a huge step forward, at least for those patients who are able to undergo surgical resection.”
Nonhematologic grade 3/4 AEs were observed with greater frequency in the mFOLFIRINOX arm (75.5% vs 51.1% for mFOLFIRINOX and gemcitabine, respectively) and included diarrhea, sensory peripheral neuropathy, fatigue, vomiting, and mucosis.3Because of the rigor of this treatment program, only patients with good performance status should be considered for it, investigators have said.
“FOLFIRINOX was more toxic than gemcitabine, but it’s still a safe regimen with manageable toxicities,” said lead author Thierry Conroy, MD, the director of the Institut de Cancérologie de Lorraine in Vandoeuvre-lès-Nancy, France, during a presentation at the ASCO Annual Meeting. “The FOLFIRINOX regimen should be considered the new standard of care after pancreatic cancer resection in patients with good performance status.”
Neoadjuvant Therapy
Because there are few disease-specific symptoms or early-warning signs associated with pancreatic cancer, more than 80% of patients present with tumors that are unresectable.4In cases that are borderline resectable and have a high risk of R1 resection, systemic treatment in the neoadjuvant setting is proving to be an effective strategy.
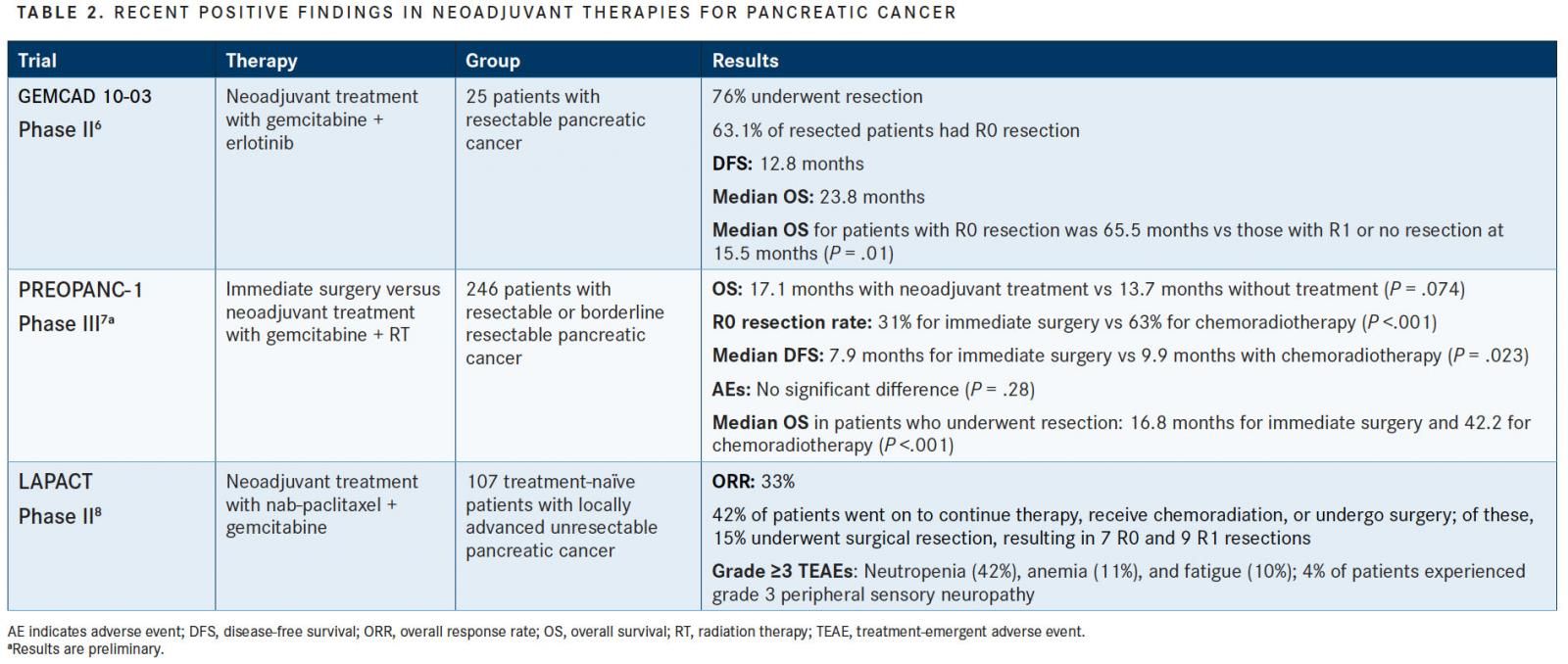
The goal of new research studies evaluating neoadjuvant treatment (TABLE 2) is to increase the number of patients who are candidates for surgical resection. Neoadjuvant treatment also has the potential to treat micrometastases earlier, and re-evaluation following neoadjuvant therapy can be helpful in identifying those that have progressive disease, thus preventing patients from undergoing unnecessary surgery.5
Neoadjuvant treatment is emerging as a tumor-shrinking strategy that can facilitate downstaging, and it should be considered either in borderline resectable cases or for resectable diseases that contain high-risk features. Patients, particularly those in borderline cases, should be monitored by CT imaging in the event that tumor shrinkage alters their resectability status.1
The application of neoadjuvant therapy in the treatment of pancreatic cancer is an active area of research, and evidence to support its efficacy is building. John L. Marshall, MD, the chief of the Division of Hematology/Oncology at MedStar Georgetown University Hospital in Washington, DC, said in an interview, “The fact that we have new medicines that can shrink the cancer and possibly convert [patients] to resectable is a new area for us.”
Recently, the phase II GEMCAD 10-03 trial evaluated the outcomes of patients who were given neoadjuvant treatment of gemcitabine plus erlotinib (Tarceva).6Patients were treated with 3 cycles of gemcitabine plus daily treatment with erlotinib, and if disease did not progress, the patient received 5 more weeks of gemcitabine plus erlotinib plus radiotherapy. Of the 25 patients enrolled in the study, 76% were able to undergo resection, and 63.1% of the resected patients experienced an R0 resection. The median OS was 23.8 months, and it was significantly longer in patients with R0 resection compared with those with an R1 or no resection (65.5 vs 15.5 months, respectively;P= .01).6
Initial results from the PREOPANC-1 phase III study were reported at the 2018 ASCO Annual Meeting, and they revealed promising statistics in favor of preoperative treatment with chemotherapy and radiation in patients with resectable or borderline resectable pancreatic cancer.7The trial is comparing the treatment of these patients with either immediate surgery (arm A) or preoperative chemoradiotherapy (arm B; 2.4 Gy + gemcitabine 1000 mg/m2 on days 1, 8, and 15, followed by a cycle of gemcitabine), both of which were followed by adjuvant chemotherapy.7
Of the 246 participants, the R0 resection rate (31% for immediate surgery vs 63% for neoadjuvant chemoradiotherapy;P<.001) and DFS (median 7.9 vs 9.9 months, respectively; HR, 0.71;P= .023) were improved with neoadjuvant treatment, with no difference in AEs. Disease progression was seen in 80% of patients who immediately underwent resection, but only in 50% of those who received neoadjuvant therapy (P= .002). OS was improved with neoadjuvant treatment, but this difference was not significant at the time of data cutoff (17.1 months with neoadjuvant treatment vs 13.7 months without; HR, 0.74;P= .074).7
The data are preliminary and the study is ongoing, but the lead study author, Geertjan van Tienhoven, MD, PhD, the principal investigator with the Dutch Pancreatic Cancer Group, said, “These preliminary results…suggest a benefit of preoperative chemoradiotherapy over immediate surgery followed by adjuvant chemotherapy.”7
The 2018 LAPACT phase II trial evaluated the combination of nab-paclitaxel (Abraxane) plus gemcitabine in patients with unresectable pancreatic cancer. Of the 107 patients who were treated, the overall response rate (ORR) was 33%, and 42% of patients went on to continue therapy, receive chemoradiation, or undergo surgery. Of these, 15% underwent surgical resection, resulting in 7 R0 and 9 R1 resections.8
Patients who have received neoadjuvant therapy can also be candidates for subsequent adjuvant therapy, although few data are evaluating this situation. NCCN guidelines recommend multidisciplinary review to determine the employment of adjuvant therapy in these cases, but in general, adjuvant chemotherapy or chemoradiation should be considered only for patients who have recovered from surgery and have no evidence of recurrence or metastasis.1
Immunotherapy
Immunotherapies are emerging as powerful treatment options for a variety of cancer types. Phase I trials measuring the efficacy of single-agent antiPD-1 receptor and anti–PD-L1 immune checkpoint inhibitors yielded discouraging results, with ORRs of 0% for patients with advanced pancreatic cancer.9Similar results were seen in a trial for cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) inhibitors in patients with metastatic pancreatic cancer.1“It’s been a complete disappointment,” Kim said.
Despite this, checkpoint inhibitors have been proven to be effective against other tumor types and exhibit low levels of toxicity. These features make them attractive for use in combination therapies with other systemic agents that are effective in treating pancreatic cancer. In addition, there is building preclinical evidence that indicates the potential efficacy of immunotherapy-plus-systemic-therapy combinations.9These therapies are being explored in more than 35 ongoing and 12 prospective clinical trials.9
This past summer, investigators from Penn Medicine’s Abramson Cancer Center shed new light on the heterogeneous responses that pancreatic tumors exhibit when treated with immunotherapy. The team, led by Ben Stanger, MD, PhD, hypothesized that differences in the tumor microenvironment dictate how a tumor responds to therapy; it sought to identify these differences in a mouse model of pancreatic cancer.10The team generated a cell line that, when injected into a mouse, will generate tumors that simulate the variable degree of T-cell infiltration seen in the tumors of patients with pancreatic cancersome have high levels of T cells, and some have very low levels. Using this model, the study revealed that tumors with low T-cell levels did not respond to immune blockade therapy, but half the tumors with high T-cell levels did respond.10
The team also found that tumors with low T-cell levels express CXCL1, which is a chemokine that prevents T-cell infiltration into the tumor microenvironment, ultimately blocking the activity of immunotherapies. The investigators found that if CXCL1 is knocked out in these tumors with low T cells, they can no longer prevent T-cell invasion and will therefore become susceptible to immunotherapies.10,11
In addition, an ongoing phase II trial is evaluating the combination of gemcitabine plus nab-paclitaxel, durvalumab (a PD-L1 inhibitor; Imfinzi), and tremelimumab (a CTLA-4 inhibitor) (NCT02879318). The median OS has not been reached, but 8 of 11 patients in the study were reported to have a partial response to the therapy with a median duration of 7.4 months. Grade ≥2 AEs included hypoalbuminemia, anemia, abnormal lipase levels, fatigue, abnormal white blood cell counts, and hyponatremia.12A phase III trial evaluating this combination therapy is also planned.
Based on the finding that cancers with mismatch repair deficiency (dMMR) are more susceptible to immune checkpoint blockade, ASCO now recommends pembrolizumab (Keytruda), a PD-1 inhibitor, as a second-line therapy for the treatment of patients who test positive for dMMR.13,14
Systemic Therapy for Locally Advanced and Metastatic Pancreatic Cancer
New developments have also been made in the systemic treatment of locally advanced and metastatic disease. Gemcitabine, once the only drug available for pancreatic cancer treatment, is being integrated into successful combination therapies.1
The randomized phase II SCALOP trial evaluated the combination of gemcitabine or capecitabine with radiation to treat patients with locally advanced pancreatic cancer. Patients treated with capecitabine and radiation had improved median OS (17.6 months), compared with those who were treated with gemcitabine and radiation (14.6 months; HR, 0.68; 95% CI, 0.38-1.21;P= .185).15
A 2016 phase II trial evaluated the efficacy of S-1, an oral fluoropyrimidine derivative.16 S-1 has been approved in Japan for the treatment of several cancer types that include pancreatic, gastric, and colorectal cancers. Compared with gemcitabine, S-1 was shown to provide better 5-year survival (24.4% for the gemcitabine group vs 44.1% for the S-1 group) and was also accompanied by fewer AEs, although higher rates of stomatitis and diarrhea were observed with the investigational agent.16 S-1 is actively being investigated in Japanese clinical trials.
Another recent phase III trial in 214 patients with advanced disease in South Korea found that the combination of gemcitabine plus capecitabine exhibited a trend toward improved median OS compared with treatment with gemcitabine alone, but this difference was not significant (10.3 vs 7.5 months;P= .06).17
BRCAmutStatus
It is well established that mutations within theBRCAgene predispose patients to several cancers, including pancreatic cancer. Recent studies indicate that patients withBRCAmutations may be particularly sensitive to platinum-based therapeutics. In a retrospective study of patients with metastatic pancreatic cancer, survival for patients treated with platinum chemotherapy, who also had a family history of breast, ovarian, or pancreatic cancer, was also strongly associated with the number of relatives with aBRCA-related malignancy (test of trend P = .009).18A 2016 phase II trial showed that adjuvant treatment with gemcitabine/cisplatin facilitated a median OS of 35.5 months.19
In addition, the FDA recently granted orphan drug designation to olaparib, a PARP inhibitor, for the treatment of patients with pancreatic cancer. This approval was based on data from the ongoing phase III POLO trial (NCT02184195), which is evaluating the efficacy of olaparib versus placebo as maintenance therapy for patients with BRCA mutations and metastatic cancer that did not progress on first-line platinum therapy. A total of 145 patients are enrolled in this trial.
Molecular profiling is becoming increasingly accessible, and testing for dMMR and microsatellite instability is recommended by the ASCO guidelines.14This type of technology provides patients and clinicians with insight into the details of the patient’s gene expression, and it can present opportunities to make treatment decisions that are tailored to the patient’s specific tumor profile.
These recent advancements have expanded the treatment options for pancreatic cancer, a disease once considered to be incurable. New therapeutics and treatment strategies will continue to increase the number of patients who respond well to treatment. As Marshall has said, “This is nice progress. We, of course, are frustrated. We want more with pancreatic cancer. We want more medicines. We want better outcomes. We want earlier detection if we [can] get it. But to go from where we were to where we are is true progress.”
References:
- Tempero MA, Malafa MP, Al-Hawary M, et al. NCCN Clinical Practice Guidelines in Oncology. Pancreatic Adenocarcinoma. Version 2.2018 https://www.nccn.org/professionals/ physician_gls/pdf/pancreatic.pdf. Published July 10, 2018. Accessed October 16, 2018.
- Neoptolemos JP, Palmer DH, Ghaneh P, et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): a multicentre, open-label, randomised, phase 3 trial. Lancet. 2017;389(10073):1011- 1024. doi: 10.1016/S0140-6736(16)32409-6.
- Conroy T, Hammel P, Hebbar M, et al. Unicancer GI PRODIGE 24/CCTG PA.6 trial: a multicenter international randomized phase III trial of adjuvant mFOLFIRINOX versus gemcitabine (gem) in patients with resected pancreatic ductal adenocarcinomas. J Clin Oncol. 2018;36(suppl; abstr LBA4001). meetinglibrary.asco.org/record/159164/abstract.
- Li D, Xie K, Wolff R, Abbruzzese JL. Pancreatic cancer. Lancet. 2004;363(9414):1049- 1057. doi: 10.1016/S0140-6736(04)15841-8.
- Breslin TM, Hess KR, Harbison DB, et al. Neoadjuvant chemoradiotherapy for adenocarcinoma of the pancreas: treatment variables and survival duration. Ann Surg Oncol. 2001;8(2):123-132.
- Maurel J, Sánchez-Cabús S, Laquente B, et al. Outcomes after neoadjuvant treatment with gemcitabine and erlotinib followed by gemcitabine-erlotinib and radiotherapy for resectable pancreatic cancer (GEMCAD 10-03 trial) [Published online September 17, 2018]. Cancer Chemother Pharmacol. 2018. doi: 10.1007/s00280-018-3682-9.
- Tienhoven GV, Versteijne E, Suker M, et al. Preoperative chemoradiotherapy versus immediate surgery for resectable and borderline resectable pancreatic cancer (PREOPANC-1): a randomized, controlled, multicenter phase III trial. J Clin Oncol. 2018;36(suppl; abstr LBA4002). meetinglibrary.asco.org/record/160063/abstract.
- Hammel P, Lacy J, Portales F, et al. Phase II LAPACT trial of nab-paclitaxel (nab-P) plus gemcitabine (G) for patients with locally advanced pancreatic cancer (LAPC). J Clin Oncol. 2018;36(suppl; abstr 204). meetinglibrary.asco.org/record/155730/abstract.
- Gong J, Hendifar A, Tuli R, et al. Combination systemic therapies with immune checkpoint inhibitors in pancreatic cancer: overcoming resistance to single-agent checkpoint blockade. Clin Transl Med. 2018;7(1):32. doi: 10.1186/s40169-018-0210-9.
- Penn study reveals secrets of “hot” and “cold” pancreatic cancer tumors [news release]. Philadelphia, PA: Penn Medicine News; June 27, 2018. www.pennmedicine.org/news/ news-releases/2018/june/penn-study-reveals-secrets-of-hot-and-cold-pancreatic-cancer-tumors. Accessed October 16, 2018.
- Li J, Byrne KT, Yan F, et al. Tumor cell-intrinsic factors underlie heterogeneity of immune cell infiltration and response to immunotherapy. Immunity. 2018;49(1):178-193.e7. doi: 10.1016/j.immuni.2018.06.006.
- Renouf DJ, Dhani NC, Kavan P, et al. The Canadian Cancer Trials Group PA.7 trial: results from the safety run in of a randomized phase II study of gemcitabine (GEM) and nab-paclitaxel (nab-P) versus GEM, nab-P, durvalumab (D), and tremelimumab (T) as first-line therapy in metastatic pancreatic ductal adenocarcinoma (mPDAC). J Clin Oncol. 2018;36(suppl; abstr 349). meetinglibrary.asco.org/record/156003/abstract.
- Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409-413. doi: 10.1126/science. aan6733.
- Sohal DPS, Kennedy EB, Khorana A, et al. Metastatic pancreatic cancer: ASCO clinical practice guideline update. J Clin Oncol. 2018;36(24):2545-2556. doi: 10.1200/ JCO.2018.78.9636.
- Hurt CN, Falk S, Crosby T, et al. Long-term results and recurrence patterns from SCALOP: a phase II randomised trial of gemcitabine- or capecitabine-based chemoradiation for locally advanced pancreatic cancer. Br J Cancer. 2017;116(10):1264-1270. doi: 10.1038/ bjc.2017.95.
- Uesaka K, Boku N, Fukutomi A, et al. Adjuvant chemotherapy of S-1 versus gemcitabine for resected pancreatic cancer: a phase 3, open-label, randomised, non-inferiority trial (JASPAC 01). Lancet. 2016;388(10041):248-257. doi: 10.1016/S0140-6736(16)30583-9.
- Lee HS, Chung MJ, Park JY, et al. A randomized, multicenter, phase III study of gemcitabine combined with capecitabine versus gemcitabine alone as first-line chemotherapy for advanced pancreatic cancer in South Korea. Medicine (Baltimore). 2017;96(1):e5702. doi: 10.1097/MD.0000000000005702.
- Fogelman D, Sugar EA, Oliver G, et al. Family history as a marker of platinum sensitivity in pancreatic adenocarcinoma. Cancer Chemother Pharmacol. 2015;76(3):489-498. doi: 10.1007/s00280-015-2788-6.
- Postlewait LM, Ethun CG, Kooby DA, et al. Combination gemcitabine/cisplatin therapy and ERCC1 expression for resected pancreatic adenocarcinoma: results of a phase II prospective trial. J Surg Oncol. 2016;114(3):336-341. doi: 10.1002/jso.24317.
- US FDA grants Lynparza orphan drug designation for pancreatic cancer [news release]. Kenilworth, NJ: AstraZeneca; October 16, 2018
The Impact of the Gut Microbiome in Young Patients With Colorectal Cancer
February 15th 2021In season 2, episode 2 of Targeted Talks, Cathy Eng, MD, speaks with Benjamin Weinberg, MD, about the gut microbiome, and how the presence of certain microbiota impact the onset and intensity of disease as well as the potential response to certain treatments.
Listen
Novel Approaches Focus on Limiting Toxicity in Older Patients With ALL
April 22nd 2024The major challenges for clinicians treating older patients with acute lymphoblastic leukemia surround the emergence of resistance to existing therapies and the toxicities associated with current chemotherapies.
Read More