Recent Studies Point to a Greater Role for PARP Inhibitors in Solid Tumors
The early development of PARP inhibitors in 2003 focused on their use in combination with cytotoxic chemotherapy agents, but this was eventually abandoned because of excess toxicity.
Timothy A. Yap, MBBS, PhD, MRCP(UK), BSc(Hons)
The early development of PARP inhibitors in 2003 focused on their use in combination with cytotoxic chemotherapy agents, but this was eventually abandoned because of excess toxicity. Approximately 2 years later, data emerged showing synthetic lethality between PARP inhibitors andBRCAmutations in ovarian cancer, leading the way to treatment ofBRCA-mutated tumors in other cancer types.1,2Investigators have since determined that PARP inhibitors are active across a range of tumor types harboringBRCAmutations and can also evoke responses in subsets ofBRCA-negative cancers.1
According to Timothy A. Yap, MBBS, PhD, MRCP(UK), BSc(Hons), associate professor in the Department of Investigational Cancer Therapeutics and associate director of translational research at the Khalifa Institute for Personalized Cancer Therapy, both at The University of Texas MD Anderson Cancer Center in Houston, PARP inhibitors show synthetic lethality when combined with germline abnormalities in many DNA damage response (DDR) pathway components, such as mutations in theATM, ATR, BRCA1/2, CHK1, CHK2, PALB2,andRAD51genes. Some of these mutations may eventually be shown to “predict robustly for antitumor response,” he said.
Yap, who is also medical director of the Institute for Applied Cancer Science at MD Anderson, is investigating whether PARP inhibitors demonstrate similar synthetic lethality when combined with other targeted DDR therapies or different classes of anticancer drugs. He said he is particularly excited about efforts to combine PARP inhibitors “with immunotherapies such as PD-1 or PD-L1 inhibitors, other DDR agents such as ATR inhibitors, and other molecularly targeted agents that may be used to induce a state of chemical ‘BRCAness’ in HR [homologous recombination]proficient cancers, such as antiangiogenic agents.” Yap said studies have already provided “promising early results with rational combinations and responses beyondBRCA1/2-mutated tumors.”
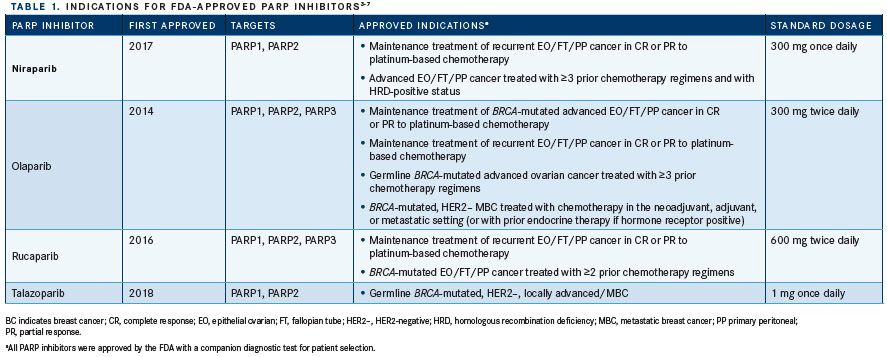
The FDA has approved 4 PARP inhibitors as single-agent therapies for breast or ovarian cancer (TABLE 1): olaparib (Lynparza), rucaparib (Rubraca), niraparib (Zejula), and talazoparib (Talzenna).3-7PARP inhibitors are a family of enzymes essential for many cellular processes in addition to DNA repair, including chromatin remodeling, transcription, messenger RNA processing, and replication fork stabilization.2,7,8PARP inhibitors primarily target the PARP1 and PARP2 isoforms, which detect and promote repair of DNA single- or double-strand breaks (DSBs) to maintain genomic stability.7,8Tumors harboring DDR pathway aberrations that prevent homologous recombination repair of DSBs depend on PARP to repair single-strand DNA breaks and prevent them from progressing to irreparable, synthetically lethal DSBs.9
PARP INHIBITION IN PROSTATE CANCER
Investigators are still elucidating the complex mechanisms of PARP inhibitors, but, in addition to inhibiting PARP catalytic activity, these agents are known to trap PARP at sites of damaged DNA. The resulting PARP-DNA complexes are highly efficient at preventing DNA repair, resulting in greater accumulation of DSBs; replication fork collapse; and, ultimately, cell death.1,7“The antitumor efficacy of the PARP-trapping PARP inhibitors is probably not dissimilar,” Yap said. Veliparib, an investigational PARP inhibitor in late-stage clinical trial development, appears to be a weak PARP trapper, which may explain its less robust single-agent activity in preclinical trials.10Yap noted that PARP inhibitors mainly differ in their toxicities: “For example, alopecia with talazoparib, transient elevations in liver transaminases with rucaparib, and hypertension with niraparib.”
Single-Agent Studies
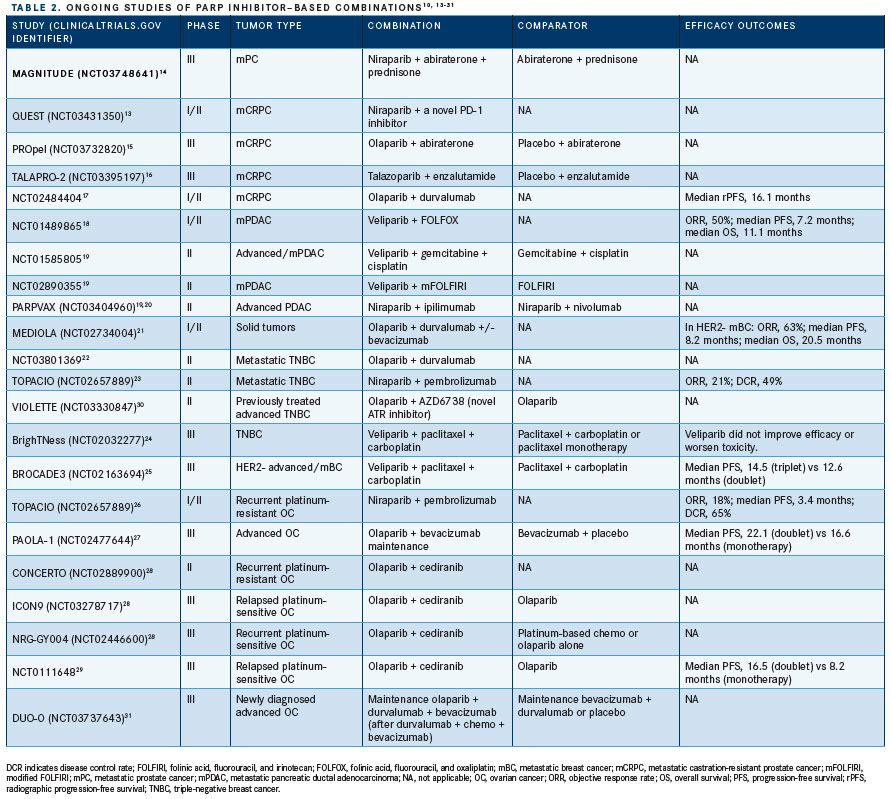
Despite recent treatment advances, metastatic castration-resistant prostate cancer (mCRPC) remains incurable.11An estimated 20% to 25% of men with metastatic prostate cancer harbor a homologous recombination pathway mutation,10 and 1 study found that 11% of prostate cancer specimens had pathogenicBRCA1/2 mutations.12Homologous recombination deficiency (HRD) mutations are associated with more aggressive disease and worse prognosis.13 Ongoing clinical trials are exploring PARP inhibitors as monotherapy or in combination with other therapies for prostate cancer (TABLE 2).10,13-31
Several trials have evaluated single-agent olaparib in mCRPC featuring a DDR alteration, and findings suggest that olaparib is more active in patients withBRCA1/2mutations than in those with mutations in other DDR genes, such asATM.32The open-label phase III PROfound trial (NCT02987543) enrolled 245 patients withBRCA1/2orATMmutations (cohort A) and 142 with other DDR mutations (cohort B) whose dis- ease had progressed after abiraterone acetate (Zytiga) or enzalutamide (Xtandi).33 Patients were randomly assigned 2:1 to olaparib or physician’s choice of enzalutamide or abiraterone.33
Preliminary data presented at the European Society for Medical Oncology (ESMO) 2019 Congress associated olaparib with significant improvement in 12-month progression-free survival (PFS) compared with treatment of physician’s choice (TPC) in cohort A (7.39 vs 3.55 months, respectively; HR, 0.34; 95% CI, 0.25-0.47;P<.0001).33In cohort A, olaparib also produced significant improvements in objective response rate (ORR) and median time to pain progression. Although the overall survival (OS) data for cohort A showed a favorable trend, the difference did not achieve a predefined boundary for statistical significance, possibly because of the high crossover rate from the TPC arm.33Olaparib did not significantly improve outcomes in cohort B.34The most common treatment-related adverse events (TRAEs) in the olaparib cohort were anemia (46%), nausea (41%), decreased appetite (30%), and fatigue (26%).33
Data from the phase II TOPARP-B trial (NCT01682772) presented at the 2019 American Society of Clinical Oncology (ASCO) Annual Meeting confirmed the activity of olaparib in progressive mCRPC harboring a DDR mutation.35Patients were randomly assigned to 300 or 400 mg of olaparib twice daily. The overall response rate in the 400-mg group was 54% (95% CI, 39%-69%) compared with 37% (95% CI, 23%-53%) in the 300-mg group.35The highest ORRs occurred in patients withBRCA1/2orPALB2mutations (80% and 57%, respectively). Overall median PFS was 5.4 months.35
The ongoing phase II open-label Galahad trial (NCT02854436) is assessing niraparib in patients with previously treated mCRPC and a DDR mutation, approximately half of whom have aBRCA1/2mutation.36An interim analysis showed that pronounced clinical activity was largely confined to theBRCA1/2cohort, which had an ORR of 41%, a composite response rate of 63%, and median radiographic PFS (rPFS) of 8.2 months.36Common grade 3/4 TRAEs were anemia (29%), thrombo- cytopenia (15%), and neutropenia (7%).
PARP InhibitorBased Combinations
In 2018, rucaparib received breakthrough therapy designation forBRCA1/2-mutated mCRPC based on promising efficacy in the phase II TRITON2 trial (NCT02952534).37Updated data presented at ESMO 2019 Congress confirmed rucaparib’s activity.38Among patients with aBRCA1/2mutation, the prostate-specific antigen response rate was 52% and the ORR was 44%, with a 5% complete response (CR) rate.38Responses were durable, persisting ≥24 weeks in 60% of patients. Common grade ≥3 TRAEs included anemia (11%) and asthenia/ fatigue (7%).38The phase III TRITON3 trial (NCT02975934) will compare rucaparib with TPC in progressive mCRPC with aBRCA1/2orATMmutation.38,39
PARP INHIBITION IN PANCREATIC CANCER
The phase III TALAPRO-2 trial (NCT03395197) is evaluating talazoparib with enzalutamide in unselected patients with mCRPC.16Preliminary data indicated promising activity and a manageable safety profile. Interim results from an ongoing phase I/II study of olaparib plus the PD-L1 inhibitor durvalumab (Imfinzi) in mCRPC also showed promising activity, particularly in patients with a DDR alteration.17Other combination trials are QUEST (NCT03431350), which is evaluating niraparib plus a novel antiPD-1 monoclonal antibody13; MAGNITUDE (NCT03748641), which combines niraparib with abiraterone and prednisone14; and PROPEL (NCT03732820), which is investigating first-line therapy with olaparib plus abiraterone.15
Single-Agent Studies
Between 4% and 7% of patients with pancreatic cancer have a germlineBRCA1/2mutation,40 and up to 3% have anATMmutation. Treatment options after progression of advanced pancreatic ductal adenocarcinoma (PDAC) are limited, and the presence of homologous recombination defects provides a rationale for evaluating PARP inhibitors in this disease.41
Interim results from the phase III POLO trial (NCT02184195) suggest that olaparib holds promise as a maintenance therapy for patients with metastatic PDAC who have a germlineBRCA1/2mutation and no progression after platinum-based chemotherapy.40At data cutoff, median PFS was significantly longer in patients randomly assigned to olaparib versus those given placebo (7.4 vs 3.8 months; HR, 0.53; 95% CI, 0.35-0.82;P= .004).40 However, the ORR, median OS, and health-related quality of life were not significantly different between the 2 arms.40Approximately one-fourth of patients treated with olaparib experienced a serious adverse event (AE).
Although the phase II RUCAPANC trial (NCT02042378) of rucaparib monotherapy for previously treated advanced PDAC harboring aBRCA1/2mutation was terminated early because of poor response, investigators noted that the 16% of patients who responded had durable, clinically significant responses.42Efficacy was enhanced in platinum-sensitive patients.42A recent interim analysis of a phase II trial (NCT03140670) of maintenance rucaparib showed greater success in platinum-sensitive patients with advanced PDAC and aBRCA1/2orPALB2mutation.43The ORR for the 19 evaluable patients was 37%, which included 6 partial responses (PRs) and 1 CR. Responses were confined to patients with aBRCA2orPALB2mutation. The disease control rate (DCR) was 90% for ≥8 weeks, and median PFS was 9.1 months.43Common AEs possibly related to treatment were nausea, dysgeusia, and fatigue.
An ongoing phase I dose-escalation trial (NCT01286987) is assessing the single-agent activity of talazoparib in advanced solid tumors that harbor a germlineBRCA1/2mutation.44Preliminary data for 13 patients with pancreatic cancer revealed an ORR of 20% (all PRs) and a clinical benefit rate of 30%.44Just 1 patient remained on talazoparib at data cutoff.
PARP InhibitorBased Combinations
Final data from a phase II trial combining veliparib with FOLFOX (folinic acid, fluorouracil, and oxaliplatin) chemotherapy showed an ORR of 50%, median PFS of 7.2 months, and median OS of 11.1 months in patients with metastatic PDAC and a DDR mutation.18Outcomes were slightly better in platinum-naïve patients with a family history of pancreatic cancer and a DDR mutation.18Additional trials are evaluating veliparib combined with gemcitabine and cisplatin (NCT01585805) and with FOLFIRI (folinic acid, fluorouracil, and irinotecan) chemotherapy (NCT02890355). In the phase II PARPVAX trial (NCT03404960), niraparib is being explored with an immune checkpoint inhibitor (nivolumab [Opdivo] or ipilimumab [Yervoy]) for advanced platinum-sensitive PDAC.19,20Immune checkpoint inhibitors have shown limited activity in pancreatic cancer, prompting investigators to look for synergistic combinations.45
PARP INHIBITION IN BREAST CANCER
Olaparib and talazoparib are the only PARP inhibitors approved for breast cancer.3,6Although the single-agent activity of PARP inhibitors in BRCA1/2-mutated breast cancer is well established, interest in expanding their use to other settings is burgeoning (TABLE 2).30
Single-Agent Studies
Regulatory approval of olaparib for germlineBRCA1/2-mutated, HER2-negative metastatic breast cancer (mBC) was based on results of the randomized phase III OlympiAD trial, showing significant improvement in median PFS with olaparib versus physician’s choice of chemotherapy (7.0 vs 4.2 months, respectively; HR, 0.58; 95% CI, 0.43-0.80;P= .0009).3Follow-up data showed no significant improvement in OS with olaparib, however.46 The randomized phase III OlympiA trial (NCT02032823) is assessing single-agent olaparib as adjuvant therapy in high-riskBRCA1/2-mutated, HER2-negative nonmetastatic breast cancer.47
Talazoparib received FDA approval based on conclusions from the randomized phase III EMBRACA trial, which compared talazoparib with TPC in BRCA1/2-mutated advanced breast cancer.48 Median PFS was significantly longer in the talazoparib arm versus the TPC arm (8.6 vs 5.6 months; HR, 0.54; 95% CI, 0.41-0.71;P<.001), but median OS did not differ significantly between the groups.48Subsequently, a small trial suggested that neoadjuvant talazoparib could reduce tumor volume in early-stage breast cancer, leading to a larger trial (NCT03499353) to investigate the regimen’s efficacy in triple-negative breast cancer (TNBC) with a germlineBRCA1/2mutation.30Talazoparib has also been studied in platinum-sensitive advanced HER2-negative breast cancer with homologous recombination pathway alterations besidesBRCA1/2, producing an ORR of 25% in 12 evaluable patients.49
PARP InhibitorBased Combinations
Although the phase III BRAVO trial assessing single-agent niraparib in locally advanced or metastatic, HER2-negative,BRCA1/2-mutated breast cancer ended prematurely because of poor patient adherence,30ongoing studies continue to evaluate niraparib for breast cancer. The phase II ABC Study (NCT02826512) is testing niraparib in advanced, locally recurrent, BRCA-like, HER2-negative breast cancer, and a phase I trial (NCT03329937) is evaluating neoadjuvant niraparib in a similar population.50
In the MEDIOLA trial (NCT02734004), olaparib was administered with durvalumab to women with germlineBRCA1/2-mutated HER2-negative mBC.21The ORR was 63%, median PFS was 8.2 months, and median OS was 20.5 months. The same combination is being studied in another phase II trial (NCT03801369) but for metastatic BRCA wild-type TNBC.22The ongoing VIOLETTE trial (NCT03330847) is combining olaparib with an investigational ATR inhibitor in previously treated advanced TNBC.30
The phase II TOPACIO/KEYNOTE-162 trial (NCT02657889) investigated the combination of niraparib plus the antiPD-1 monoclonal antibody pembrolizumab (Keytruda).23The ORR was 21%, which included 5 CRs and 5 PRs, and the DCR was 49%.23Activity was stronger inBRCA1/2- mutated tumors, producing an ORR of 47% and a DCR of 80%.23Common TRAEs were anemia (18%), thrombocytopenia (15%), and fatigue (7%); 15% of patients experienced immune-related AES.23
PARP INHIBITION IN OVARIAN CANCER
Veliparib combinations have been evaluated in multiple trials. The phase III BrighTNess trial (NCT02032277) randomly assigned patients with TNBC to receive paclitaxel alone; paclitaxel plus carboplatin; or paclitaxel, carboplatin, and veliparib.24Veliparib did not enhance the efficacy of paclitaxel/carboplatin, nor did it substantially increase toxicity.24The BROCADE3 trial (NCT02163694) assessed the same triplet regimen but in patients with HER2-negative, advanced or metastatic, germlineBRCA1/2-mutated breast cancer.25This time, adding veliparib to carboplatin and paclitaxel significantly prolonged PFS versus carboplatin and paclitaxel alone (14.5 vs 12.6 months, respectively; HR, 0.71; 95% CI, 0.57-0.88;P= .002) but failed to significantly improve OS or ORR.25The 3-year PFS rate was more than doubled in the veliparib arm.25
Single-Agent Studies
Olaparib, rucaparib, and niraparib are all indicated for ovarian, fallopian tube, and primary peritoneal cancers.3-5Niraparib was first approved as maintenance therapy for recurrent platinum-sensitive disease and received an expanded indication in October 2019 for advanced disease.51
Data were presented at the 2019 ASCO meeting from the randomized phase III SOLO3 trial (NCT02282020), which compared single-agent olaparib with TPC in germline BRCA1/2-mutated platinum-sensitive relapsed ovarian cancer.52Outcomes significantly favored olaparib over TPC, with ORRs of 72% vs 51% (HR, 2.53; 95% CI, 1.40-4.58;P= .002) and median PFS of 13.4 vs 9.2 months (HR, 0.62; 95% CI, 0.43- 0.91;P= .013).52SOLO1 (NCT01844986) is an earlier trial of maintenance olaparib in newly diagnosed advanced ovarian cancer with a confirmedBRCA1/2mutation, and which showed a 70% reduction in disease progression or death versus placebo (HR, 0.30; 95% CI, 0.23-0.41;P<.001).53
PARP InhibitorBased Combinations
Approval of niraparib for advanced ovarian cancer in patients with HRD was based on results of QUADRA, a single-arm phase II trial (NCT02354586) in heavily pretreated patients.54The ORR in a subset of patients with HRD and platinum sensitivity was 28% (95% CI, 15.6%-42.6%; 1-sidedP= .00053), and the DCR was 69%.54The most common grade ≥3 TRAEs were small intestinal obstruction (7%), thrombocytopenia (7%), and vomiting (6%). The phase III PRIMA study (NCT02655016) compared niraparib with placebo in newly diagnosed, advanced, platinum-sensitive ovarian cancer regardless of BRCA mutation status.55 Niraparib was associated with significantly longer PFS than placebo overall and in a subset of patients with HRD.55
Combinations featuring niraparib or olaparib are also being investigated in ovarian cancer. TOPACIO/KEYNOTE-162 (NCT02657889) included 62 women with ovarian cancer, regardless of BRCA mutation status, and administered a combination of niraparib and pembrolizumab.26 The ORR was 18% (90% CI, 11%-29%), median PFS was 3.4 months (95% CI, 2.1-5.1 months), and the DCR was 65% (90% CI, 54%-75%), which included 3 CRs and 8 PRs.26Responses occurred even in patients without HRD.
The phase III PAOLA-1/ENGOT-ov25 trial (NCT02477644) assessed the addition of olaparib to bevacizumab (Avastin) maintenance therapy in unselected patients with advanced ovarian cancer who had a stable CR or PR after first-line platinum-based therapy plus bevacizumab. Overall median PFS was significantly longer with the doublet versus bevacizumab monotherapy (22.1 vs 16.6 months, respectively; HR, 0.59; 95% CI, 0.49-0.72;P<.001).27Patient cohorts with aBRCAmutation or HRD achieved even longer PFS.
Several ongoing trials are evaluating olaparib in combination with the antiangiogenic agent cediranib in various ovarian cancer settings (Table).28 Recently published findings from a randomized phase II trial in women with relapsed platinum-sensitive ovarian cancer associated the combination of olaparib plus cediranib with significantly longer median PFS and OS than single-agent olaparib, primarily in patients without a BRCA1/2 mutation.29
Conclusions
Studies continue to reveal the promising antitumor activity of PARP inhibitors beyond breast and ovarian cancers. Beyond the expansion of PARP inhibitors to prostate and pancreatic cancers, investigators are also now evaluating these agents in solid tumors such as lung and gastric cancers, as well as in hematologic malignancies. Combining PARP inhibition with agents that target other DDR pathways or have complementary mechanisms, such as immune checkpoint inhibitors and DNA-damaging chemotherapeutics, may provide the best opportunity to deliver the clinical benefits of PARP inhibition to more patients. In the future, biomarkers will likely play an even greater role in identifying those patients most likely to respond to PARP inhibitors.
References:
- Yap TA, Plummer R, Azad NS, Helleday T. The DNA damaging revolution: PARP inhibitors and beyond.Am Soc Clin Oncol Educ Book.2019:185-195. doi: 10.1200/edbk_238473.
- Morales J, Li L, Fattah FJ, et al. Review of poly (ADP-ribose) polymerase (PARP) mechanisms of action and rationale for targeting in cancer and other diseases.Crit Rev Eukaryot Gene Expr.2014;24:15-28.
- Lynparza [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP, 2019.
- Rubraca [package insert]. Boulder, CO: Clovis Oncology, 2018.
- Zejula [package insert]. Waltham, MA: Tesaro, Inc, 2019.
- Talzenna [package insert]. New York, NY: Pfizer Labs, 2019.
- Murthy P, Muggia F. PARP inhibitors: clinical development, emerging differences, and the current therapeutic issues.Cancer Drug Resist.2019;2:665-679. doi: 10.20517/cdr.2019.002.
- Michelena J, Lezaja A, Teloni F, Schmid T, Imhof R, Altmeyer M. Analysis of PARP inhibitor toxicity by multidimensional fluorescence microscopy reveals mechanisms of sensitivity and resistance.Nature Comm.2018;9(1):267 doi: 10.1038/s41467-018-05031-9.
- Noordermeer SM, van Attikum H. PARP inhibitor resistance: a tug-of-war in BRCA-mutated cells.Trends Cell Biol.2019;29(10):820-834. doi: 10.1016/j.tcb.20107.008.
- Mateo J, Lord CJ, Serra V, et al. A decade of clinical development of PARP inhibitors in perspective.Ann Oncol.2019;30(9):1437-1447. doi: 1093/annonc/mdz192.
- Lang SH, Swift SL, White H, Misso K, Kleijnen J, Quek RGW. A systematic review of the prevalence of DNA damage response gene mutations in prostate cancer.Int J Oncol.2019;55(3):597-616. doi: 10.3892/ijo.2019.4842.
- Agarwal N, Sokol ES, Lara P Jr., et al. Pan-cancer assessment of BRCA1/2 genomic alterations (GAs) by comprehensive genomic profiling (CGP) of tissue and circulating tumor DNA (ctDNA).Ann Oncol.2018;29(suppl 8). doi: 10.1093/annonc/mdy269.049.
- Prostate cancer. OncologyPro website. oncologypro.esmo.org/Oncology-in-Practice/Anti-Cancer-Agents-and-Biological-Therapy/PARP-inhibition-and-DNA-Damage-Response-DDR/PARP-inhibitors/Clinical-Activity/Prostate-Cancer. Updated July 25, 2019. Accessed October 26, 2019.
- A Study of Niraparib in Combination With Abiraterone Acetate and Prednisone Versus Abiraterone Acetate and Prednisone for Treatment of Participants With Metastatic Prostate Cancer (MAGNITUDE). clinicaltrials.gov/ct2/show/NCT03748641. Updated October 17, 2019. Accessed October 30, 2019.
- Clarke NW, Armstrong AJ, Thiery-Vuillemin A, et al. PROPEL: a randomized, phase III trial evaluating the efficacy and safety of olaparib combined with abiraterone as first-line therapy in patients with metastatic castration-resistant prostate cancer (mCRPC).J Clin Oncol.2019;37(suppl 7; abstr TPS340). doi: 10.1200/JCO.2019.37.7_suppl.TPS340.
- Agarwal N, Shore ND, Dunshee C, et al. Clinical and safety outcomes of TALAPRO-2: a two-part phase III study of talazoparib (TALA) in combination with enzalutamide (ENZA) in metastatic castration-resistant prostate cancer (mCRPC).J Clin Oncol.2019;37(suppl 15; abstr 5076). doi: 10.1200/JCO.2019.37.15_suppl.5076.
- Karzai F, VanderWeele D, Madan RA, et al. Activity of durvalumab plus olaparib in metastatic castration-resistant prostate cancer in men with and without DNA damage repair mutations.J Immunother Cancer.2018;6:141. doi: 10.1186/s40425-018-0463-2.
- Pishvaian MJ, Wang H, Parenti S, et al. Final report of a phase I/II study of veliparib (Vel) in combination with 5-FU and oxaliplatin (FOLFOX) in patients (pts) with metastatic pancreatic cancer (mPDAC).J Clin Oncol.2019;37(suppl 15; abstr 4015). doi: 10.1200/JCO.2019.37.15_suppl.4015.
- Pancreatic cancer. OncologyPro website. oncologypro.esmo.org/Oncology-in-Practice/Anti-Cancer-Agents-and-Biological-Therapy/PARP-inhibition-and-DNA-Damage-Response-DDR/PARP-inhibitors/Clinical-Activity/Pancreatic-Cancer. Updated July 25, 20 Accessed October 26, 2019.
- Reiss KA, Mick R, O'Hara MH, et al. A randomized phase II trial of niraparib plus either nivolumab or ipilimumab in patients with advanced pancreatic cancer whose cancer has not progressed on platinum-based therapy.J Clin Oncol.2019;37(suppl 15; abstr TPS4161). doi: 10.1200/JCO.2019.37.15_suppl.TPS4161.
- Domchek S, Postel-Vinay S, Im S-A, et al. Phase II study of olaparib (O) and durvalumab (D) (MEDIOLA): updated results in patients (pts) with germline BRCA-mutated (gBRCAm) metastatic breast cancer (MBC).Ann Oncol.2019;30(suppl 5). doi: 10.1093/annonc/mdz253.017.
- Mitri ZI, Vuky J, Kemmer KA, et al. A phase II trial of olaparib and durvalumab in metastatic BRCA wild type triple-negative breast cancer.J Clin Oncol.2019;37(suppl 15; abstr TPS1111). doi: 10.1200/JCO.2019.37.15_suppl.TPS1111.
- Vinayak S, Tolaney SM, Schwartzberg L, et al. Open-label clinical trial of niraparib combined with pembrolizumab for treatment of advanced or metastatic triple-negative breast cancer.JAMA Oncol.2019;5(8):1132-1140. doi: 10.1001/jamaoncol.2019.1029.
- Loibl S, O’Shaughnessy J, Untch M, et al. Addition of the PARP inhibitor veliparib plus carboplatin or carboplatin alone to standard neoadjuvant chemotherapy in triple-negative breast cancer (BrighTNess): a randomised, phase 3 trial.Lancet Oncol.2018;19(4):497-509. doi: 10.1016/S1470-2045(18)30111-6.
- Diéras VC, Han HS, Kaufman B, et al. Phase III study of veliparib with carboplatin and paclitaxel in HER2-negative advanced/metastatic gBRCA-associated breast cancer.Ann Oncol.2019;30(suppl 5; abstr LBA9). doi: 10.1093/annonc/mdz394.008.
- Konstantinopoulos PA, Waggoner S, Vidal GA, et al. Single-arm phases 1 and 2 trial of niraparib in combination with pembrolizumab in patients with recurrent platinum-resistant ovarian carcinoma.JAMA Oncol.2019;5(8):1141-1149. doi: 10.1001/jamaoncol.2019.1048.
- Ray-Coquard IL, Pautier P, Pignata S, et al. Phase III PAOLA-1/ENGOT-ov25 trial: olaparib plus bevacizumab (bev) as maintenance therapy in patients (pts) with newly diagnosed, advanced ovarian cancer (OC) treated with platinum-based chemotherapy (PCh) plus bev.Ann Oncol.2019;30(suppl 5; abstr LBA_PR). doi: 10.1093/annonc/mdz394.053.
- Boussios S, Karihtala P, Moschetta M, et al. Combined strategies with poly (ADP-ribose) polymerase (PARP) inhibitors for the treatment of ovarian cancer: a literature review.Diagnostics (Basel).2019;9(3):pii:E87. doi: 10.3390/diagnostics9030087.
- Liu JF, Barry WT, Birrer M, et al. Overall survival and updated progression-free survival outcomes in a randomized phase II study of combination cediranib and olaparib versus olaparib in relapsed platinum-sensitive ovarian cancer.Ann Oncol.2019;30:551-557. doi: 10.1093/annonc/mdz018.
- Breast cancer. OncologyPro website. oncologypro.esmo.org/Oncology-in-Practice/Anti-Cancer-Agents-and-Biological-Therapy/PARP-inhibition-and-DNA-Damage-Response-DDR/PARP-inhibitors/Clinical-Activity/Breast-Cancer/Ongoing-Developments. Updated July 25, 2019. Accessed October 26, 2019.
- Harter P, Bidziński M, Colombo N, et al. DUO-O: a randomized phase III trial of durvalumab (durva) in combination with chemotherapy and bevacizumab (bev), followed by maintenance durva, bev and olaparib (olap), in newly diagnosed advanced ovarian cancer patients.J Clin Oncol.2019;37(suppl 15; abstr TPS5598). doi: 10.1200/JCO.2019.37.15_suppl.TPS5598.
- Marshall CH, McNatty A, Eisenberger MA, Bryce AH, Antonarakis ES. Response to PARP inhibitor therapy in metastatic castrate-resistant prostate cancer (mCRPC) patients withBRCA1/2versus ATMmutations. J Clin Oncol.2019;37(suppl 7; abstr 154). doi: 10.1200/JCO.2019.37.7_suppl.154.
- Hussain M, Mateo J, Fizazi K, et al. PROfound: phase III study of olaparib versus enzalutamide or abiraterone for metastatic castration-resistant prostate cancer (mCRPC) with homologous recombination repair (HRR) gene alterations.Ann Oncol.2019;30(suppl 5; abstr LBA12_PR). doi: 10.1093/annonc/mdz394.039.
- Inman S. Olaparib doubles rPFS in heavily pretreated HRR+ mCRPC. OncLive®website. onclive.com/conference-coverage/esmo-2019/olaparib-improves-outcomes-in-heavily-pretreated-hrr-mcrpc. Published September 30, 2019. Accessed October 30, 2019.
- Mateo J, Porta N, McGovern UB, et al. TOPARP-B: a phase II randomized trial of the poly(ADP)-ribose polymerase (PARP) inhibitor olaparib for metastatic castration resistant prostate cancers (mCRPC) with DNA damage repair (DDR) alterations.J Clin Oncol.2019;37(suppl 15; abstr 5005). doi: 10.1200/JCO.2019.37.15_suppl.5005.
- Smith MR, Sandhu SK, Kelly WK, et al. Pre-specified interim analysis of GALAHAD: a phase II study of niraparib in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) and biallelic DNA-repair gene defects (DRD).Ann Oncol.2019;30(suppl 5; abstr LBA59). doi: 10.1093/annonc/mdz394.043.
- Adashek JJ, Jain RK, Zhang J. Clinical development of PARP inhibitors in treating metastatic castration-resistant prostate cancer.Cells.2019;8(8):pii:E860. doi: 10.3390/cells8080860.
- Abida W, Campbell D, Patnaik A, et al. Preliminary results from the TRITON2 study of rucaparib in patients (pts) with DNA damage repair (DDR)-deficient metastatic castration-resistant prostate cancer (mCRPC): updated analyses.Ann Oncol.2019;30(suppl 5). doi: 10.1093/annonc/mdz248.003.
- Ryan CJ, Abida W, Bryce AH, et al. TRITON3: an international, randomized, open-label, phase III study of the PARP inhibitor rucaparib vs. physician’s choice of therapy for patients with metastatic castration-resistant prostate cancer (mCRPC) associated with homologous recombination deficiency (HRD).J Clin Oncol.2018;36(suppl 6; abstr TPS389). doi: 10.1200/JCO.2018.36.6_suppl.TPS389.
- Golan T, Hammel P, Reni M, et al. Maintenance olaparib for germlineBRCA-mutated metastatic pancreatic cancer.N Engl J Med.2019;381(4):317-327. doi: 10.1056/NEJMoa1903387.
- Pilarski R. The role ofBRCAtesting in hereditary pancreatic and prostate cancer families.Am Soc Clin Oncol Educ Book.2019:79-86. doi: 10.1200/edbk_238977.
- Shroff RT, Hendifar A, McWilliams RR, et al. Rucaparib monotherapy in patients with pancreatic cancer and a known deleteriousBRCAmutation. JCO Precis Oncol.2018;2018. doi: 10.1200/PO.17.00316.
- Reiss Binder K, Mick R, O'Hara M, et al. A phase II, single arm study of maintenance rucaparib in patients with platinum-sensitive advanced pancreatic cancer and a pathogenic germline or somatic mutation inBRCA1,BRCA2orPALB2. Abstract presented at: American Association for Cancer Research Annual Meeting 2019; March 29-April 3, 2019; Atlanta, GA. Abstract CT234.cancerres.aacrjournals.org/content/79/13_Supplement/CT234.
- de Bono J, Ramanathan RK, Mina L, et al. Phase I, dose-escalation, two-part trial of the PARP inhibitor talazoparib in patients with advanced germlineBRCA1/2mutations and selected sporadic cancers. Cancer Discov.2017;7(6):620-629. doi: 10.1158/2159-8290.Cd-16-1250.
- Torphy RJ, Zhu Y, Schulick RD. Immunotherapy for pancreatic cancer: barriers and breakthroughs.Ann Gastroenterol Surg.2018;2(4):274-281. doi: 10.1002/ags3.12176.
- Robson ME, Tung N, Conte P, et al. OlympiAD final overall survival and tolerability results: olaparib versus chemotherapy treatment of physician’s choice in patients with a germline BRCA mutation and HER2-negative metastatic breast cancer.Ann Oncol.2019;30(4):558-566. doi: 10.1093/annonc/mdz012.
- Tutt A, Kaufman B, Garber J, et al. OlympiA: a randomized phase III trial of olaparib as adjuvant therapy in patients with high-risk HER2-negative breast cancer (BC) and a germline BRCA1/2 mutation (gBRCAm).Ann Oncol.2017;28(suppl 5). doi: 10.1093/annonc/mdx362.065.
- Litton JK, Rugo HS, Ettl J, et al. Talazoparib in patients with advanced breast cancer and a germlineBRCAmutation.N Engl J Med.2018(8);379:753-763. doi: 10.1056/NEJMoa1802905.
- Gruber JJ, Afghahi A, Hatton A, et al. Talazoparib beyond BRCA: a phase II trial of talazoparib monotherapy inBRCA1and BRCA2wild-type patients with advanced HER2-negative breast cancer or other solid tumors with a mutation in homologous recombination (HR) pathway genes.J Clinl Oncol.2019;37(suppl 15; abstr 3006). doi: 10.1200/JCO.2019.37.15_suppl.3006.
- Shao N, Shi Y, Yu L, et al. Prospect for application of PARP inhibitor in patients with HER2 negative breast cancer.Int J Biol Sci.2019;15(5):962-972. doi: 10.7150/ijbs.30721.
- FDA approves niraparib for HRD-positive advanced ovarian cancer. FDA website. www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-niraparib-hrd-positive-advanced-ovarian-cancer. Updated October 23, 2019. Accessed October 29, 2019.
- Penson RT, Valencia RV, Cibula D, et al. Olaparib monotherapy versus (vs) chemotherapy for germline BRCA-mutated (gBRCAm) platinum-sensitive relapsed ovarian cancer (PSR OC) patients (pts): phase III SOLO3 trial.J Clin Oncol.2019;37(suppl 15; abstr 5506). doi: 10.1200/JCO.2019.37.15_suppl.5506.
- Moore K, Colombo N, Scambia G, et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer.N Engl J Med.2018;379(26):2495-2505. doi: 10.1056/NEJMoa1810858.
- Moore KN, Secord AA, Geller MA, et al. Niraparib monotherapy for late-line treatment of ovarian cancer (QUADRA): a multicentre, open-label, single-arm, phase 2 trial.Lancet Oncol.2019;20(5):636-648. doi: 10.1016/S1470-2045(19)30029-4.
- González-Martín A, Pothuri B, Vergote I, et al; PRIMA/ENGOT-OV26/GOG-3012 Investigators. Niraparib in patients with newly diagnosed advanced ovarian cancer [published online September 28, 2019].N Engl J Med.doi: 10.1056/NEJMoa1910962.

Advances in Subsequent Therapies Shake Up Sequencing of ccRCC Treatment
April 25th 2024With the approval of belzutifan and other newer data for treating patients with recurrent renal cell carcinoma, the state of subsequent therapies is advancing beyond the reuse of frontline options with impacts on duration of response and quality of life.
Read More
Novel Approaches Focus on Limiting Toxicity in Older Patients With ALL
April 22nd 2024The major challenges for clinicians treating older patients with acute lymphoblastic leukemia surround the emergence of resistance to existing therapies and the toxicities associated with current chemotherapies.
Read More