Bell Discusses Options for a Patient With MSI-High, dMMR Endometrial Cancer
During a Targeted Oncology™ Case-Based Roundtable™ event, Maria Bell, MD, MPH, MBA, moderated a discussion on biomarker testing and treatment for a patient with extensive, well-differentiated, grade 1 endometrioid adenocarcinoma.
Maria Bell, MD, MPH, MBA
MODERATOR
Chief Medical Officer
Sanford Development and Research
Sanford Health
Sioux Falls, SD

EVENT REGION Great Plains
PARTICIPANTS Vinay Raja, MD | Peter Silberstein, MD | Bassam I. Mattar, MD | Radhakrishna Vegunta, MD | Anastas Provatas, MD | Hilary I. Ufearo, MBBS | Rabih Fahed, MD
CASE SUMMARY
In August 2021, a 64-year-old postmenopausal woman presented to her gynecologist with abnormal uterine bleeding lasting 4 months. She underwent menopause at 55 years of age. She is a widow, has no children, and lives alone. She has a medical history of polycystic ovary syndrome, arthritis, obesity (body mass index = 40), and hypertension well controlled with medication. Her 2020 Papanicolaou test was negative, and ECOG performance status was 1. A bimanual examination detected a mildly enlarged uterus and bilateral, inguinal lymphadenopathy.
In her diagnostic workup, a transvaginal ultrasound showed an endometrial echo of 7 mm with a subtle inhomogeneous pattern and scant amount of fluid. A pipelle biopsy was performed under local anesthesia with aspiration of sufficient tissue from the uterine walls. Histopathology was positive for extensive, well-differentiated, grade 1 endometroid adenocarcinoma. Lastly, an abdomen/pelvis CT showed no indications of secondary involvement.
DISCUSSION QUESTIONS
- Should every patient with endometrial cancer undergo molecular testing?
- For biomarker testing:
- When do you order testing?
- What type of testing do you order and for which biomarkers?
- How are you applying test results?
- How do you assess prognostic risk?
- Do you recommend mismatch repair deficiency (MMR) testing for all patients with endometrial cancer?
BELL: Should every patient with endometrial cancer undergo molecular testing? I do [test] in my practice when patients are diagnosed with endometrial cancer; many times they already have a biopsy. Once they’ve done the hysterectomy, I typically get POLE, TP53, HER2 [testing] if it’s serous disease, and estrogen receptor [ER] status. And that’s right at the beginning of their diagnosis. Who is typically doing molecular testing at the time of diagnosis?
RAJA: I agree. I [would be] doing the MMR testing for this patient and doing the ER status and checking the biomarkers.
SILBERSTEIN: I think it’s more important to do it when you’re going to use [it]—it’s more important in metastatic disease when you’re going to start to use these drugs. You may want to rebiopsy them when they have metastatic disease and do the testing at that time.
BELL: The reason why I do it early on is because we’re using molecular testing to triage on adjuvant treatment even for stage I and II disease. That’s why I typically do that, because if they’re POLE-positive for stage I or II disease, they don’t need any further treatment. TP53 disease behaves much more aggressively, so even for early stage I with TP53-mutant disease, we would be discussing systemic chemotherapy with those patients. That’s why we do it at the time of diagnosis. I don’t do the total next-generation sequencing testing.
I just do those select biomarkers because it will triage our adjuvant treatment.
CASE UPDATE
The patient was counseled on surgical options, then scheduled for total laparoscopic hysterectomy with bilateral salpingo-oophorectomy and sentinel lymph node dissection. She had stage IA, grade 1 endometrial cancer. Immunohistochemistry/molecular testing results showed an MMR deficiency (dMMR) and microsatellite instability–high status (MSI-H), and 3+ ER-positive.
For postoperative follow-up, the patient was counseled on surveillance every 6 months for 1 year, and annually thereafter. In August 2022, she reported intermittent pelvic pain over prior 4 weeks. CT scan of the chest, abdomen, and pelvis (CAP) suggested relapsed or metastatic disease with involvement of 1 right external iliac lymph node. Carboplatin/paclitaxel (6 cycles every 3 weeks) was administered, chemotherapy was well tolerated, and a complete response was recorded at end of regimen.
In April 2023, disease relapse was documented on routine follow-up. CT CAP scan showed heterogeneously enhancing mass in right suprarenal space, multiple bilateral pulmonary nodules, and a new right internal iliac lymph node (in addition to the previously observed positive lymph node). Radiologically guided fine-needle aspirate of the suprarenal mass confirmed metastatic endometrioid adenocarcinoma. The patient was counseled about systemic therapy options, during which she repeatedly expressed concerns about adverse events.
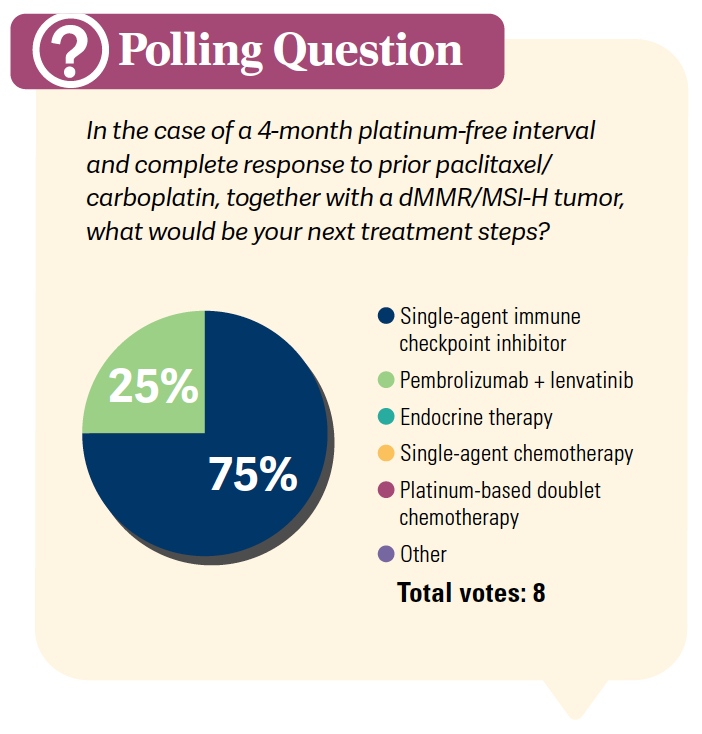
DISCUSSION QUESTIONS:
- What are the systemic therapy options for patients with metastatic endometrial cancer and MSI-H/dMMR disease?
- What are your determining factors in selecting next-line therapy? What if this patient had a partial response to carboplatin-paclitaxel? Would you order repeat molecular testing for this patient?
MATTAR: The time of the prior chemotherapy is very important for me. Typically if [relapse is] within a year, for sure within 6 months, I will not go back to the same agent. At this age with other comorbidity problems, I always like a checkpoint inhibitor in this setting. I feel like with lenvatinib [Lenvima], we may have to use it in MMR-proficient patients and it will have more adverse events, so this would not be my choice in this setting.
VEGUNTA: In this patient, if they progress on single-agent chemotherapy and if they have an MSI-high tumor, I would use a single-agent immune checkpoint inhibitor. If they’re not MSI high, I would look at the location of the disease and also their performance status, and will consider immune checkpoint inhibitor with lenvatinib [as a] treatment option.
PROVATAS: I would consider pembrolizumab [Keytruda] and lenvatinib as an option.
BELL: Would you repeat molecular testing?
SILBERSTEIN: We didn’t used to use it. Initially, we got the MSI status, and we did not use that information. If we have the MSI at this time and all these other things, we will use the information. This patient is MSI high; [I would use] pembrolizumab as a majority would do.
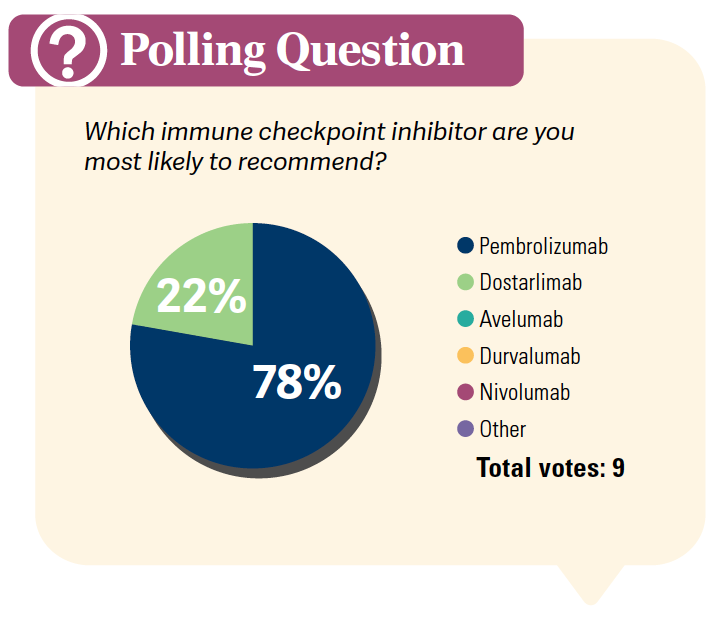
BELL: Any difference in choosing one or the other? Is it just because of more use of pembrolizumab in the past? What are your thoughts about the differences between the 2 drugs?
RAJA: [I have] more experience with pembrolizumab, and I’m not used to dostarlimab. I think they’re all equally good, but if there are any differences, I would like to hear from you. But I think they’re equal.
BELL: I would say it’s similar between them. It reminds me of the PARP [inhibitors] where people were very familiar with olaparib [Lynparza] and then the others came, so I think it’s what you’re used to using. That’s typically what I see.
[Is anyone] having issues with insurance coverage for one immune checkpoint inhibitor over the other?
UFEARO: I’ve not used a lot of dostarlimab; I’ve used mainly pembrolizumab and the coverage has been good.
BELL: Is anyone using dostarlimab?
SILBERSTEIN: Insurance companies are very picky these days.... [I choose] very carefully, and I can win every time with the insurance companies, but I try not to [deviate from] the National Comprehensive Cancer Network guidelines.
BELL: I’m having a similar experience with the insurance providers as well.
MATTAR: If I want to use it in combination with chemotherapy, I’d probably go with [dostarlimab]. But in general, I use pembrolizumab alone and combine [dostarlimab] with paclitaxel/carboplatin.
FAHED: I echo what Dr Mattar said.
BELL: Are you seeing more toxicity if you use pembrolizumab with chemotherapy?
FAHED: Not really. I’m more impressed with the overall survival data with dostarlimab compared with pembrolizumab in combination with chemotherapy.

Roundtable Roundup: Treatment for Metastatic pMMR Endometrial Cancer
July 23rd 2024In separate, live virtual events, Michael J. Birrer, MD, PhD, and Jubilee Brown, MD, surveyed participants on the treatment of a postmenopausal woman with stage IVA endometrial cancer after first-line chemotherapy.
Read More
Depth of Response With Quadruplet Regimens Considered in Newly Diagnosed Multiple Myeloma
July 18th 2024During a Case-Based Roundtable® event, Timothy Schmidt, MD, and participants discussed treatment selection for a 54-year-old patient with transplant eligible R-ISS stage 2/R2-ISS stage 3 IgG-κ myeloma.
Read More
Rossetti Reviews Myelofibrosis Risk Stratification and Outcome Data for Pacritinib
July 17th 2024During a Case-Based Roundtable® event, James M. Rossetti, DO, discussed the role of risk scoring and stratification tools and treatment for a patient with declining hemoglobin and platelet counts due to primary myelofibrosis.
Read More