Multiple Myeloma: Top 10 Advances in the Past 10 Years
Treatment options have expanded widely for patients with multiple myeloma over the past several years, resulting in significantly improved outcomes for these patients.
Kenneth C. Anderson, MD

Treatment options have expanded widely for patients with multiple myeloma
over the past several years, resulting in significantly improved outcomes for these patients.
Two decades ago, the median survival was approximately 3 years, and now it is 8 to 10 years1 — and can be even longer for many patients, according to Kenneth C. Anderson, MD, program director of the Jerome Lipper Multiple Myeloma Center and LeBow Institute for Myeloma Therapeutics at Dana-Farber Cancer Institute and the Kraft Family Professor of Medicine at Harvard Medical School, both in Boston, Massachusetts. The reason? Anderson attributed it to combining classes of drugs like proteasome inhibitors, immunomodulatory drugs (IMiDs), and monoclonal antibodies for newly diagnosed patients and those with relapsed/refractory multiple myeloma. “We can increase the frequency of response and the extent of response, so we get very deep responses,” he said.
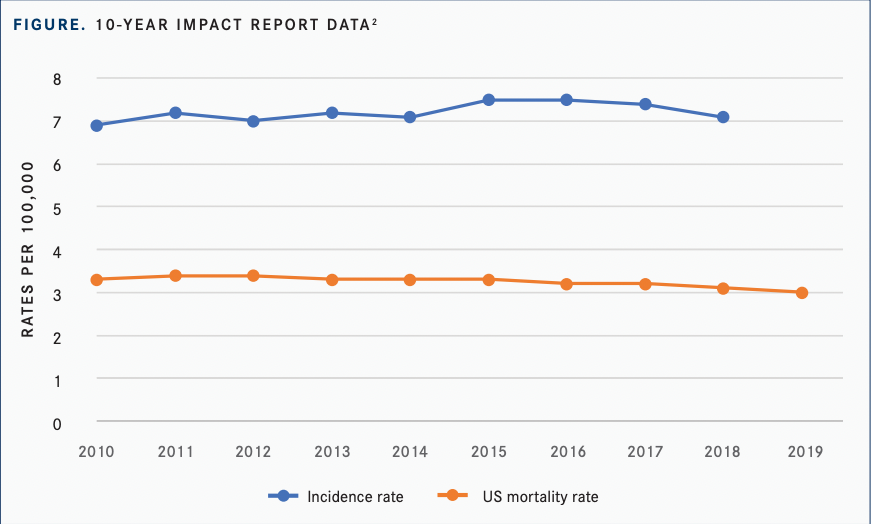
In 2021, there were an estimated 34,910 new cases of myeloma, accounting for 1.8% of all new cancer cases. Last year, approximately 12,410 individuals died from myeloma. Death rates have decreased slightly in the past decade, from 3.4 per 100,000 in 2012 to 3.0 per 100,000 in 2019, the last year data were available (FIGURE).2
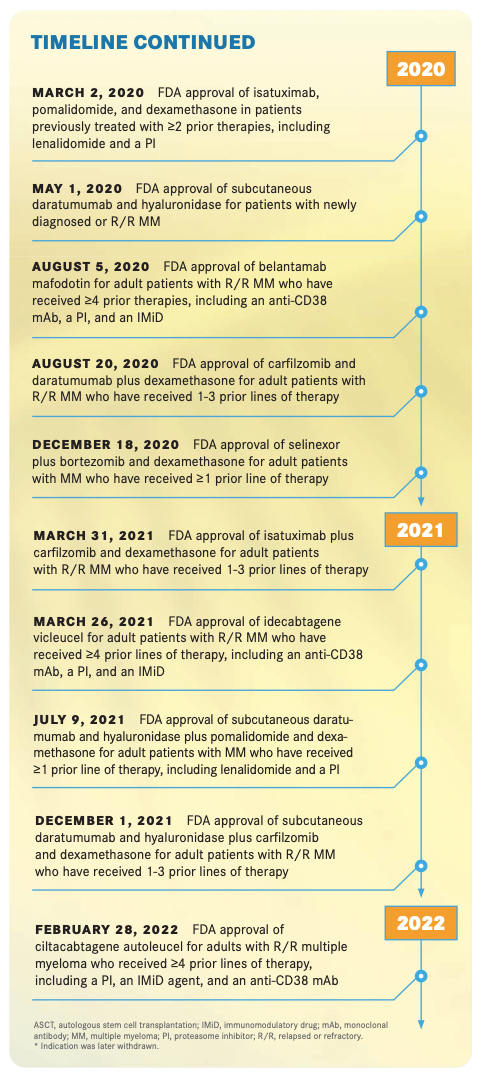
In the past decade, the FDA approved 13 new agents and 29 treatments for multiple myeloma, which have transformed the treatment paradigm for patients with newly diagnosed and relapsed/ refractory multiple myeloma, according to Anderson. “Most importantly, it’s changed the outlook and the outcome for our patients,” he said in an interview with Targeted Therapies in OncologyTM.
What follows are the 10 top advances in multiple myeloma from the past decade.
The Starting Point
In the 1980s, doctors relied on high-dose chemotherapy and autologous stem cell transplantation (ASCT) from bone marrow, Anderson said. In the 1990s, treatment included high-dose therapy and peripheral blood as a source of stem cell transplant.3 By the late 1990s and the first decade of 2000, IMiDs and proteosome inhibitor drugs were approved for use in multiple myeloma.
Around 2012, lenalidomide (Revlimid) and dexamethasone (Rd), followed by ASCT, became a standard-of-care treatment option. Intermediateor high-risk patients might have received a bortezomib (Velcade)–based induction therapy, followed by ASCT, with bortezomib-based maintenance. Patients ineligible for ASCT might have received Rd for standard-risk disease or a bortezomib-based treatment for intermediate- or high-risk disease.4
Sagar Lonial, MD, FACP

“The big debate at that time was, ‘Was is it good to combine our 2 most active agents, or should we try and sequence agents so that we preserve or save treatments for later on?’” Sagar Lonial, MD, FACP, said. Lonial is chief medical officer of Winship Cancer Institute of Emory University and professor and chair of the Department of Hematology and Medical Oncology as well as the Anne and Bernard Gray Family Chair in Cancer at Emory University School of Medicine in Atlanta, Georgia.
These patients may have experienced increased survival rates from the treatment, but almost all would relapse.5
Redefining Whom to Treat
The past decade saw paradigm shifts in myeloma management, including early intervention. The International Myeloma Working Group updated its diagnostic criteria in 2014 to add validated biomarkers beyond the standard CRAB features (hypercalcemia, renal failure, anemia, and bone lesions) for defining multiple myeloma.6 Additionally, treatment options were added for some patients with smoldering multiple myeloma; patients could be treated earlier, based on their risk of end-organ damage, according to Shaji Kumar, MD, a professor of medicine and consultant for the Division of Hematology at Mayo Clinic in Rochester, Minnesota.
Shaji Kumar, MD

“Clinical trials have shown that there’s an advantage to starting to treat patients with smoldering multiple myeloma who are at a very high risk of disease progression,” Kumar said.
Previously, patients with smoldering myeloma were treated only when they developed complications, such as high blood calcium, kidney dysfunction, anemia, or bone disease, Anderson said. With the advent of effective and well-tolerated novel agents, these complications may be prevented.
Current Frontline Treatments
The current standard induction regimen of lenalidomide, bortezomib, and dexamethasone (RVd)7 and single ASCT for eligible patients, followed by risk-adaptive maintenance, has shown median progression-free survival (PFS) of 65 months for all patients and 76.5 months for standard-risk patients. The median overall survival (OS) is 126.6 months.8
“I think what allowed us to do that was combining drugs together, getting deep responses, building on those deep responses with transplant, and then using risk-adapted maintenance,” Lonial said. Physicians currently debate the need for 4-drug regimens, such as with the addition of the anti-CD38 monoclonal antibody daratumumab (Darzalex), to further improve outcomes.
Transplant remains a standard of care for those who are eligible. “If you put the RVd and daratumumab before and after a transplant, you can get minimal residual disease [MRD]–negative responses in 70% of patients, which is quite remarkable, and that does correlate with much better outcomes,” Anderson said.9
RVd lite is a modified regimen with a lower dose of lenalidomide, which also has high response rates and a long PFS, with a favorable adverse event profi le for nontransplant candidates, Anderson said. The median PFS with the modified regimen was found to be 35.1 months,10 and this triplet as well as the daratumumab with lenalidomide and dexamethasone triplet have become preferred regimens for patients who are ineligible for transplant.7
Eliminating ASCT is a topic of discussion but not currently advised.11 “I would be cautious about throwing out transplant,” Lonial said. “We’ve been trying to do that for 15 years, and it continues to survive and thrive even in the context of new treatments.” That is because every trial comparing transplant to no transplant, even with new drugs, has shown that transplant deepens the response and offers significant benefits, he said.
Proteosome Inhibitors
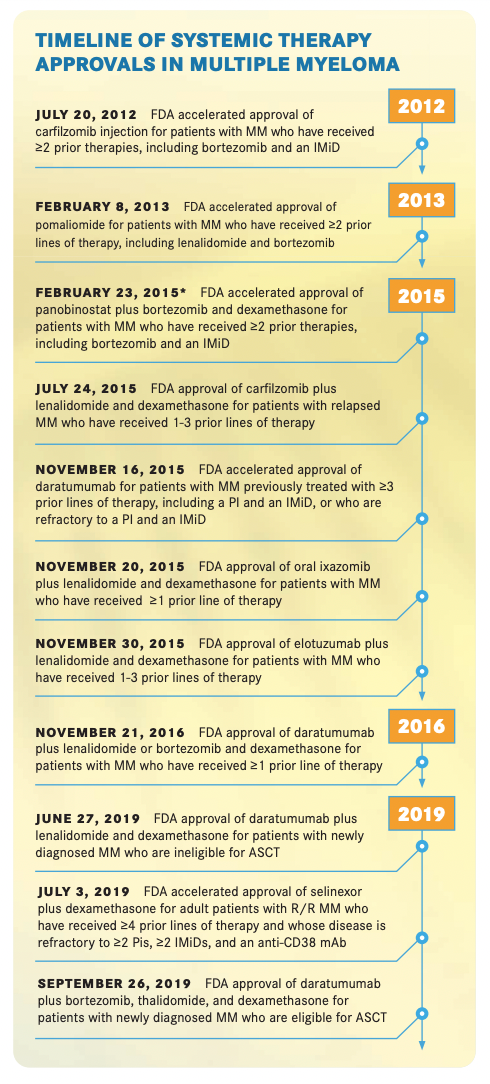
In the past decade, the proteosome inhibitor class of drugs has expanded, with the FDA approval of carfilzomib (Kyprolis) in 201212 and ixazomib (Ninlaro) in 2015,13 the first FDA-approved oral proteasome inhibitor (TIMELINE). Carfilzomib acts as a single agent or a doublet with dexamethasone.14
“Pomalidomide [Pomalyst] and carfilzomib are playing a much greater role in the management of patients throughout their disease course now than they [did] a decade ago, and they have significant advantages in terms of toxicity as well as potency that I think makes them...great drugs for us to have available for our patients,” Lonial said.
Carfilzomib is considered the more potent of the proteasome inhibitors.14-16 In the ENDEAVOR trial (NCT01568866), carfilzomib was compared with the first-generation proteasome inhibitor bortezomib, both in combination with dexamethasone, in patients with relapsed/refractory multiple myeloma. The median PFS was approximately double for carfilzomib/dexamethasone compared with bortezomib/dexamethasone at 18.7 months vs 9.4 months, respectively (HR, 0.53; 95% CI, 0.44-0.65; P < .0001).15
Ixazomib is advantageous as an oral agent, with the phase 3 TOURMALINE-MM1 trial (NCT01564537) showing a PFS advantage for oral ixazomib plus lenalidomide/dexamethasone vs lenalidomide/dexamethasone alone (20.6 months vs 14.7 months, respectively; HR, 0.74; P = .01) in patients with relapsed/ refractory disease.16 Although patients can get some peripheral neuropathy, recent clinical trials show it is not as potent as with some other proteosome inhibitors,17 Kumar noted.
Immune-Based Therapies
Anderson defines this past decade as the “immune era,” and Lonial agrees, noting that the revolution in myeloma treatment during the past 10 years has been in immune-based therapies. That includes daratumumab, which has become important for patients who are ineligible for ASCT, based on the results of the MAIA trial (NCT02252172).18 Investigators found a significant PFS benefit for daratumumab plus lenalidomide and dexamethasone vs lenalidomide and dexamethasone alone in transplant-ineligible patients with newly diagnosed multiple myeloma (not reached vs 34.4 months, respectively; HR, 0.53; 95% CI, 0.43-0.66; P < .0001). “The average remission is around 5 years. That’s better than anything else we’ve seen in that patient population,” Lonial said. “But it’s continuous therapy.”
Daratumumab has single-agent activity and “is potent in pretty much every combination we tested, whether it’s a proteosome inhibitor or an IMiD or in combination with other immune therapies,” Lonial noted.
Daratumumab targeting CD38 and elotuzumab (Empliciti) targeting SLAMF7 were both approved in 2015.19,20 Elotuzumab “doesn’t have single-agent activity but does have effects in combination with both lenalidomide and pomalidomide in phase 3 trials, demonstrating superiority over lenalidomide and pomalidomide alone,” Lonial said.
In the phase 3 ELOQUENT-2 trial (NCT01239797), elotuzumab in combination with lenalidomide and dexamethasone showed a significant increase in PFS compared with lenalidomide and dexamethasone alone (19.4 months vs 14.9 months, respectively; HR, 0.70; 95% CI, 0.57-0.85; P < .001).21
A new formulation of daratumumab with hyaluronidase-fi hj (Darzalex Faspro) was approved in 2021.22 “[It] has been a big game changer for myeloma because it’s been combined with a variety of different drugs, and the combinations of it are quite effective,” Kumar said, noting that it is also well tolerated. Usage of the subcutaneous formulation became especially important during the COVID-19 pandemic, when oncologists sought to reduce the number of office visits to safeguard patients’ health.
Pomalidomide was approved in 2013 as a second-generation IMiD that can overcome resistance to lenalidomide and thalidomide (Thalomid).23 In the phase 3 NIMBUS/MM-003 trial (NCT01311687) that enrolled patients with relapsed/refractory multiple myeloma and at least 2 prior therapies, pomalidomide and low-dose dexamethasone showed an improved PFS compared with high-dose dexamethasone (4.0 months vs 1.9 months, respectively; HR, 0.48; 95% CI, 0.39-0.60; P < .0001).24
Other Treatment Classes
In the past decade, patients with multiple myeloma benefi ted from development and approvals in other treatment classes, including antibody-drug conjugates (ADC), nuclear transport inhibitors, and IMiDs. The ADC belantamab mafodotin-blmf (Blenrep) was approved in 202025; it has single-agent activity in relapsed/refractory myeloma and is being considered in combinations as well. The agent is labeled for use after at least 4 prior therapies, but the prescribing label comes with a boxed warning for alterations in vision. Lonial highlighted that it is the first of the B-cell maturation antigen (BCMA)–directed treatments, a promising novel therapeutic target in multiple myeloma due to its expression on mature B lymphomas and limited expression on hematopoietic stem cells.26
The XP01 nuclear transport inhibitor selinexor (Xpovio) was approved in 2019.27 It has a new target and mechanism of action, Lonial noted, and is approved alone, with dexamethasone, in combination with proteasome inhibitors, and in earlier lines of relapse.
The histone deacetylase inhibitor panobinostat (Farydak) received accelerated approval in 2015 from the FDA for previously treated multiple myeloma,28 but the indication was withdrawn in 2021 because the manufacturer could not complete the necessary postapproval clinical research to maintain approval.29 “At this point, it’s something that cannot be easily used [for myeloma],” Kumar said.
CAR T and BiTEs
One of the decade’s big stories in myeloma is chimeric antigen receptor (CAR) T-cell and bispecific T-cell engager (BiTE) therapies. The CAR T-cell therapy idecabtagene vicleucel (Abecma) was approved in 2021.30 The anti-BCMA targeted therapy “clearly has activity and depth of response in a refractory patient population and is being tested in earlier lines of therapy as well,” Lonial said. He added that the average response is approximately a year and that the average response for ciltacabtagene autoleucel (cilta-cel; Carvykti), which was recently FDA approved,31 looks to be about 2 years. “CAR T cells offer us a window into a way to use BCMA as a mechanism by which to eradicate the clone,” he said. Cilta-cel was approved by the FDA on February 28, 2022, for adults with relapsed or refractory multiple myeloma after 4 or more prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.32
“The 2 CAR T-cell therapies have been remarkable in that they have achieved complete and even MRD-negative responses in patients with far advanced relapsed/refractory myeloma, who literally have no other options,” Anderson said.
BiTEs are not yet FDA approved but look promising. “I think many of us were skeptical that [BiTEs] would be active in myeloma, given that T-cell health may be suboptimal in a refractory myeloma patient population. But that’s clearly not the case, with response rates between 60% and 80% with any of the BCMA-directed CAR T cell or bispecifics,” Lonial said, noting that he is excited about the potential of having an off-the-shelf immune-stimulator product, which could be a great adjunct to CAR T cells.
Between 6 and 12 BiTEs are in clinical trials for myeloma, Anderson said. Most CAR T and BiTE products target BCMA, as does belantamab mafodotin.26 “It’s a good target, and right now it’s only being targeted in far-advanced myeloma—but it’s going to be used earlier in the disease course,” he said.
MRD: The Potential End to Continuous Therapy
Clinicians are using end points like MRD to begin to potentially move away from continuous therapy, Lonial said. “With the availability of potentially sustained MRD negativity as an end point, we may be getting to a point where we can stop therapy,” he said.
MRD is assessed with multicolor flow cytometry or gene sequencing,33 Anderson said. Triplet and quadruplet treatments can achieve MRD negativity in patients with newly diagnosed and relapsed/refractory multiple myeloma, he noted, and a durable response correlates with marked PFS and OS advantages.
Cytogenetics-Based Stratification Standards
Myeloma is a heterogenous disease, and this newer revelation is leading to genetic risk stratification at diagnosis to determine therapy, Kumar said. “A one-size-fits-all approach may not be the best way to deal with patients with multiple myeloma,” he said. Different prognostic factors allow clinicians to better defi ne potential outcomes, helping them formulate individual treatment strategies.
Fluorescence in situ hybridization testing is an accepted standard to help define whether a patient has standard- or high-risk multiple myeloma. This has markedly improved the prognostication in myeloma, Anderson said.
There’s also a move toward molecular testing with gene sequencing or mutation analysis to determine disease risk, as well as mass spectrometry to determine treatment paths based on protein analyses. “Because mass spectrometry is a little bit more reproducible, it’s a little bit more objective,” Lonial said, noting that can make a difference when using antibodies like daratumumab and elotuzumab and potentially the BiTEs.
Looking Ahead
Each therapy is a building block to new treatments. The words “cure” or “functional cure” are now in play, Kumar said. By extending the duration that myeloma is under control, at least for older patients, myeloma may not be the cause of death. Due to drug combinations and better supportive care, each treatment line can control the disease for 3 to 6 years, so patients are living longer with myeloma, he noted.
Perhaps ASCT will be eliminated if BiTEs and/or CAR T cells can show better results than ASCT. The future for multiple myeloma treatment includes determining optimal therapy combinations based on disease characteristics, with the possibility of discontinuing treatment. “But, of course, our goal is to try and cure the disease,” Kumar said.
There’s also a move toward molecular testing with gene sequencing or mutation analysis to determine disease risk, as well as mass spectrometry to determine treatment paths based on protein analyses. “Because mass spectrometry is a little bit more reproducible, it’s a little bit more objective,” Lonial said, noting that can make a difference when using antibodies like daratumumab and elotuzumab and potentially the BiTEs.
References
1. GullaA, Anderson KC. Multiple myeloma: the (r)evolution of current therapy and a glance into future. Haematologica. 2020;105(10):2358-2367. doi:10.3324/haematol.2020.247015
2. Cancer stat facts: myeloma. National Cancer Institute Surveillance, Epidemiology, and End Results Program. Accessed February 11, 2022.https://seer.cancer.gov/statfacts/html/mulmy.html
3. Al Hamed R, Bazarbachi AH, Malard F, Harousseau JL, Mohty M. Current status of autologous stem cell transplantation for multiple myeloma. Blood Cancer J.2019;9(4):44. doi:10.1038/s41408-019-0205-9
4. Rajkumar SV. Multiple myeloma: 2012 update on diagnosis, risk-stratification, and management Am J Hematol.2012;87(1):78-88. Published correction appears in Am J Hematol.2012;87(4):452. Published correction appears in Am J Hematol. 2014;89(6):669.
5. Mahindra A, Laubach J, Raje N,Munshi N, Richardson PG, Anderson K. Latest advances and current challenges in the treatment of multiple myeloma. Nat Rev Clin Oncol.2012;9(3):135-143. doi:10.1038/nrclinonc.2012.15
6. Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol.2014;15(12):e538-e548. doi:10.1016/S1470-2045(14)70442-5
7. NCCN Clinical Practice Guidelines in Oncology. Multiple myeloma,version 4.2022. Accessed February 12, 2022. https://bit.ly/3h865Zj
8. Joseph NS, Kaufman JL, Dhodapkar MV, et al. Long-term follow-up results of lenalidomide, bortezomib, and dexamethasone induction therapy and risk-adapted maintenance approach in newly diagnosed multiple myeloma. J Clin Oncol.2020;38(17):1928-1937. doi:10.1200/JCO.19.02515
9. Abdallah N, Kumar SK. Daratumumab in untreated newly diagnosed multiple myeloma. Ther Adv Hematol.2019;10:2040620719894871. doi:10.1177/2040620719894871
10. O’Donnell EK, Laubach JP, Yee AJ, et al. A phase 2 study of modified lenalidomide, bortezomib, and dexamethasone in transplant-ineligible multiple myeloma. Br J Haematol. 2018;182(2):222-230. doi:10.1111/bjh.15261
11. Devarakonda S, Efebera Y, Sharma N. Role of stem cell transplantation in multiple myeloma. Cancers. 2021;13(4):863. doi:10.3390/cancers13040863
12. Carfilzomib. FDA. Updated May 4, 2016. Accessed February 12, 2022. https://bit.ly/3sUg0as
13. Ixazomib. FDA. Updated November 20, 2015. Accessed February 12, 2022. https://bit.ly/3vbmKDp
14. Yee AJ. The role of carfilzomib in relapsed/refractory multiple myeloma. Ther Adv Hematol. 2021;12:20406207211019612. doi:10.1177/20406207211019612
15. Dimopoulos MA, Moreau P, Palumbo A, et al; ENDEAVOR Investigators. Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): a randomised, phase 3, open-label, multicentre study. Lancet Oncol. 2016;17(1):27-38. doi:10.1016/S1470-2045(15)00464-7
16. Moreau P, Masszi T, Grzasko N, et al; TOURMALINE-MM1 Study Group. Oral ixazomib, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;374(17):1621-1634. doi:10.1056/NEJMoa1516282
17. Brayer J, Baz R. The potential of ixazomib, a second-generation proteasome inhibitor, in the treatment of multiple myeloma. Ther Adv Hematol. 2017;8(7):209-220. doi:10.1177/2040620717710171
18. Facon T, Kumar SK, Plesner T, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol.2021;22(11):1582-1596. doi:10.1016/ S1470-2045(21)00466-6
19. Daratumumab injection. FDA. Updated November 16, 2015. Accessed February 12, 2022. https://bit.ly/3s8CjtH
20. Elotuzumab. FDA. Updated November 30, 2015. Accessed February 12, 2022. https://bit.ly/3tgRTmz
21. Lonial S, Dimopoulos M, Palumbo A, et al; ELOQUENT-2 Investigators. Elotuzumab therapy for relapsed or refractory multiple myeloma. N Engl J Med. 2015;373(7):621-631. doi:10.1056/NEJMoa1505654
22. FDA approves daratumumab and hyaluronidase-fihj with pomalidomide and dexamethasone for multiple myeloma. FDA. Updated July 12, 2021. Accessed February 12, 2022. https://bit.ly/3v3DEDR
23. Pomalidomide. FDA. Updated October 9, 2015. Accessed February 12, 2022. https://bit.ly/3LVrvac
24. Ríos-Tamayo R, Martin-García A, Alarcón-Payer C, et al. Pomalidomide in the treatment of multiple myeloma: design, development and place in therapy. Drug Des Devel Ther. 2017;11:2399-2408. doi:10.2147/DDDT.S115456
25. FDA granted accelerated approval to belantamab mafodotin-blmf for multiple myeloma. FDA. Updated August 6, 2020. Accessed February 12, 2022. https://bit.ly/3JOreUx
26. Sanchez L, Dardac A, Madduri D, Richard S, Richter J. B-cell maturation antigen (BCMA) in multiple myeloma: the new frontier of targeted therapies. Ther Adv Hematol. 2021;12:2040620721989585. doi:10.1177/2040620721989585
27. FDA grants accelerated approval to selinexor for multiple myeloma. FDA. Updated July 3, 2019. Accessed February 12, 2022. https://bit.ly/3vcApu4
28. Panobinostat. FDA. Updated February 24, 2015. Accessed February 12, 2022. https://bit.ly/3pa43w0
29. Secura Bio announces U.S.withdrawal of Farydak(panobinostat) NDA. News release. Secura Bio Inc.November 30, 2021. Accessed February 12, 2022. https://prn.to/3BKsN32
30. FDA approves idecabtagene vicleucel for multiple myeloma. FDA. Updated March 29, 2021. Accessed February 12, 2022. https://bit.ly/3vhR7bz
31. U.S. FDA Approves CARVYKTI™ (ciltacabtagene autoleucel), Janssen’sFirst Cell Therapy, a BCMA-Directed CAR-T Immunotherapy for the Treatment of Patients with Relapsed or Refractory Multiple Myeloma. News release. Accessed February 4, 2022. https://bit.ly/3CgPGvu
32. Kostopoulos IV, Ntanasis-Stathopoulos I, Gavriatopoulou M, Tsitsilonis OE, Terpos E. Minimal residual disease in multiple myeloma: current landscape and future applications with immunotherapeutic approaches. Front Oncol. 2020;10:860. doi:10.3389/fonc.2020.00860
Leon-Ferre Explores Targeting of PIK3CA Alterations in ER+ Breast Cancer
July 24th 2024During a live Community Case Forum event in partnership with the Minnesota Society of Clinical Oncology, Roberto A. Leon-Ferre, MD, discussed drugs targeting PIK3CA alterations in patients with ER+ metastatic breast cancer.
Read More
Khouri Discusses Selinexor Dosing and Management Approaches for R/R MM
July 22nd 2024During a Case-Based Roundtable® event, Jack Khouri, MD, and participants discussed the use of selinexor with dose modifications to manage toxicity in patients with relapsed/refractory multiple myeloma.
Read More
Depth of Response With Quadruplet Regimens Considered in Newly Diagnosed Multiple Myeloma
July 18th 2024During a Case-Based Roundtable® event, Timothy Schmidt, MD, and participants discussed treatment selection for a 54-year-old patient with transplant eligible R-ISS stage 2/R2-ISS stage 3 IgG-κ myeloma.
Read More
Rossetti Reviews Myelofibrosis Risk Stratification and Outcome Data for Pacritinib
July 17th 2024During a Case-Based Roundtable® event, James M. Rossetti, DO, discussed the role of risk scoring and stratification tools and treatment for a patient with declining hemoglobin and platelet counts due to primary myelofibrosis.
Read More