EP. 1A: The Role of Imaging and Genomic Testing in Prostate Cancer Treatment
In this first video of the series, Neeraj Agarwal, MD, of the Huntsman Cancer Institute of the University of Utah explains the role of imaging and genomic testing in the diagnosis and treatment of prostate cancer.
Episodes in this series

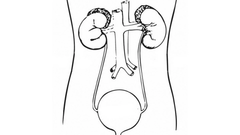
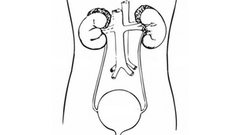
After the initial diagnosis of prostate cancer, depending upon the risk of the prostate cancer—[but] especially in those who belong to intermediate- [or] high-risk localized prostate cancer—according to [the] NCCN guidelines, we obtain [a] bone scan [and/or a] CT scan of the pelvis, plus [or] minus [the] abdomen … [B]asically, we want to rule out regional or distant metastasis. But if you look at intermediate- and high-risk prostate cancer, when the risk of metastasis is higher, clearly there is a definite role of bone scan and CT scan. So the first step is to rule out metastasis. Once you have ruled out [metastasis], and as long as these patients are well—have a life expectancy of more than ten years—there are 3 genomic tests which can be performed.
[O]verall, what these tests … are doing is that they are helping us in determining the likelihood of metastasis and death because of prostate cancer in our patients who are considered to have localized prostate cancer today. [W]hen you are doing this testing—genomic testing—they’re actually helping us in determining whether we should treat these patients with definitive therapy or [if] it [is] safe for these patients to undergo active surveillance … That is one of the reasons we are doing this testing.
So, questions may be asked in this context. If you already [have] NCCN risk stratification in place, you have CAPRA [Cancer of the Prostate Risk Assessment] scores in place, why do you need these tests? Studies have shown that Decipher [Prostate] …, Oncotype DX [Genomic Prostate Score assay], and Prolaris testing can independently predict or correlate with metastasis and prostate cancer–specific mortality versus traditional NCCN risk stratification or CAPRA risk.
So, I think because of the independent nature of being able to predict prostate cancer–specific mortality, onset of metastasis, [and] onset of biochemical recurrence, there is a value of doing those tests.
These genomic tests can independently help our patients in deciding whether they want to pursue active surveillance or definitive therapy with surgery or radiation by telling them their risk of recurrence—either biochemical recurrence or metastatic disease—or their risk of dying because of prostate cancer.