Case 2: The Diagnosis of Polycythemia Vera
Episodes in this series

Srdan Verstovsek, MD, PhD: Let’s have a nice discussion here on a number of topics. Let’s start with the diagnosis here again. Rami, we had a case here where the hematocrit level was 63%. The patient didn’t have a bone marrow biopsy, but is it absolutely necessary?
Rami Komrakji, MD: No. So you bring a good point. I don’t think you need a bone marrow for establishing the diagnosis in those cases. If the hematocrit is high enough, with the presence of a clonal marker like JAK2, I think one can make a diagnosis of polycythemia vera [PV]. However, we do get bone marrows at baseline typically, and I do them for more prognostic value and as a baseline to have as the patient’s disease progresses down the road to compare to. There are studies that look at the presence of fibrosis, for example, on the bone marrow in p vera, and that could be prognostic. I like to get a baseline because if there’s a question about transformation, I would like to compare at baseline. However, in certain cases, when the hematocrit is high enough, you have a clonal marker positive, you do not need the bone marrow for establishing the diagnosis.
Srdan Verstovsek, MD, PhD: So in this case, for example, the very high hematocrit level, with a JAK2 mutation positivity on blood, with the low serum erythropoietin, perhaps even iron deficiency, you would have enough there to say it is a PV.
Rami Komrakji, MD: Yes.
Srdan Verstovsek, MD, PhD: Now, in terms of bone marrow, so you have some prognosis there, 20% of the patients may have a grade 2 fibrosis already at the time of diagnosis. You can also do the cytogenetic analysis to see occasionally whether there are some patients with chromosomal abnormalities. Prithviraj, is there a role of a genetic testing—expensive genetic testing—other than the JAK mutation? There is some information, but is it practical to test for it? Do we need it in short order? Yes? No? Maybe?
Prithviraj Bose, MD: That’s a great question as well. I think the role of that is, right now, more of a research role because there are some mutations, SRSF2, ASXL1, and IDH, that are adverse in PV. But, again, you’re not going to act on them right now. So this is quite often a debated topic, whether everyone should be tested. Now, in research institutions such as ours, we do that, and it’s important from a research standpoint I think. But right now, from a patient care standpoint, perhaps not in a universal manner.
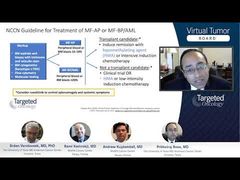
Srdan Verstovsek, MD, PhD: When it comes to therapy, hydroxyurea comes up as a drug of choice, although the guidelines that, Andrew, you just showed very well, would suggest hydroxyurea or interferon. Is hydroxyurea preferred in your hands, and are there cases for interferon, and once you start the cytoreductive therapy, what are the goals? Is it just hematocrit or is it something else? Is it everything else in blood cell counts, is it symptoms with the spleen? Give us a little bit of a summary here quickly. What do you choose and what do you aim for?
Andrew Kuykendall, MD: I think when starting patients on therapy, we mentioned the hydroxyurea and interferon are both first-line options. Typically speaking, I think that we often consider interferon more for younger patients—that we think there’s maybe a little bit of a better role for using this long term in those, and hydroxyurea more for the older patients. Now, I think they are both options, and certainly they’re very different and there’s a lot of discussion as far as which one is more appropriate in different circumstances. As far as hydroxyurea, we know it’s been used for a long time. It’s easy, we have expected adverse effects that we know how to manage, it’s relatively cheap, it’s oral and easy to take. Interferon, for the most part we’re using pegylated interferon, which is a subcutaneous injection given weekly. The cost of that is a little bit different, so sometimes, it factors into that as well.
But the goal of this is to really improve the quality of life of the patient and prevent thrombosis. In general, polycythemia vera has a favorable prognosis, so we have to think about not just improving quantity of life or reducing the risk of thrombotic events, but also maintaining a quality of life as well. So, when you mention the aspects of what do we consider uncontrolled disease in the setting of cytoreductive therapy, I think there are easy things. We want to prevent thrombosis, we want to be able to control blood counts, we want to keep people functional and active. But there’s also a lot of other things that go into this. Hydroxyurea, ruxolitinib was very good at controlling symptoms of the disease, and symptoms of the disease are something that is what bothers patients on a day-to-day basis. In someone whose blood counts are controlled, but have debilitating symptoms or symptoms that are bothersome, I think that’s a failure of our therapy and we need to consider other options. In the same manner, if we’re on some sort of treatment that is requiring the patient to have phlebotomies every month, I think that can also be considered a failure of therapy.
Transcript edited for clarity.