Case 4: The Importance of Early Intervention in MPN
Episodes in this series

Srdan Verstovsek, MD, PhD: Rami, you have seen in this slide that it seems that the characteristics of a patient who transforms, at the time of transformation, indicates that the molecular evolution is the main reason. But cytogenetics have a role. There are other possible biological parameters. What are they, and how can we use them for perhaps therapeutic decision-making in that setting?
Rami Komrokji, MD: I think that’s a good question. Obviously, in most cases, what we observe in transformation is clonal evolution. However, there are cases where we, for example, see the JAK2 mutation disappearing, indicating that probably subclones have emerged. I think it will be important to try to identify, as you were alluding to, who those patients are and if can we intervene earlier in those patients to try to alter that. In my mind, the accelerated phase is just a transition, and those patients will get leukemia eventually. For example, as Prithviraj showed, there is a subset enriched with the IDH mutation, even those can happen in almost 20%, 30% of the patients at the time of transformation. Maybe if we are starting to see those mutations, an intervention with an IDH inhibitor would have a role. But in most of the cases, the disease is clonal evolution. Many of those mutations so far are not targetable so that we can do anything about it.
To your point of thinking of identifying those patients, the only therapy we have is really allogeneic stem-cell transplant. To your point, if we can identify ET [essential thrombocythemia] patients who have a high risk, should we be thinking of transplanting those patients if we have a solidarity of those models predicting the disease progressing to leukemia, or at the point where we see a molecular change that will suggest that the transformation is coming and inevitable?
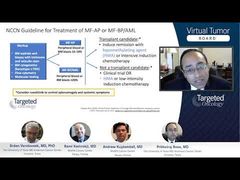
Srdan Verstovsek, MD, PhD: That’s a very good point, but unfortunately, we and others are not really allowed, because of the cost issue, to monitor patients’ molecularity, to do molecular testing and see whether there is evolution to intervene earlier. To me, it seems that the standard way of looking at the people changing is just basically looking at the blasts. Can you look at the blasts? Prithviraj showed in the slide set that most people slowly evolve over time and get to 20%. If 20% is a cutoff for calling patients transformed, but the patients are transforming as you pointed out through accelerated phase, that appears to be the time to intervene, not to wait, and that percentage of blasts, 20%, is relatively insignificant in a sense that we don’t need to wait for it. Prithviraj, you pointed out hypomethylating agents as what NCCN [National Comprehensive Cancer Network] Guidelines would be suggesting over chemotherapy. Where would you use chemotherapy? Is there a role for chemotherapy at all in the management of patients with accelerated and blastic phase?
Prithviraj Bose, MD: I think the role is very limited. Again, it’s in your young, fit patient where you have a donor lined up and where you know that transplant is your goal. That’s where the role is. It’s there, but it’s very small.
Srdan Verstovsek, MD, PhD: The JAK2 inhibitors were mentioned as well. Andrew, we use them to control the signs and symptoms, and we utilize perhaps 10 mg twice a day of ruxolitinib. And we stick with it, to control what we can, for quality of life. What’s your take on that? Is that how it should be done?
Andrew Kuykendall, MD: In general, we do that. But it’s a case-by-case basis. Certainly I don’t think the JAK inhibitor is there to induce remissions, and it’s unclear if it provides any role in that. Early on, ruxolitinib was looked at in AML [acute myeloid leukemia], and I think there was less than a handful of responders. But in general, it’s there to improve the symptoms related to the disease. Because as Rami mentioned, this is a molecularly complex disease that has subclones that are responsible for the progression to blast phase and AML, but certainly the clones that are there that led to the constitutional symptoms and splenomegaly are still there, and those are still symptoms that the patient has to deal with despite receiving treatment for acute leukemia. So we typically do that as well. We try to keep that on board in patients who were either benefiting from that previously or have signs or symptoms that would suggest they would benefit from that. We certainly titrate that up and down to manage the cytopenias that come along with that to maximize the patient quality of life.
Srdan Verstovsek, MD, PhD: This is really a good summary. If I may, it seems to me that the theme here is earlier intervention, either thinking with a bone marrow transplant when the blasts are coming up, or intervening with hypomethylating agents with or without JAK inhibitors, and not to wait for 20% blasts because most people will slowly transition. So we need earlier intervention, something akin of early intervention in myelofibrosis, not to wait for them to be high risk and in bad shape. Same kind of thoughts here with the transformation process.
Thank you very much for this lovely discussion. It was very valuable for our participants. Thank you for your thoughts and ideas how to manage this terrible condition when people are transforming.
Srdan Verstovsek, MD, PhD: I’d like to thank our panel for your case presentations and for a lively, informative discussion. It was a real pleasure. Thank you very much. To our viewing audience, thank you for joining us for this Targeted Oncology® Virtual Tumor Board® presentation. We hope today’s discussion was a valuable use of your time and that you acquired some practical knowledge that you can take back to your clinic. Thank you, and have a good day.
Transcript edited for clarity.