Case 2: The RESPONSE Trial of Ruxolitinib for PV
Episodes in this series

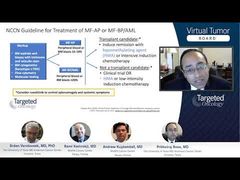
Andrew Kuykendall, MD: We’ll talk a little bit about ruxolitinib, which I mentioned is an option in the second-line setting in polycythemia vera, and this was based on the RESPONSE trial, which is a phase 3 trial of patients with polycythemia vera, requiring phlebotomies who had previously been on hydroxyurea. Patients were randomized 1-to-1 to either ruxolitinib or standard therapy, and this is a study that now has 5-year data that’s been presented and published recently.
The primary end point of this study was a composite end point between hematocrit control and spleen response. As you can see here, about 21% of patients in the ruxolitinib group were able to achieve both hematocrit control and spleen response. Broken down into spleen response and hematocrit control, you can see that those percentages are both superior to standard therapy, with 40% of patients being able to achieve a spleen response and 60% of patients being able to achieve hematocrit control. Always a key end point in myeloproliferative disease trials is symptom reduction. You can see here that ruxolitinib showed very impressive symptom benefit looking at it as a whole, with a 14-point symptom score on the MPN-SAF [Myeloproliferative Neoplasm Symptom Assessment Form],as well as clustered into cytokine clusters, hyperviscosity, and splenomegaly.
As I mentioned, this has long-term results that have been published recently. These were presented in 2019 and published earlier this year. These are the 5-year data in the RESPONSE study, and what you can see here is the response rates were impressive, but also the durability of response is impressive here. At 224 weeks, it was expected that over 70% of patients would be able to maintain the hematocrit control and maintain the spleen volume reduction. Looking at complete hematologic response, this was also maintained in over 50% of patients at the 224-week mark.
I’d be remiss to not mention the RESPONSE-2. The RESPONSE study enrolled patients that had large spleens, and so the RESPONSE-2 was an answer to the question of whether or not this was a drug that was predominantly good for patients who had enlarged spleens, and the answer unequivocally was yes. You can see here in the RESPONSE-2, patients without splenomegaly were able to achieve a very similar degree of hematocrit control and complete hematologic response as well as symptom improvements, compared to what was seen in the RESPONSE setting, and that was superior to what was seen with best available therapy. So, this is not a drug that is just for those with enlarged spleens, as it is able to give benefit to those who did not have splenomegaly as well.
Interestingly, we think about the toll that phlebotomies take on a patient, and by repeatedly phlebotomizing patients, we make them iron deficient, and that comes with a whole set of symptoms that can be quite debilitating and problematic for patients. So it’s challenging when that’s the 1 treatment option that we offer, is to solve 1 problem, but creates others.
So, this is an interesting study looking at the effect of ruxolitinib therapy on iron markers and iron deficiencies, and if you focus on the MCV [corpuscular volume], these are broken down into 4 lines and it looks at patients who started with normal and abnormal iron levels, whether or not they were on ruxolitinib or best available therapy. But you can see in the patients that had low MCVs and low iron saturations, suggestive of significant iron deficiency, those numbers improved over time while on the ruxolitinib therapy to a degree not seen in the best available therapy arm.
Then looking at the exposure-adjusted rates of common adverse effects with ruxolitinib, you can see in the polycythemia vera setting, the rates of anemia and thrombocytopenia are quite low. While there are many different adverse effects listed here, the rates of grade 3/4 adverse effects are quite low, but we need to mention that there is an increased risk of herpes zoster reactivation or shingles reactivation that’s seen with ruxolitinib at a higher rate than what was seen with the best available therapy. That’s certainly something that we counsel patients on and talk to them about—especially the need for the new shingles vaccine and keeping an eye out for this potentiality.
While the primary end point or secondary was not really included, there are thromboembolic events that were not really included as a primary end point within the study. That is often the goal of treatment in patients with polycythemia vera—to prevent further thromboembolic events. But looking at this, you can see that the rates of thromboembolic events with ruxolitinib were quite low, occurring in 1% of patients, compared to a higher rate that was seen with best available therapy. And then the patients that crossed over from best available therapy over to the ruxolitinib arm, the rate was quite low as well. So, I think that’s very reassuring to know that this is often what we’re trying to prevent from happening, and that while not included as a primary end point, this was something that was seen in a very small amount of patients.
As far as safety goes, we worry about the potential for secondary malignancies in patients who are treated with ruxolitinib, and here you can see that the rate of this was about 7 per 100 patient-years in patients originally randomized to ruxolitinib. That was a little bit more than what we saw with best available therapy, where that rate was 4.1 per 100 patient-years, and a little bit in between that in the crossover group. And also, the rates of nonmelanoma skin cancer were 5.1 in the original patients who were randomized to ruxolitinib, and that was about double than what was seen with the best available therapy arm as well. So, while I know the 4 of us are in Texas and Florida, we always recommend that patients see their dermatologists pretty regularly. But this is something to counsel patients on while they are on ruxolitinib.
Transcript edited for clarity.